PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1403971

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1403971
Interventional Neurology Devices - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts 2024 - 2029
The interventional neurology devices market is anticipated to register a CAGR of 8.97% over the forecast period.
COVID-19 significantly impacted people with brain-related disorders due to limitations in the diagnostic and therapeutic processes that influenced ongoing research activities. For example, according to a study published in February 2021 in PubMed, patients with severe COVID-19 demonstrated a higher D-dimer level, suggesting an altered coagulation system state. Furthermore, the ICU-admitted COVID-19 patients showed significant thrombotic complications, including acute pulmonary embolism, ischemic stroke, deep-vein thrombosis, and systemic arterial embolism. Hence, the demand for interventional neurology devices was high during the pandemic, significantly impacting the market growth. The studied market reached its pre-pandemic nature regarding demand for interventional neurology devices as COVID-19-related restrictions were eased. Moreover, the market is believed to be registering significant growth in the coming years due to post-COVID neurological complications and new product approvals and launches.
Factors such as an increase in the incidence of neurological diseases and a rise in the adoption of minimally invasive treatment will likely fuel the market growth. For instance, as per the British Epilepsy Association data published in 2022, there were about 600,000 people with epilepsy living in the UK, around one in every 100 people. Every day, 87 people are expected to be diagnosed with epilepsy. With the high number of people living with epilepsy in the country, the demand for interventional neurology devices is increasing, contributing to market growth. Similarly, according to the 2022 update from the Parkinson's Foundation, nearly one million people in the United States were living with Parkinson's disease (PD). This number is expected to rise to 1.2 million by 2030. Parkinson's is the second-most common neurodegenerative disease after Alzheimer's disease. Hence, the high prevalence of neurological disease is likely to boost the market growth.
Additionally, the recent developments in the market, such as product reimbursement, will lead to significant growth. For instance, in June 2021, Terumo France/Europe and MicroVention Europe received reimbursement for the Roadsaver carotid stent system due to the positive assessment from the French National Authority for Health (HAS), highlighting the device's therapeutic benefit. Such initiatives will boost the market. Similarly, in September 2022, Penumbra, Inc., a global healthcare company focused on innovative therapies, publicized that its RED Reperfusion Catheters secured CE Mark (Conformite Europeenne) and are available in Europe. The catheters are part of the company's Penumbra System. It is a fully integrated mechanical aspiration thrombectomy system to restore blood flow in acute ischemic stroke (AIS) patients. Also, in March 2021, Innovative Cardiovascular Solutions (ICS) reported positive results from European clinical cases using the next generation of the EMBLOK Embolic Protection System in patients undergoing transcatheter aortic valve replacement (TAVR).
Therefore, due to the increase in neurology diseases and a surge in product launches and approvals, the studied market is expected to witness significant growth over the forecast period. However, the strict rules of regulatory bodies for adopting new technologies and the high cost of devices are expected to hinder the market's growth.
Interventional Neurology Devices Market Trends
Neuro Thrombectomy Devices Segment is Expected to Show Significant Growth Over the Forecast Period
A neurothrombectomy device is intended to retrieve or destroy blood clots in the cerebral microvasculature by mechanical, laser, ultrasound technologies, or a combination of technologies. Ischemic acute stroke is associated with side effects and higher health burdens. Neurothrombectomy equipment is recommended for patients receiving treatment that may include limited functionality. The currently available neuro-thrombectomy machines offer interesting treatment options to patients with acute ischemic stroke. The neuro-thrombectomy device segment will likely witness significant growth over the forecast period. It is due to factors such as an increase in neurological diseases and a rise in neuro-thrombectomy device launches coupled with research and development.
Moreover, countries like India are facing the challenge of TBI caused by road accidents, as the number of road accidents in these countries is increasing yearly. For instance, as per the data by National Crime Records Bureau published in August 2022, the number of road accidents in India increased from 3,68,828 in 2020 to 4,22,659 in 2021. Furthermore, other data from the Indian Head Injury Foundation published in its 2021 report stated that over one million people across the country suffer from severe head injuries every year, and around 60% of TBI in India are caused by road accidents. Hence, observing the given statistics, it is believed that increasing road accidents in India causing TBIs will contribute significantly to the growth of neuro-thrombectomy devices over the forecast period.
Furthermore, neurothrombectomy machines offer many potential benefits beyond pharmacologic thrombolysis, including the success of rapid reconstruction, improved efficiency in treating large vessels, and a lower risk of bleeding events. Introducing neurothrombectomy devices will lead to an increase in market share, thus driving the market. For example, in January 2021, Vesalio received the FDA 510k approval and its 4th CE approval to remove a thrombus from nearby blood vessels that expand the stage of neuro-vascular thrombectomy. Similarly, in February 2022, MicroPort NeuroTech Limited received marketing approval from China's National Medical Products Administration (NMPA) for its self-developed Neurohawk Stent Thrombectomy Device (Neurohawk). Neurohawk is a retrievable, self-expandable clot stent retriever used in endovascular invasive thrombectomy procedures to remove large clots in blood vessels.
Hence, with increased neurology diseases, product launches, and several product approvals, the studied segment is expected to grow significantly over the forecast period.
North America Anticipated to Hold a Significant Market Share Over the Forecast Period
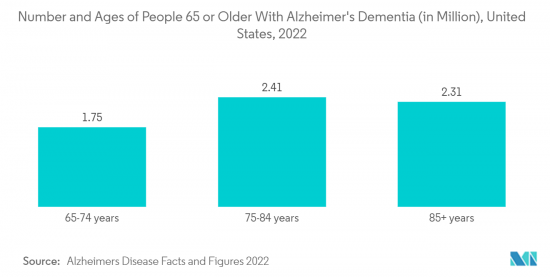
North America is expected to witness significant growth in the studied market owing to factors such as the increasing burden of neurodegenerative diseases like Alzheimer's, Parkinson's disease, etc., product launches, and increasing demand for minimally invasive treatment. For instance, the increasing number of neurological disorders in North America is one of the major drivers for the market. According to the report published by the Brain Injury Association of America in March 2021, around 3.5 million Americans yearly suffer from traumatic brain injuries. The same source also reported that about 2.8 million people sustain traumatic brain injuries annually, with some 280,000 resulting in hospitalizations. Furthermore, in 2022, Heart & Stroke published a new study in the Canadian Journal of Neurological Sciences, which revealed that annual stroke occurrence rates in Canada increased to 108,707, or approximately one every five minutes.
Furthermore, as per the report published by the CDC in January 2022, about 0.6% of children aged 0-17 years had active epilepsy in 2021. The same source also stated that around 470,000 children had active epilepsy and around 3.0 million adults in the United States had active epilepsy in 2021. In addition, the incidence of epilepsy in the United States in seniors is up to 240 per 100,000 per year. Thus, the rising prevalence of epilepsy propels the demand for interventional neurology devices, which will increase market growth.
Additionally, a rise in product launches, strategic activities by the key players, and government initiatives and awareness campaigns are expected to drive market growth. For instance, in October 2022, the Society of Vascular and Interventional Neurology publicized a 40-second stroke awareness challenge. During the challenge, each person was required to create a 40-second video doing as many push-ups as they could safely do. Such awareness challenges make people more cautious about their health and, thereby, driving the market growth.
Therefore, with increased neurology diseases and awareness programs, North America is expected to witness significant growth over the forecast period.
Interventional Neurology Devices Industry Overview
The interventional neurology devices market is fragmented in nature. The global key players are manufacturing the majority of interventional neurology devices. Market leaders with more funds for research and a better distribution system established their position in the market. The key players in the market are Abbott, B Braun SE, Medtronic, Boston Scientific Corporation, and Stryker Corporation.
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope of the Study
2 RESEARCH METHODOLOGY
3 EXECUTIVE SUMMARY
4 MARKET DYNAMICS
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Increasing Incidence of Neurological Disorders
- 4.2.2 Risng Demand for Minimally Invasive Treatment
- 4.3 Market Restraints
- 4.3.1 High Cost of Devices
- 4.3.2 Stringent Regulatory Scenario
- 4.4 Porter's Five Forces Analysis
- 4.4.1 Bargaining Power of Suppliers
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Threat of New Entrants
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5 MARKET SEGMENTATION (Market Size by Value - USD)
- 5.1 By Product Type
- 5.1.1 Cerebral Balloon Angioplasty and Stenting Systems
- 5.1.1.1 Carotid Artery Stents
- 5.1.1.2 Embolic Protection Systems
- 5.1.2 Neurothrombectomy Devices
- 5.1.3 Aneurysm Coiling and Embolization Devices
- 5.1.4 Micro-catheters
- 5.1.5 Guidewires
- 5.1.1 Cerebral Balloon Angioplasty and Stenting Systems
- 5.2 By Application
- 5.2.1 Artery Stenosis
- 5.2.2 Ischemic Strokes
- 5.2.3 Brain Aneurysm
- 5.2.4 Vein Stenosis
- 5.2.5 Others
- 5.3 Geography
- 5.3.1 North America
- 5.3.1.1 United States
- 5.3.1.2 Canada
- 5.3.1.3 Mexico
- 5.3.2 Europe
- 5.3.2.1 Germany
- 5.3.2.2 United Kingdom
- 5.3.2.3 France
- 5.3.2.4 Italy
- 5.3.2.5 Spain
- 5.3.2.6 Rest of Europe
- 5.3.3 Asia Pacific
- 5.3.3.1 China
- 5.3.3.2 Japan
- 5.3.3.3 India
- 5.3.3.4 Australia
- 5.3.3.5 South Korea
- 5.3.3.6 Rest of Asia-Pacific
- 5.3.4 Middle East and Africa
- 5.3.4.1 GCC
- 5.3.4.2 South Africa
- 5.3.4.3 Rest of Middle East and Africa
- 5.3.5 South America
- 5.3.5.1 Brazil
- 5.3.5.2 Argentina
- 5.3.5.3 Rest of South America
- 5.3.1 North America
6 COMPETITIVE LANDSCAPE
- 6.1 Company Profiles
- 6.1.1 Abbott
- 6.1.2 B. Braun SE
- 6.1.3 Boston Scientific Corporation
- 6.1.4 Johnson & Johnson
- 6.1.5 Medtronic
- 6.1.6 Merit Medical Systems.
- 6.1.7 Penumbra, Inc.
- 6.1.8 Stryker Corporation
- 6.1.9 Terumo Corporation
- 6.1.10 Integer Holdings Corporation
- 6.1.11 Canon Inc.
- 6.1.12 MicroPort Scientific Corporation
7 MARKET OPPORTUNITIES AND FUTURE TRENDS




