PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1273425

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1273425
Patient Derived Xenograft Models Market - Growth, Trends, and Forecasts (2023 - 2028)
The patient-derived xenograft models market is expected to register a CAGR of 12.5% over the forecast period.
During the COVID-19 pandemic, cancer research was halted as the majority of studies were focused on developing drugs for COVID-19. However, post-pandemic, the majority of the focus of researchers has been on developing innovative therapies for cancer. This is expected to support the growth of the industry.
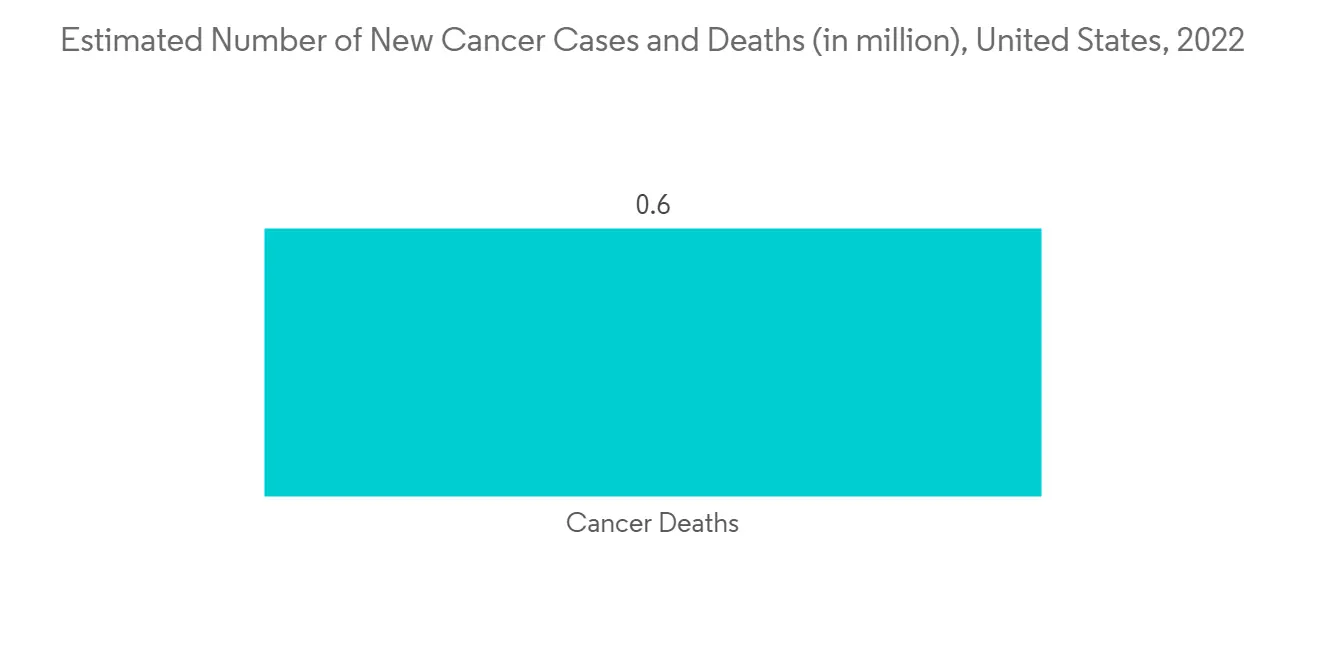
The rising cases of cancer, rising research and development activities in the pharmaceutical industry, and continuous support for cancer research from the public, as well as the private sector, are the major factors driving the market growth. For instance, according to the GLOBOCAN Report 2020, globally, around 19,292,789 new cancer cases have been reported, and 9,958,133 reported cancer deaths. The majorly affecting cancers are breast cancer, lung cancer, colorectum cancer, and prostate cancer.
In addition, the increasing research using mouse models for drug discovery for cancer is also anticipated to contribute to market growth. For example, as per the article published in April 2022 by Oncoimmunology, the researchers utilized the Diversity Outbred (DO) and Collaborative Cross (CC) mouse models to investigate the outcomes of the immune checkpoint inhibitors (ICI) for a variety of malignancies. The researchers of the study purchased the DO mice models from the Jackson Laboratory and all CC mice from the Systems Genetics Core Facility at the University of North Carolina. Such adoption of mice models in cancer research is expected to contribute to market growth over the forecast period.
However, the high cost of personalized patient-derived xenograft models and stringent regulations towards the use of animal models are likely to impede market growth over the forecast period.
Patient Derived Xenograft Models Market Trends
Mice Model Segment is Expected to Witness Growth Over the Forecast Period
The mouse is the leading mammalian model for the study of human disease and human health. The mouse is small, which makes it an economical choice, and it also breeds very well. Scientists have amassed a vast knowledge of mouse physiology, anatomy, and genes from more than 100 years of working with them. Furthermore, scientists can manipulate genes, and the mouse is among the first species of mammals whose genes have been modified by molecular tools. Mouse models are among the most valuable tools in cancer research. Researchers use them for many types of studies, from identifying potential new cancer treatments to finding new clues about cancer biology.
The increasing application of the mice model for cancer treatment and rising investments by private and public sectors for research and development are the major factors propelling the segment's growth. For instance, as per the study published in March 2021, by Cancer Management and Research, a genetically engineered mouse model (GEMM) was designed to induce tumorigenesis by promoting the expression of oncogenes (such as BRAF V600E in melanoma) or deleting tumor suppressor genes (such as PTEN in prostate cancer) through genetic engineering. Therefore, such innovations may induce segment growth.
Furthermore, according to the report published by the European Commission in December 2021, European Union-based companies increased their investments in research and development in health by up to 10.3%. It also reported that companies in the United States and China increased their overall research and development investments by 9.1% and 18.1%, respectively. The United States and China-based companies have expanded their share of research and development in the healthcare sector during the coronavirus crisis, in which the United States increased up to 17.9% and China increased up to 30.7%. Therefore, such a scenario is anticipated to accelerate the research works related to diseases and novel treatments for combatting diseases.
Therefore, owing to the above-mentioned factors, significant segment growth is anticipated over the forecast period.
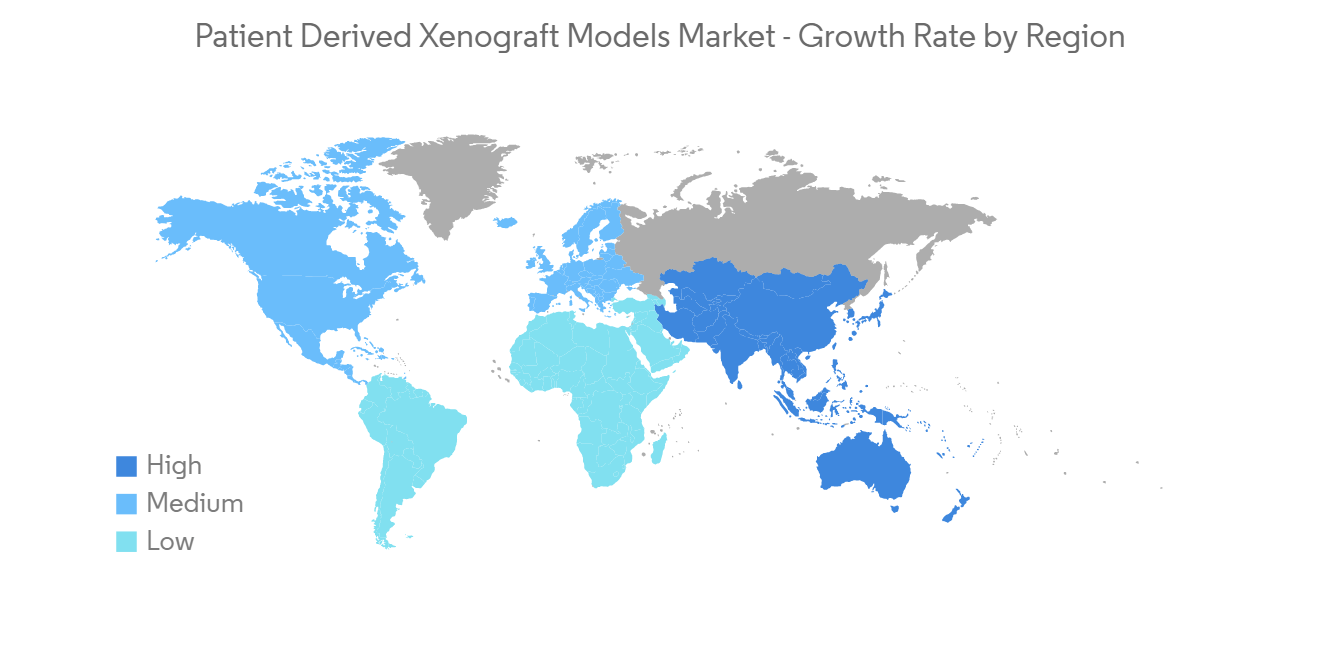
North America is Expected to Dominate the Patient Derived Xenograft Models Market Over the Forecast Period
North America is expected to dominate the overall patient-derived xenograft models market over the forecast period. The growth is due to factors such as the increasing cases of cancer, rising investments by private and public sectors, and the presence of key players in the region. For instance, in May 2022, the Breast Cancer Society of Canada granted USD 350,000 to Precision Oncology Research. The breast cancer research project selected for funding will focus on advances in screening, detection, precision oncology, and patient-reported outcomes, thereby improving the care of Canadian women with breast cancer. Similarly, as per the report published by the WHO in February 2021, it is estimated that around 28,000 Canadians will be diagnosed with breast cancer in 2022. Therefore, such instances are anticipated to propel market growth in this region.
Key product launches, high concentration of market players or manufacturer's presence, and acquisition & partnerships among major players, and rising cases of cancer in the United States are some of the factors driving the growth of the patient-derived xenograft models market in the country. For instance, in March 2022, KIYATEC presented new clinical correlation data at American Association for Cancer Research. KIYATEC's clinical and preclinical technology platforms, 3D Predict and KIYA-PREDICT, respectively, are leaders in functional precision oncology with published evidence of predictive response correlating with clinical outcomes. The six abstracts accepted for presentation at the conference include applications in various tumor types, responses to immunotherapy, and ex vivo application of PDX for preclinical drug development. Therefore, such advancements are anticipated to propel market growth.
Therefore, owing to the aforesaid factors, the growth of the studied market is anticipated in the North America Region.
Patient Derived Xenograft Models Industry Overview
The patient-derived xenograft market is consolidated in nature due to the presence of a few key companies operating globally as well as regionally. The competitive landscape includes an analysis of a few international as well as local companies which hold significant market shares and are well known, including Champions Oncology, Inc., Crown Bioscience, Inc., Charles River Laboratories, Inc., EPO Berlin-Buch GmbH, Hera BioLabs, Oncodesign, Pharmatest Services Ltd, Urolead and Xentech among others.
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope of the Study
2 RESEARCH METHODOLOGY
3 EXECUTIVE SUMMARY
4 MARKET DYNAMICS
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Rising Cases of Cancer
- 4.2.2 Rising R&D Activities in the Pharmaceutical Industry
- 4.2.3 Continuous Support for Cancer Research From Public as Well as Private Sector
- 4.3 Market Restraints
- 4.3.1 High Cost of Personalized Patient Derived Xenograft Models
- 4.3.2 Stringent Regulations Towards Use of Animals Models
- 4.4 Porter Five Forces
- 4.4.1 Threat of New Entrants
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Bargaining Power of Suppliers
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5 MARKET SEGMENTATION (Market Size by Value - USD Million)
- 5.1 By Type
- 5.1.1 Mice Model
- 5.1.2 Rats Model
- 5.2 By Tumor Type
- 5.2.1 Gastrointestinal Tumor Model
- 5.2.2 Gynecological Tumor Model
- 5.2.3 Respiratory Tumor Model
- 5.2.4 Other Tumor Model
- 5.3 By End User
- 5.3.1 Pharmaceutical & Biotechnology Companies
- 5.3.2 Academic & Research Institutions
- 5.3.3 Others
- 5.4 Geography
- 5.4.1 North America
- 5.4.1.1 United States
- 5.4.1.2 Canada
- 5.4.1.3 Mexico
- 5.4.2 Europe
- 5.4.2.1 Germany
- 5.4.2.2 United Kingdom
- 5.4.2.3 France
- 5.4.2.4 Italy
- 5.4.2.5 Spain
- 5.4.2.6 Rest of Europe
- 5.4.3 Asia-Pacific
- 5.4.3.1 China
- 5.4.3.2 Japan
- 5.4.3.3 India
- 5.4.3.4 Australia
- 5.4.3.5 South Korea
- 5.4.3.6 Rest of Asia-Pacific
- 5.4.4 Rest of the World
- 5.4.1 North America
6 COMPETITIVE LANDSCAPE
- 6.1 Company Profiles
- 6.1.1 Champions Oncology, Inc.
- 6.1.2 Charles River Laboratories, Inc.
- 6.1.3 Crown Bioscience, Inc.
- 6.1.4 EPO Berlin-Buch GmbH
- 6.1.5 Hera BioLabs
- 6.1.6 Oncodesign
- 6.1.7 Pharmatest Services Ltd
- 6.1.8 Urolead
- 6.1.9 Xentech
7 MARKET OPPORTUNITIES AND FUTURE TRENDS




