PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1273421

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1273421
Oseltamivir Market - Growth, Trends, and Forecasts (2023 - 2028)
The oseltamivir market is expected to register a CAGR of 3.2% over the forecast period.
COVID-19 has significantly impacted the oseltamivir market. Most COVID-19 patients who were symptomatic during the ongoing coronavirus illness have used oseltamivir. For instance, as per the article published in the Annals of Medical surgery in May 2022, it has been observed that the administration of oseltamivir in COVID-19 patients resulted in a shorter length of hospital stay, earlier recovery and discharge from hospitals and a lower mortality rate. In addition, as per the same source, a lower incidence of ICU hospitalization and mechanical ventilation within the first hours of admission was observed in patients administered with oseltamivir in combination therapy. However, flu activity was unusually low throughout the 2020-2021 flu season, both in the United States and globally, despite high levels of testing. From September 2020 to May 2021 in the United States, only 0.2% of 818,939 respiratory specimens tested by U.S. clinical laboratories were positive for an influenza virus. This decline was largely due to the COVID-19 mitigation measures such as wearing face masks, staying home, hand washing, school closures, reduced travel, increased ventilation of indoor spaces, and physical distancing that contributed to the decline in 2020-2021 flu incidence, hospitalizations, and deaths. Hence, the pandemic has impacted the market during peak times. However, the market started to recover as COVID-19 cases started to decline, along with the removal of mitigation measures.
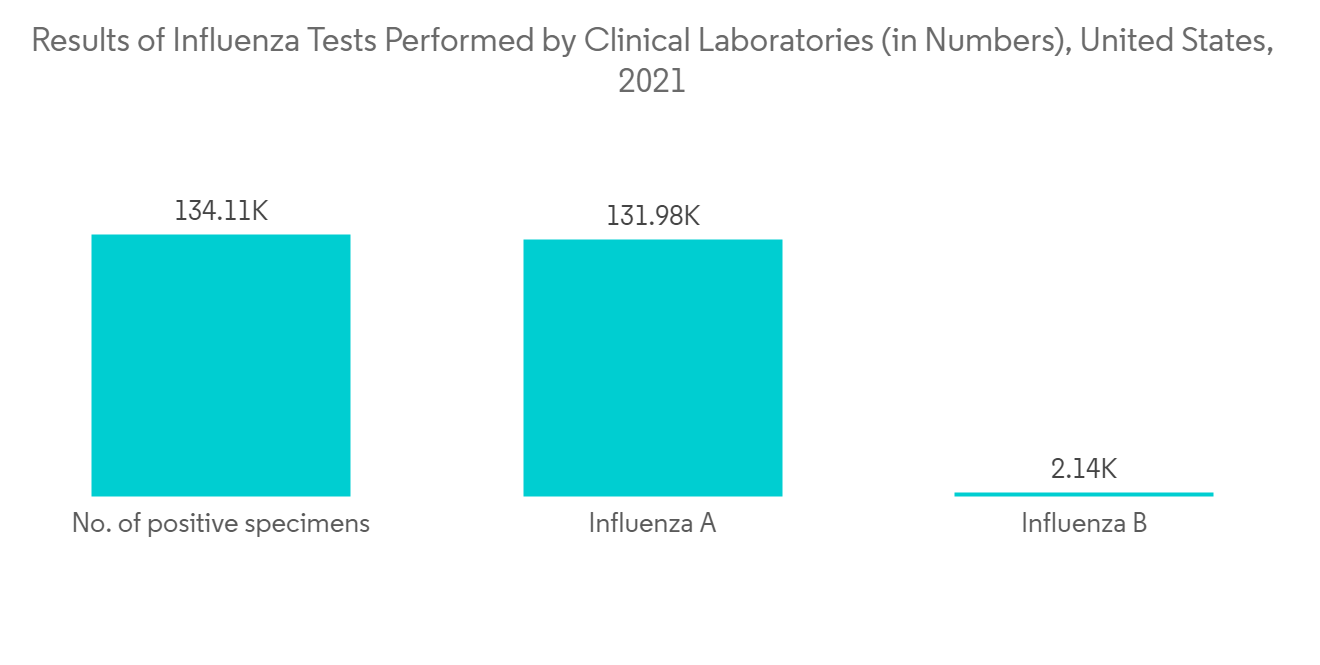
The factors that primarily drive the market are the increasing prevalence of influenza virus infection. Influenza is recognized as a significant cause of morbidity and mortality in humans, prompting the development and implementation of treatment to reduce health and economic burden. For instance, per the data published by CDC, from early October 2021 through mid-June 2022, influenza virus infection resulted in 8-13 million symptomatic illnesses, 3.7-6.1 million medical visits, 82,000-170,000 hospitalizations, and 5,000-14,000 deaths. Clinical laboratories tested more than 2.8 million respiratory specimens for the influenza virus. Overall, 4.5% of specimens tested positive, including 98.6% for influenza A and 1.4% for influenza B. Each week, between 0.1% and 9.9% of the specimens tested positive for influenza.
Furthermore, as per the WHO data published in February 2021, WHO GISRS laboratories tested over 490,516 specimens during 2021-2022, among which 12,368 individuals tested positive for flu, with 8,423 for influenza A and 3,945 for influenza B. Thus, the significant burden of influenza infection among the population is expected to increase the demand for the oseltamivir drug for its treatment, thereby propelling the market growth.
Besides, the introduction of the products is expected to further propel the growth of the market. For instance, in February 2023, Zhongchao Inc. announced that Chongqing Xinjiang Pharmaceutical Co., Ltd. obtained the general distribution rights in Mainland China for the anti-influenza drug from Natco Pharma Limited ("Natco"). The first batch of cargo has arrived at the port in Chongqing City, and the drug will be introduced to the market for domestic treatment and prevention of Type A and Type B influenza viruses after the inspection and quarantine. The anti-influenza drug introduced by Xinjiang Pharmaceutical is Naiditawei, commonly known as Oseltamivir Phosphate Capsules.
However, the side effects associated with oseltamivir treatment are likely to impede the market growth over the forecast period.
Oseltamivir Market Trends
Influenza A Segment Expects to Register a High CAGR Over the Forecast Period
Oseltamivir is used to treat infections brought on by the influenza A virus. Oseltamivir can be used to treat and prevent influenza, as well as to reduce its symptoms. The influenza A segment is expected to witness significant growth in the oseltamivir market over the forecast period due to the factors such as the rising prevalence of influenza A virus infection and the growing company activities in developing medications.
Additionally, from an article published in the Vaccines Journal in May 2022, it has been observed that an estimated 68,077 cases caused by the H3N2 Darwin type A influenza strain were reported in Brazil (between November and December 2021). Furthermore, an article published in ISIRV in September 2021 stated that the infants with influenza treated with oseltamivir were found to have the less viral load in nasopharyngeal secretions and shortened duration and severity of symptoms. Thus, oseltamivir is a well-tolerated drug that reduces the duration of symptomatic illness in patients with naturally acquired influenza, which is anticipated to increase segment growth over the forecast period.
Thus, owing to the factors mentioned above, the segment is expected to grow significantly during the forecast period.
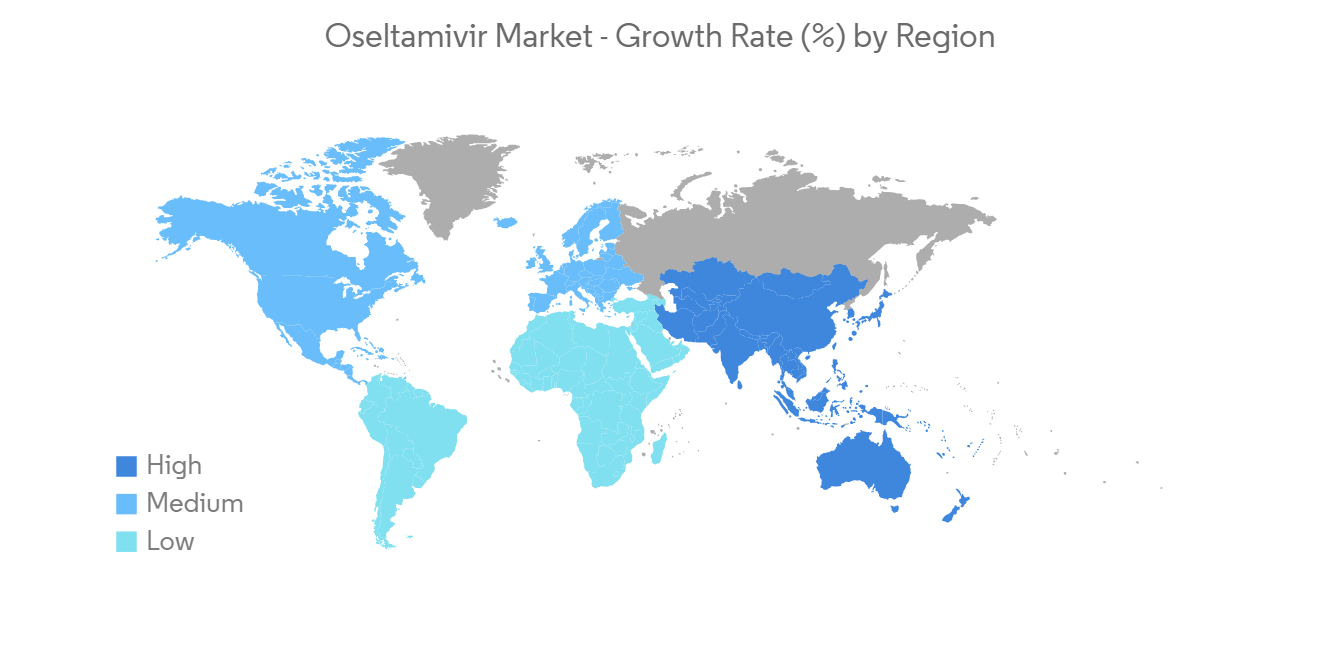
North America is Expected to Hold a Significant Share in the Market during the Forecast Period
North America is expected to hold a major share of the oseltamivir market due to the increasing prevalence of influenza infections, high adoption rates of treatment, presence of established key players, and rising company focus on developing effective drugs for treating influenza infections,
In addition, the high healthcare expenditures in the region are creating opportunities that are expected to support market growth. For instance, according to the OECD data published in June 2022, United States healthcare spending in 2021 was 17.8% of the country's total GDP.
Besides, the rising burden of influenza infections is the key factor driving the market growth. For instance, as per the data published by CDC in February 2023, there were 18,000 flu-related fatalities, 25 million illnesses, and 280,000 hospitalizations occurred in 2022. 54.2% of the influenza cases were influenza A(H3N2), and 45.8% were influenza A cases. (H1N1). Thus, the huge burden of influenza in the country is expected to propel the growth of the market over the forecast period.
Additionally, government initiatives to increase access to oseltamivir are expected to boost market growth. For instance, in December 2022, the U.S. Department of Health and Human Services (HHS), through the Administration for Strategic Preparedness and Response (ASPR), announced that they are making the additional supply of Tamiflu available to jurisdictions to respond to an increased demand for the antiviral during this flu season, including through the Strategic National Stockpile (SNS).
Thus, the market studied is expected to grow significantly during the forecast period due to the aforementioned factors.
Oseltamivir Industry Overview
The oseltamivir market is fragmented and competitive and consists of several major players. In terms of market share, a few of the major players are currently dominating the market, which include F. Hoffmann-La Roche Ltd, Cipla Inc., Zydus Cadila, NATCO Pharma Limited, Macleods Pharmaceuticals Ltd, Amneal Pharmaceuticals LLC, and Alembic Pharmaceuticals Limited.
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope of the Study
2 RESEARCH METHODOLOGY
3 EXECUTIVE SUMMARY
4 MARKET DYNAMICS
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Rising Prevalence of Influenza Virus Infections
- 4.2.2 Increase in the Number of Research and Development Activities
- 4.3 Market Restraints
- 4.3.1 Side Effects Associated with Oseltamivir Treatment
- 4.4 Porter Five Forces
- 4.4.1 Threat of New Entrants
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Bargaining Power of Suppliers
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5 MARKET SEGMENTATION (Market Size by Value - USD million)
- 5.1 By Product Type
- 5.1.1 Capsule
- 5.1.2 Suspension
- 5.1.3 Other Product Types
- 5.2 By Application
- 5.2.1 Influenza A
- 5.2.2 Influenza B
- 5.2.3 Other Applications
- 5.3 Geography
- 5.3.1 North America
- 5.3.1.1 United States
- 5.3.1.2 Canada
- 5.3.1.3 Mexico
- 5.3.2 Europe
- 5.3.2.1 Germany
- 5.3.2.2 United Kingdom
- 5.3.2.3 France
- 5.3.2.4 Italy
- 5.3.2.5 Spain
- 5.3.2.6 Rest of Europe
- 5.3.3 Asia-Pacific
- 5.3.3.1 China
- 5.3.3.2 Japan
- 5.3.3.3 India
- 5.3.3.4 Australia
- 5.3.3.5 South Korea
- 5.3.3.6 Rest of Asia-Pacific
- 5.3.4 Middle East and Africa
- 5.3.4.1 GCC
- 5.3.4.2 South Africa
- 5.3.4.3 Rest of Middle East and Africa
- 5.3.5 South America
- 5.3.5.1 Brazil
- 5.3.5.2 Argentina
- 5.3.5.3 Rest of South America
- 5.3.1 North America
6 COMPETITIVE LANDSCAPE
- 6.1 Company Profiles
- 6.1.1 F. Hoffmann-La Roche Ltd
- 6.1.2 Cipla Inc.
- 6.1.3 Zydus Cadila
- 6.1.4 NATCO Pharma Limited
- 6.1.5 Macleods Pharmaceuticals Ltd
- 6.1.6 Amneal Pharmaceuticals LLC
- 6.1.7 Alembic Pharmaceuticals Limited
- 6.1.8 Hetero Healthcare Limited
- 6.1.9 Lupin Limited
- 6.1.10 Strides Pharma Science Limited
- 6.1.11 Gilead Sciences, Inc.
7 MARKET OPPORTUNITIES AND FUTURE TRENDS




