PUBLISHER: Coherent Market Insights | PRODUCT CODE: 1378566

PUBLISHER: Coherent Market Insights | PRODUCT CODE: 1378566
U.S. Intravenous Solutions Market, By Solution Type (Saline, Dextran, Lactated Ringers, Amino Acid, Vitamins & Minerals, Heparin and trace elements, Mixed Solutions), Packaging (Ampules, Vials, Cartridges, Bottles, Bags), Application, End User
The U.S. Intravenous Solutions Market is estimated to be valued at US$ 3,171.1 Mn in 2023 and is expected to exhibit a CAGR of 7.2% during the forecast period (2023-2030).
| Report Coverage | Report Details | ||
|---|---|---|---|
| Base Year: | 2022 | Market Size in 2023: | US$ 3,171.1 Mn |
| Historical Data for: | 2018 to 2021 | Forecast Period: | 2023 - 2030 |
| Forecast Period 2023 to 2030 CAGR: | 7.20% | 2030 Value Projection: | US$ 5,165.3 Mn |
Intravenous solution is given to patients to replenish the water, sugar, and salt levels in their body in case of an operation or when an individual can't eat or drink normally. Intravenous solutions are also known as intravenous fluids. Intravenous solutions are of the following types: saline, dextran, lactated Ringer's, amino acids, vitamins and minerals, heparin and trace elements, and mixed solutions. Intravenous (IV) solutions play a crucial role in modern medicine, providing a means to deliver fluids, electrolytes, medications, and nutrients directly into a patient's bloodstream. These solutions are administered through a vein using a sterile IV line and are employed for various medical purposes. One of the primary functions of IV solutions is to maintain a patient's hydration and electrolyte balance. This is particularly important for individuals who are unable to consume fluids orally due to illness, surgery, or other medical conditions. IV solutions can help prevent dehydration, which can lead to serious complications. IV solutions are administered using IV sets, which consist of tubing, a needle or catheter, and a bag or bottle containing the solution. The rate of administration is carefully controlled to ensure the patient receives the prescribed amount.
Market Dynamics
The key players in the market are focused on organic strategies, such as the product approval of intravenous (IV) formulations, which is expected to drive growth in the U.S. intravenous solutions market over the forecast period. For instance, in May 2022, SIGA Technologies, Inc., a commercial-stage pharmaceutical company, announced that the U.S. Food and Drug Administration (FDA) had approved the intravenous (IV) formulation of TPOXX for the treatment of smallpox. For patients who are unable to ingest TPOXX oral pills, the IV formulation offers a significant alternative.
Key features of the study:
- This report provides an in-depth analysis of the U.S. intravenous solutions market and provides market size (US$ Mn) and compound annual growth rate (CAGR) for the forecast period (2023-2030), considering 2022 as the base year
- It elucidates potential revenue opportunities across different segments and explains attractive investment proposition matrices for this market.
- This study also provides key insights about market drivers, restraints, opportunities, new product launches or approval, market trends, regional outlook, and competitive strategies adopted by key players.
- It profiles key players in the U.S. intravenous solutions market based on the following parameters- company highlights, products portfolio, key highlights, financial performance, and strategies.
- Key companies covered as a part of this study Baxter International Inc., ICU Medical, Inc., Braun Melsungen Ag and Grifols, S.A.
- Insights from this report would allow marketers and the management authorities of the companies to make informed decisions regarding their future product launches, type up-gradation, market expansion, and marketing tactics
- U.S. intravenous solutions market report caters to various stakeholders in this industry including investors, suppliers, product manufacturers, distributors, new entrants, and financial analysts
- Stakeholders would have ease in decision-making through various strategy matrices used in analyzing the U.S. intravenous solutions market.
Detailed Segmentation:
- By Solution Type:
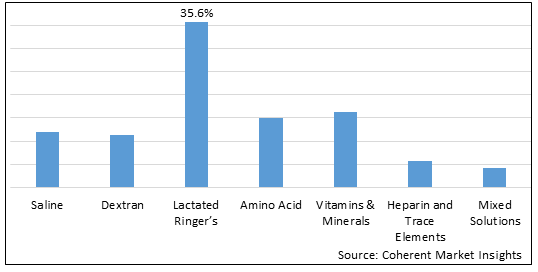
- Saline
- Normal Saline (0.9% NaCl)
- Hypertonic Solution
- Dextran
- D5W 5% dextrose in water
- Analog Hypertonic Saline
- Lactated Ringer's
- Amino Acid
- Vitamins & Minerals
- Heparin and trace elements
- Mixed Solutions
- By Packaging:
- Ampules
- Vials
- Cartridges
- Bottles
- Bags
- Large Volume Bags (greater than 250ml)
- Small Volume Bags (less than 250ml)
- By Application:
- Basic IV Solution
- Nutritional IV Solution
- Blood IV Solution
- Drug IV Solution
- Irrigation IV Solution
- By End User:
- Hospital
- Clinics
- Ambulatory Surgical Centers
- Home Care Settings
- Others
- Company Profiles
- Baxter International Inc.
- ICU Medical, Inc.
- Braun Melsungen Ag
- Grifols, S.A.
Table of Contents
1. Research Objective and Assumption
- Research Objectives
- Assumptions
- Abbreviations
2. Market Purview
- Report Description
- Market Definition and Scope
- Executive Summary
- Market Snippet, By Solution Type
- Market Snippet, By Packaging
- Market Snippet, By Application
- Market Snippet, By End User
- Market Snippet, By Region
- Coherent Opportunity Map (COM)
3. Market Dynamics, Regulations, and Trends Analysis
- Market Dynamics
- Drivers
- Restraints
- Market Opportunities
- Impact Analysis
- Market Trends
- Key Developments
- Regulatory Landscape
- Epidemiology Analysis
- Porter's Five Forces model
4. U.S. Intravenous Solutions Market- COVID-19 Impact Analysis
- Economic Impact
- COVID-19 Epidemiology
- Impact on Supply and Demand
5. U.S. Intravenous Solutions Market, By Solution Type, 2018-2030, (US$ Mn)
- Introduction
- Market Share Analysis, 2023 and 2030 (%)
- Y-o-Y Growth Analysis, 2018-2030
- Segment Trends
- Saline
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- Normal Saline (0.9% NaCl)
- Hypertonic Saline
- Dextran
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- D5W 5% dextrose in water
- Analog Hypertonic Saline
- Lactated Ringer's
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- Amino Acid
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- Vitamins & Minerals
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- Heparin and trace elements
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- Mixed Solutions
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
6. U.S. Intravenous Solutions Market, By Packaging, 2018 - 2030, (US$ Mn)
- Introduction
- Market Share Analysis, 2023 and 2030 (%)
- Y-o-Y Growth Analysis, 2018-2030
- Segment Trends
- Ampules
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- Vials
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- Cartridges
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- Bottles
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- Bags
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- Large Volume Bags (greater than 250ml)
- Small Volume Bags (less than 250ml)
7. U.S. Intravenous Solutions Market, By Application, 2018 - 2030, (US$ Mn)
- Introduction
- Market Share Analysis, 2023 and 2030 (%)
- Y-o-Y Growth Analysis, 2018-2030
- Basic IV Solution
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- Nutritional IV Solution
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- Blood IV Solution
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- Drug IV Solution
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- Irrigation IV Solution
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
8. U.S. Intravenous Solutions Market, By End User, 2018 - 2030, (US$ Mn)
- Introduction
- Market Share Analysis, 2023 and 2030 (%)
- Y-o-Y Growth Analysis, 2018-2030
- Segment Trends
- Hospital
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- Clinics
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- Ambulatory Surgical Centers
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- Home Care Settings
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
- Others
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Mn)
9. Competitive Landscape
- Company Profiles
- Baxter International Inc.
- Company Overview
- Product Portfolio
- Key Highlights
- Financial Overview
- Strategies
- ICU Medical, Inc.
- Company Overview
- Product Portfolio
- Key Highlights
- Financial Overview
- Strategies
- Braun Melsungen Ag
- Company Overview
- Product Portfolio
- Key Highlights
- Financial Overview
- Strategies
- Grifols, S.A.
- Company Overview
- Product Portfolio
- Key Highlights
- Financial Overview
- Strategies
10. Section
- References
- Research Methodology
- About us and Sales Contact




