PUBLISHER: Coherent Market Insights | PRODUCT CODE: 1378568

PUBLISHER: Coherent Market Insights | PRODUCT CODE: 1378568
Small Molecule Prefilled Syringes Market, By Type, By Material Type, By Application (Diabetes, Cancer, Opthalmology, Anaphylaxis, Rheumatoid Arthritis and Others ), By Distribution Channel, By Region
Global small molecule prefilled syringes market is estimated to be valued at US$ 18.98 Bn in 2023, and is expected to exhibit a CAGR of 5.2% during the forecast period (2023-2030).
| Report Coverage | Report Details | ||
|---|---|---|---|
| Base Year: | 2022 | Market Size in 2023: | US$ 18.98 Bn |
| Historical Data for: | 2018 to 2021 | Forecast Period: | 2023 - 2030 |
| Forecast Period 2023 to 2030 CAGR: | 5.20% | 2030 Value Projection: | US$ 27.06 Bn |
A disposable needle-based injection system with a pre-filled syringe that has a specific medication already inside is known as prefilled syringe (PFS). A prefilled glass syringe is frequently used in auto-injectors to hold and administer drugs. In order to ensure the integrity and functionality of the syringe system, proper containment and drug expulsion, and the highest level of patient comfort, various components of the prefilled syringes must be tested using a variety of standard tests. There are different types of prefilled syringes (PFS) that includes glass or plastic, with or without needle. Glass prefilled syringe represents the most experience in the industry and continues to supply the majority of prefilled syringe packaging. The PFS has a multi-component complexity along with, in certain cases, life-saving benefits. It consists of a barrel, a needle, a needle shield, a plunger, and a closure, all of which must work in unison and safely with the pre-filled medicine. In order to maintain a sterile barrier against potential contaminants, the container closure integrity (CCI), which refers tothe appropriateness of the syringe closure mechanismis also crucial. So,all PFS versions are tightly governed by international standards and put to testing.
Market Dynamics
Increasing product approvals by government regulatory authority in the market is expected to drive growth of the global small molecule prefilled syringes market over the forecast period. For instance, in January 2022, Owen Mumford Pharmaceutical Services, division of Owen Mumford Ltd., a medical device manufacturer, announced that its UniSafe 1mL safety device for pre-filled syringes had been approved as a combination product in Asia Pacific. UniSafe 1ml had also received regulatory approval in Europe and is also available in the market as a combination product with a drug to treat rheumatoid arthritis. UniSafe 1ml is a prefilled syringe safety device that offers numerous benefits to both pharmaceutical companies and their patients due to its unique, spring-free, patented design.
Key features of the study:
- The report provides an in-depth analysis of the global small molecule prefilled syringes market and provides market size (US$ Bn) and compound annual growth rate (CAGR) for the forecast period (2023-2030), considering 2022 as the base year
- It elucidates potential revenue opportunities across different segments and explains attractive investment proposition matrices for the market
- This study also provides key insights about market drivers, restraints, opportunities, new product launches or approval, market trends, regional outlook, and competitive strategies adopted by key players
- It profiles key players in the global small molecule prefilled syringes market based on the following parameters- company highlights, products portfolio, key highlights, financial performance, and strategies
- Key companies covered as a part of the study include BD, Cytiva, Merck KGaA, Gaplast, Ascendia Pharmaceuticals, Sanofi, Viatris Inc. (Mylan N.V.), Pfizer Inc., Dr. Reddy's Laboratories Ltd., Fresenius Kabi AG, and McKesson Corporation
- Insights from the report would allow marketers and the management authorities of the companies to make informed decisions regarding their future product launches, type up-gradation, market expansion, and marketing tactics
- Global small molecule prefilled syringes market report caters to various stakeholders in this industry including investors, suppliers, product manufacturers, distributors, new entrants, and financial analysts
- Stakeholders would have ease in decision-making through various strategy matrices used in analyzing the global small molecule prefilled syringes market
Global Small Molecule Prefilled Syringes Market Detailed Segmentation:
- By Type:
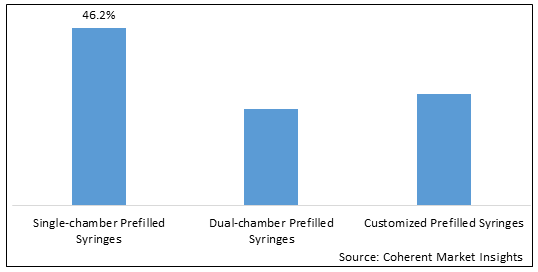
- Single-chamber Prefilled Syringes
- Dual-chamber Prefilled Syringes
- Customized Prefilled Syringes
- By Material Type:
- Glass
- Plastic & Polymer
- By Application:
- Diabetes
- Cancer
- Opthalmology
- Anaphylaxis
- Rheumatoid Arthritis
- Others (Thrombosis, etc.)
- By Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- By Region:
- North America
- Latin America
- Europe
- Asia Pacific
- Middle East
- Africa
- Company Profiles
- BD
- Cytiva
- Merck KGaA
- Gaplast
- Ascendia Pharmaceuticals
- Sanofi
- Viatris Inc. (Mylan N.V.)
- Pfizer Inc.
- Dr. Reddy's Laboratories Ltd.
- Fresenius Kabi AG
- McKesson Corporation
Table of Contents
1. Research Objective and Assumption
- Research Objectives
- Assumptions
- Abbreviations
2. Market Purview
- Report Description
- Market Definition and Scope
- Executive Summary
- Market Snippet, By Type
- Market Snippet, By Material Type
- Market Snippet, By Application
- Market Snippet, By Distribution Channel
- Market Snippet, By Region
- Coherent Opportunity Map (COM)
3. Market Dynamics, Regulations, and Trends Analysis
- Market Dynamics
- Drivers
- Restraints
- Opportunities
- Market Trends
- Mergers, Acquisitions, and Collaborations
- Regulatory Scenario
- Key Developments
- PEST Analysis
- PORTER'S Analysis
- Industry Trends
- Product Launches and Approvals
4. Global Small Molecule Prefilled Syringes Market - COVID-19 Impact Analysis
- Economic Impact
- COVID-19 Epidemiology
- Impact on Supply and Demand
5. Global Small Molecule Prefilled Syringes Market, By Type, 2018- 2030, (US$ Bn)
- Introduction
- Market Share Analysis, 2023 and 2030 (%)
- Y-o-Y Growth Analysis, 2018-2030
- Segment Trends
- Single-chamber Prefilled Syringes
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Segment Trends
- Dual-chamber Prefilled Syringes
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Segment Trends
- Customized Prefilled Syringes
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Segment Trends
6. Global Small Molecule Prefilled Syringes Market, By Material Type, 2018- 2030, (US$ Bn)
- Introduction
- Market Share Analysis, 2023 and 2030 (%)
- Y-o-Y Growth Analysis, 2018-2030
- Segment Trends
- Glass
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Segment Trends
- Plastic & Polymer
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Segment Trends
7. Global Small Molecule Prefilled Syringes Market, By Application, 2018 - 2030, (US$ Bn)
- Introduction
- Market Share Analysis, 2023 and 2030 (%)
- Y-o-Y Growth Analysis, 2018-2030
- Segment Trends
- Diabetes
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Segment Trends
- Cancer
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Segment Trends
- Opthalmology
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Segment Trends
- Anaphylaxis
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Segment Trends
- Rheumatoid Arthritis
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Segment Trends
- Others (Thrombosis, etc.)
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Segment Trends
8. Global Small Molecule Prefilled Syringes Market, By Distribution Channel, 2018 - 2030, (US$ Bn)
- Introduction
- Market Share Analysis, 2023 and 2030 (%)
- Y-o-Y Growth Analysis, 2018-2030
- Segment Trends
- Hospital Pharmacies
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Retail Pharmacies
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Online Pharmacies
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
9. Global Small Molecule Prefilled Syringes Market, By Region, 2018 - 2030, (US$ Bn)
- Introduction
- Market Share Analysis, 2023 and 2030 (%)
- Y-o-Y Growth Analysis, 2018-2030
- Segment Trends
- North America
- Market Size and Forecast, and Y-o-Y Growth, By Type, 2018 -2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Material Type, 2018 -2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Application, 2018 -2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Distribution Channel, 2018-2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2018 -2030, (US$ Bn)
- U.S.
- Canada
- Europe
- Market Size and Forecast, and Y-o-Y Growth, By Type, 2018 -2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Material Type, 2018 -2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Application, 2018 -2030, (US$ Bn)
- Market Size and Forecast, andY-o-Y Growth, By Distribution Channel, 2018-2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2018 -2030, (US$ Bn)
- U.K.
- Germany
- Italy
- France
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- Market Size and Forecast, and Y-o-Y Growth, By Type, 2018 -2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Material Type, 2018 -2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Application, 2018 -2030, (US$ Bn)
- Market Size and Forecast,Y-o-Y Growth, By Distribution Channel, 2018-2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2018 -2030, (US$ Bn)
- China
- India
- Japan
- ASEAN
- Australia
- South Korea
- Rest of Asia Pacific
- Latin America
- Market Size and Forecast, and Y-o-Y Growth, By Type, 2018 -2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Material Type, 2018 -2030, (US$ Bn)
- Market Size and Forecast, Y-o-Y Growth, By Application, 2018 -2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Distribution Channel, 2018-2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2018 -2030, (US$ Bn)
- Brazil
- Mexico
- Argentina
- Rest of Latin America
- Middle East
- Market Size and Forecast, and Y-o-Y Growth, By Type, 2018 -2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Material Type, 2018 -2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Application, 2018 -2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Distribution Channel, 2018-2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2018 -2030, (US$ Bn)
- GCC
- Israel
- Rest of Middle East
- Africa
- Market Size and Forecast, and Y-o-Y Growth, By Type, 2018 -2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Material Type, 2018 -2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Application, 2018 -2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Distribution Channel, 2018-2030, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Country/Sub-region, 2018 -2030, (US$ Bn)
- North Africa
- Central Africa
- South Africa
10. Competitive Landscape
- Company Profiles
- BD
- Company Overview
- Product Portfolio
- Key Highlights
- Financial Overview
- Strategies
- Cytiva
- Company Overview
- Product Portfolio
- Key Highlights
- Financial Overview
- Strategies
- Merck KGaA
- Company Overview
- Product Portfolio
- Key Highlights
- Financial Overview
- Strategies
- Gaplast
- Company Overview
- Product Portfolio
- Key Highlights
- Financial Overview
- Strategies
- Ascendia Pharmaceuticals
- Company Overview
- Product Portfolio
- Key Highlights
- Financial Overview
- Strategies
- Sanofi
- Company Overview
- Product Portfolio
- Key Highlights
- Financial Overview
- Strategies
- Viatris Inc. (Mylan N.V.)
- Company Overview
- Product Portfolio
- Key Highlights
- Financial Overview
- Strategies
- Pfizer Inc.
- Company Overview
- Product Portfolio
- Key Highlights
- Financial Overview
- Strategies
- Dr. Reddy's Laboratories Ltd.
- Company Overview
- Product Portfolio
- Key Highlights
- Financial Overview
- Strategies
- Fresenius Kabi AG
- Company Overview
- Product Portfolio
- Key Highlights
- Financial Overview
- Strategies
- McKesson Corporation
- Company Overview
- Product Portfolio
- Key Highlights
- Financial Overview
- Strategies
- Analysts' Views
11. Section
- References
- Research Methodology
- About us and Sales Contact




