PUBLISHER: Coherent Market Insights | PRODUCT CODE: 1392121

PUBLISHER: Coherent Market Insights | PRODUCT CODE: 1392121
Myeloproliferative Neoplasms Treatment Market, By Disease Type, By Drug Class, By Distribution Channel, By Geography
The Global Myeloproliferative Neoplasms Treatment Market is estimated to be valued at US$ 9.4 Billion in 2023 and is expected to exhibit a CAGR of 3.9% during the forecast period (2023-2030).
| Report Coverage | Report Details | ||
|---|---|---|---|
| Base Year: | 2022 | Market Size in 2023: | US$ 9.4 Bn |
| Historical Data for: | 2018 to 2021 | Forecast Period: | 2023 - 2030 |
| Forecast Period 2023 to 2030 CAGR: | 3.90% | 2030 Value Projection: | US$ 12.28 Bn |
Myeloproliferative neoplasms are a group of rare disorders of the bone marrow that cause an increase in the number of blood cells. The symptoms associated with Myeloproliferative Neoplasms include frequent headaches, tiredness, bruising or unusual bleeding, problem with eyes - such as blurred vision, ringing in the ears, getting more infectious than usual. Moreover, the diagnosis involves blood test to diagnose myeloproliferative neoplasms, the blood tests show the number of blood cells, and can look for genetic mutations. Also, there are other test which done like a bone marrow test, an ultrasound of abdomen.
Different myeloproliferative disorders affect different blood cells that form in the bone marrow. The bone marrow is the soft inner part of bones that makes the blood cells. There are number of different types of myeloproliferative disorders exist, which are;
- Chronic myeloid leukaemia (CML)
- Polycythaemia vera
- Essential thrombocythaemia
- Primary myelofibrosis
Market Dynamics
The key players in the myeloproliferative neoplasm treatment industry are focusing on research and development to develop innovative treatment for myeloproliferative neoplasm. Thus, an increase in research and developement activities in the market is expected to drive the global myeloproliferative neoplasms treatement market growth over the forecast period. For instance, on February 23, 2023, PharmaEssentia USA Corporation, a subsidiary of PharmaEssentia Corporation, a global biopharmaceutical innovator based in Taiwan leveraging deep expertise and proven scientific principles to deliver new biologics in hematology and oncology, announced that the first patient is now being dosed in a phase 2b clinical study evaluating ropeginterferon alfa-2b-njft for the treatment of adults with essential thrombocythemia (ET). BESREMi (ropeginterferon alfa-2b-njft) is innovative monopegylated, long-acting interferon. With its unique pegylation technology, BESREMi has a long duration of activity in the body and is aimed to be administered once every two weeks (or every four weeks with hematological stability for at least one year), allowing flexible dosing that helps meet the individual needs of patients.
Key features of the study:
- This report provides an in-depth analysis of the global myeloproliferative neoplasms treatment market and provides market size (US$ Billion) and compound annual growth rate (CAGR) for the forecast period (2023-2030), considering 2022 as the base year. It elucidates potential revenue opportunities across different segments and explains attractive investment proposition matrices for this market.
- This study also provides key insights about market drivers, restraints, opportunities, new product launches or approval, market trends, regional outlook, and competitive strategies adopted by key players
- It profiles key players in the global myeloproliferative neoplasms treatment market based on the following parameters- company highlights, products portfolio, key highlights, financial performance, and strategies.
- Key companies covered as a part of this study include Novartis AG, PharmaEssentia Corporation, Bristol Myers Squibb, Pfizer, Inc., Takeda Pharmaceutical Company Limited, Incyte, Johnson & Johnson, Teva Pharmaceutical Industries Ltd, F. Hoffmann-La Roche AG, Gamida Cell, Sierra Oncology, Inc, CTI BioPharma Corp, Imago BioSciences, AbbVie Inc, and Constellation Pharmaceuticals
- Insights from this report would allow marketers and the management authorities of the companies to make informed decisions regarding their future product launches, type up-gradation, market expansion, and marketing tactics
- Global myeloproliferative neoplasms treatment market report caters to various stakeholders in this industry including investors, suppliers, product manufacturers, distributors, new entrants, and financial analysts
- Stakeholders would have ease in decision-making through various strategy matrices used in analyzing the global myeloproliferative neoplasms treatment
Detailed Segmentation:
- By Disease Type:
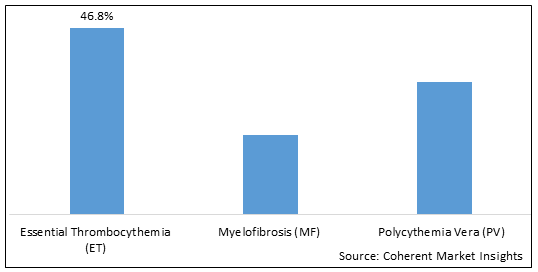
- Essential Thrombocythemia (ET)
- Myelofibrosis (MF)
- Polycythemia Vera (PV)
- By Drug Class:
- Tyrosine Kinase Inhibitors
- Janus kinase inhibitors (JAKi)
- Antimetabolites
- Others (alkylating agents, etc.)
- By Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- By Region:
- North America
- Latin America
- Europe
- Asia Pacific
- Middle East
- Africa
- Company Profiles
- Novartis AG
- PharmaEssentia Corporation
- Bristol Myers Squibb
- Pfizer, Inc.
- Takeda Pharmaceutical Company Limited
- Incyte
- Johnson & Johnson
- Teva Pharmaceutical Industries Ltd,
- F. Hoffmann-La Roche AG
- Gamida Cell
- Sierra Oncology, Inc
- CTI BioPharma Corp
- Imago BioSciences
- AbbVie Inc
- Constellation Pharmaceuticals
Table of Contents
1. Research Objective and Assumption
- Research Objectives
- Assumptions
- Abbreviations
2. Market Purview
- Report Description
- Market Definition and Scope
- Executive Summary
- Market Snippet, By Disease Type
- Market Snippet, By Drug Class
- Market Snippet, By Distribution Channel
- Market Snippet, By Region
- Coherent Opportunity Map (COM)
3. Market Dynamics, Regulations, and Trends Analysis
- Market Dynamics
- Drivers
- Restraints
- Market Opportunities
- Impact Analysis
- Regulatory Scenario
- Epidemiology
- Merger, Acquisition, and Collaborations
- Regulatory Scenario
- Key Developments
- PEST Analysis
4. Global Myeloproliferative Neoplasms Treatment Market - COVID-19 Impact Analysis
- Economic Impact
- COVID-19 Epidemiology
- Impact on Supply and Demand
5. Global Myeloproliferative Neoplasms Treatment Market, By Disease Type, 2018-2030, (US$ Bn)
- Introduction
- Market Share Analysis, 2023 and 2030 (%)
- Y-o-Y Growth Analysis, 2019-2030
- Segment Trends
- Essential Thrombocythemia (ET)
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Segment Trends
- Myelofibrosis (MF)
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Segment Trends
- Polycythemia Vera (PV)
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Segment Trends
6. Global Myeloproliferative Neoplasms Treatment Market, By Drug Class, 2018 - 2030, (US$ Bn)
- Introduction
- Market Share Analysis, 2023 and 2030 (%)
- Y-o-Y Growth Analysis, 2019-2030
- Segment Trends
- Tyrosine Kinase Inhibitors
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Segment Trends
- Janus Kinase Inhibitors
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Segment Trends
- Antimetabolites
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Segment Trends
- Others
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Segment Trends
7. Global Myeloproliferative Neoplasms Treatment Market, By Distribution Channel, 2018 - 2030, (US$ Bn)
- Introduction
- Market Share Analysis, 2023 and 2030 (%)
- Y-o-Y Growth Analysis, 2019-2030
- Segment Trends
- Hospital Pharmacies
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Retail Pharmacies
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
- Online Pharmacies
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2018 - 2030, (US$ Bn)
8. Global Myeloproliferative Neoplasms Treatment Market, By Region, 2018 - 2030, (US$ Bn)
- Introduction
- Market Share Analysis, 2023 and 2030 (%)
- Y-o-Y Growth Analysis, 2019-2030
- Segment Trends
- North America
- Introduction
- Market Size and Forecast, Y-o-Y Growth and By Disease Type, 2018 -2030, (US$ Bn)
- Market Size and Forecast, Y-o-Y Growth and By Drug Class, 2018 -2030, (US$ Bn)
- Market Size and Forecast, Y-o-Y Growth and By Distribution Channel, 2018-2030, (US$ Bn)
- Market Size and Forecast, By Country, Y-o-Y Growth and By Country, 2018 -2030, (US$ Bn)
- U.S.
- Canada
- Europe
- Introduction
- Market Size and Forecast, Y-o-Y Growth and By Disease Type, 2018 -2030, (US$ Bn)
- Market Size and Forecast, Y-o-Y Growth and By Drug Class, 2018 -2030, (US$ Bn)
- Market Size and Forecast, Y-o-Y Growth and By Distribution Channel, 2018-2030, (US$ Bn)
- Market Size and Forecast, By Country, Y-o-Y Growth and By Country, 2018 -2030, (US$ Bn)
- U.K.
- Germany
- Italy
- France
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- Introduction
- Market Size and Forecast, Y-o-Y Growth and By Disease Type, 2018 -2030, (US$ Bn)
- Market Size and Forecast, Y-o-Y Growth and By Drug Class, 2018 -2030, (US$ Bn)
- Market Size and Forecast, Y-o-Y Growth and By Distribution Channel, 2018-2030, (US$ Bn)
- Market Size and Forecast, By Country, Y-o-Y Growth and By Country, 2018 -2030, (US$ Bn)
- China
- India
- Japan
- ASEAN
- Australia
- South Korea
- Rest of Asia Pacific
- Latin America
- Introduction
- Market Size and Forecast, Y-o-Y Growth and By Disease Type, 2018 -2030, (US$ Bn)
- Market Size and Forecast, Y-o-Y Growth and By Drug Class, 2018 -2030, (US$ Bn)
- Market Size and Forecast, Y-o-Y Growth and By Distribution Channel, 2018-2030, (US$ Bn)
- Market Size and Forecast, By Country, Y-o-Y Growth and By Country, 2018 -2030, (US$ Bn)
- Brazil
- Mexico
- Argentina
- Rest of Latin America
- Middle East
- Introduction
- Market Size and Forecast, Y-o-Y Growth and By Disease Type, 2018 -2030, (US$ Bn)
- Market Size and Forecast, Y-o-Y Growth and By Drug Class, 2018 -2030, (US$ Bn)
- Market Size and Forecast, Y-o-Y Growth and By Distribution Channel, 2018-2030, (US$ Bn)
- Market Size and Forecast, By Country, Y-o-Y Growth and By Country, 2018 -2030, (US$ Bn)
- GCC
- Israel
- Rest of Middle East
- Africa
- Introduction
- Market Size and Forecast, Y-o-Y Growth and By Disease Type, 2018 -2030, (US$ Bn)
- Market Size and Forecast, Y-o-Y Growth and By Drug Class, 2018 -2030, (US$ Bn)
- Market Size and Forecast, Y-o-Y Growth and By Distribution Channel, 2018-2030, (US$ Bn)
- Market Size and Forecast, Y-o-Y Growth and By Region/Country, 2018 -2030, (US$ Bn)
- North Africa
- Central Africa
- South Africa
9. Competitive Landscape
- Company Profiles
- Novartis AG
- Company Overview
- Product Portfolio
- Key Highlights
- Financial Overview
- Strategies
- PharmaEssentia Corporation
- Bristol Myers Squibb
- Pfizer, Inc.
- Takeda Pharmaceutical Company Limited
- Incyte
- Johnson & Johnson
- Teva Pharmaceutical Industries Ltd
- F. Hoffmann-La Roche AG
- Gamida Cell
- Sierra Oncology, Inc
- CTI BioPharma Corp
- Imago BioSciences
- AbbVie Inc
- Constellation Pharmaceuticals
10. Section
- References
- Research Methodology
- About us and Sales Contact




