PUBLISHER: Coherent Market Insights | PRODUCT CODE: 1707353

PUBLISHER: Coherent Market Insights | PRODUCT CODE: 1707353
Blood Gas Analyzers Market, By Product Type, By Modality, By Application, By End User, By Geography
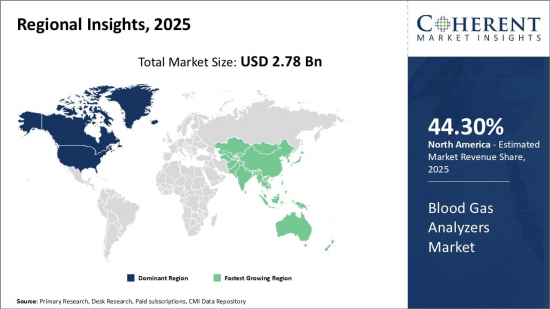
Global Blood Gas Analyzers Market is estimated to be valued at USD 2.78 Bn in 2025 and is expected to reach USD 4.13 Bn by 2032, growing at a compound annual growth rate (CAGR) of 5.8% from 2025 to 2032.
| Report Coverage | Report Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 2.78 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 5.80% | 2032 Value Projection: | USD 4.13 Bn |
Blood gas analyzers play a pivotal role in monitoring critical conditions in patients and have become an indispensable tool for clinicians across emergency rooms and intensive care units. They provide real-time analysis of blood gas, electrolytes, and metabolite parameters including pH, pCO2, pO2, sodium, potassium, ionized calcium, glucose, and lactate directly from blood samples. This helps physicians gain rapid insights into patient's oxygenation, ventilation, and acid-base status, which is vital for timely clinical decision making and management of acute and chronic respiratory diseases, cardiac illnesses, organ transplant, sepsis, trauma, and other critical care conditions. Advancements in miniaturization and portability of devices along with introduction of sophisticated technologies like optical sensing are likely to augment the adoption of blood gas analyzers globally.
Market Dynamics:
The global blood gas analyzers market is primarily driven by the rising geriatric population susceptible to chronic diseases, increasing incidence of respiratory disorders, growing number of ICU admissions worldwide, and increasing number of product launches or approvals.
For instance, in April 2021, LivaNova PLC, a global medical device company, announced that it has received U.S. Food and Drugs Administration (FDA) 510(k) clearance for B-Capta, the in-line, blood-gas monitoring system integrated into the S5 heart-lung machine (HLM). The device is intended to monitor arterial and venous blood gas values even during lengthy and complicated pediatric and adult cardiopulmonary bypass surgeries. B-Capta will now be accessible internationally after receiving CE Mark in May 2020 and completing a successful limited commercial release in Europe.
Key Features of the Study:
- This report provides an in-depth analysis of the global blood gas analyzers market, and provides market size (US$ Billion) and Compound Annual Growth Rate (CAGR %) for the forecast period (2025-2032), considering 2024 as the base year
- It elucidates potential revenue growth opportunities across different segments and explains attractive investment proposition matrices for this market
- This study also provides key insights about market drivers, restraints, opportunities, new product launches or approvals, market trends, regional outlook, and competitive strategies adopted by key players
- It profiles key players in the global blood gas analyzers market based on the following parameters such as company highlights, products portfolio, key highlights, financial performance, and strategies
- The key companies covered as a part of this study include Radiometer Medical ApS, Werfen, Nova Biomedical, OPTI Medical Systems, Inc., Accurex Biomedical Pvt. Ltd., Siemens Healthcare GmbH, Medica Corporation, ERBA Diagnostics Inc., F. Hoffmann-La Roche Ltd., Convergent Technologies, Bio-Rad Laboratories, and EKF Diagnostics
- Insights from this report would allow marketers and the management authorities of the companies to make informed decisions regarding their future product launches, type up-gradation, market expansion, and marketing tactics
- The global blood gas analyzers market report caters to various stakeholders in this industry including investors, suppliers, product manufacturers, distributors, new entrants, and financial analysts
- Stakeholders would have ease in decision-making through various strategy matrices used in analyzing the global blood gas analyzers market
Blood Gas Analyzers Market Detailed Segmentation:
- By Product Type:
- Instruments
- Consumables
- By Modality:
- Companion
- Benchtop
- Portable
- By Application:
- Anesthesia Management
- Critical Care
- Cardiovascular Procedures
- Others (Respiratory Function Testing, etc.)
- By End User:
- Hospitals and Clinics
- Diagnostic Laboratories
- Homecare Settings
- Others
- By Region:
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East
- Africa
- Company Profiles
- Radiometer Medical ApS
- Werfen
- Nova Biomedical
- OPTI Medical Systems, Inc.
- Accurex Biomedical Pvt. Ltd.
- Siemens Healthcare GmbH
- Medica Corporation
- ERBA Diagnostics Inc.
- F.Hoffmann-La Roche Ltd.
- Convergent Technologies
- Bio-Rad Laboratories
- EKF Diagnostics
Table of Contents:
1. Research Objectives and Assumptions
- Research Objectives
- Assumptions
- Abbreviations
2. Market Purview
- Report Description
- Market Definition and Scope
- Executive Summary
- Global Blood Gas Analyzers Market, By Product Type
- Global Blood Gas Analyzers Market, By Modality
- Global Blood Gas Analyzers Market, By Application
- Global Blood Gas Analyzers Market, By End User
- Global Blood Gas Analyzers Market, By Region
- Coherent Opportunity Map (COM)
3. Market Dynamics, Regulations, and Trends Analysis
- Market Dynamics
- Drivers
- Restraints
- Opportunities
- Impact Analysis
- Key Highlights
- Regulatory Scenario
- Product Launches/Approvals
- PEST Analysis
- PORTER's Analysis
- Merger and Acquisition Scenario
4. Global Blood Gas Analyzers Market - Impact of Coronavirus (COVID-19) Pandemic
- COVID-19 Epidemiology
- Supply Side and Demand Side Analysis
- Economic Impact
5. Global Blood Gas Analyzers Market, By Product Type, 2020-2032, (US$ Bn)
- Introduction
- Market Share Analysis, 2025 and 2032 (%)
- Y-o-Y Growth Analysis, 2021 - 2032
- Segment Trends
- Instruments
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (US$ Bn)
- Consumables
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (US$ Bn)
6. Global Blood Gas Analyzers Market, By Modality, 2020-2032, (US$ Bn)
- Introduction
- Market Share Analysis, 2025 and 2032 (%)
- Y-o-Y Growth Analysis, 2021 - 2032
- Segment Trends
- Benchtop
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (US$ Bn)
- Portable
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (US$ Bn)
7. Global Blood Gas Analyzers Market, By Application, 2020-2032, (US$ Bn)
- Introduction
- Market Share Analysis, 2025 and 2032 (%)
- Y-o-Y Growth Analysis, 2021 - 2032
- Segment Trends
- Anesthesia Management
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (US$ Bn)
- Critical Care
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (US$ Bn)
- Cardiovascular Procedures
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (US$ Bn)
- Others
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (US$ Bn)
8. Global Blood Gas Analyzers Market, By End User, 2020-2032, (US$ Bn)
- Introduction
- Market Share Analysis, 2025 and 2032 (%)
- Y-o-Y Growth Analysis, 2021 - 2032
- Segment Trends
- Hospitals and Clinics
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (US$ Bn)
- Diagnostic Laboratories
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (US$ Bn)
- Homecare Settings
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (US$ Bn)
- Others
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (US$ Bn)
9. Global Blood Gas Analyzers Market, By Region, 2020-2032, (US$ Bn)
- Introduction
- Market Share Analysis, 2025 and 2032 (%)
- Y-o-Y Growth Analysis, 2021 - 2032
- Segment Trends
- North America
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, By Product Type, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Modality, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Application, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By End User, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2020-2032, (US$ Bn)
- U.S.
- Canada
- Europe
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, By Product Type, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Modality, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Application, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By End User, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2020-2032, (US$ Bn)
- U.K.
- Germany
- Italy
- France
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, By Product Type, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Modality, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Application, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By End User, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2020-2032, (US$ Bn)
- China
- Japan
- India
- ASEAN
- Australia
- South Korea
- Rest of Asia Pacific
- Latin America
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, By Product Type, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Modality, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Application, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By End User, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2020-2032, (US$ Bn)
- Brazil
- Mexico
- Argentina
- Rest of Latin America
- Middle East
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, By Product Type, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Modality, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Application, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By End User, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2020-2032, (US$ Bn)
- GCC
- Israel
- Rest of Middle East
- Africa
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, By Product Type, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Modality, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Application, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By End User, 2020-2032, (US$ Bn)
- Market Size and Forecast, and Y-o-Y Growth, By Country/Region, 2020-2032, (US$ Bn)
- North Africa
- Central Africa
- South Africa
10. Competitive Landscape
- Radiometer Medical ApS
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- Werfen
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- Nova Biomedical
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- OPTI Medical Systems, Inc.
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- Accurex Biomedical Pvt. Ltd.
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- Siemens Healthcare GmbH
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- Medica Corporation
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- ERBA Diagnostics Inc.
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- Hoffmann-La Roche Ltd.
- Convergent Technologies
- Bio-Rad Laboratories
- EKF Diagnostics
11. Section
- Research Methodology
- About us




