PUBLISHER: Global Market Insights Inc. | PRODUCT CODE: 1959570

PUBLISHER: Global Market Insights Inc. | PRODUCT CODE: 1959570
Immune Checkpoint Inhibitors Market Opportunity, Growth Drivers, Industry Trend Analysis, and Forecast 2026 - 2035
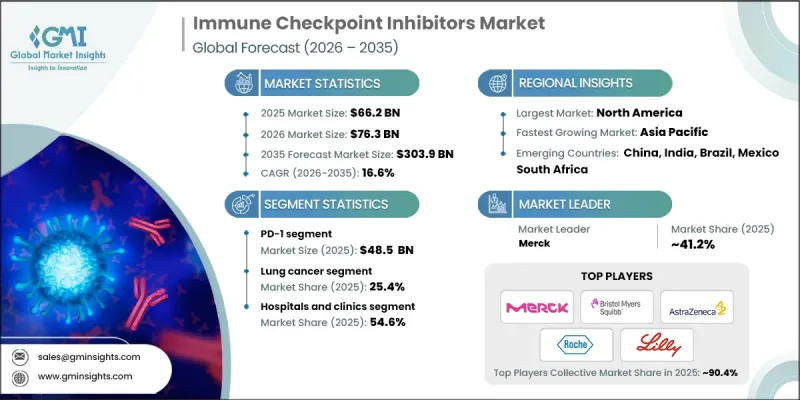
The Global Immune Checkpoint Inhibitors Market was valued at USD 66.2 billion in 2025 and is estimated to grow at a CAGR of 16.6% to reach USD 303.9 billion by 2035.
The market expansion is driven by the proven clinical effectiveness of these therapies across a broad spectrum of cancers and their increasing adoption as a standard of care in oncology. As cancer incidence continues to rise worldwide, the demand for potent and targeted treatments such as immune checkpoint inhibitors is intensifying. These therapies work by enhancing the body's immune system to combat tumor cells, specifically by blocking immune checkpoint proteins like PD-1, PD-L1, and CTLA-4, which are exploited by cancer cells to evade immune detection. By inhibiting these proteins, T-cell function is restored, enabling the immune system to effectively target and eliminate malignant cells. The rising prevalence of hard-to-treat and late-stage cancers is increasing the patient population for immunotherapy, while ongoing innovation in the development of more effective, precise, and safe inhibitors continues to propel market growth.
| Market Scope | |
|---|---|
| Start Year | 2025 |
| Forecast Year | 2026-2035 |
| Start Value | $66.2 Billion |
| Forecast Value | $303.9 Billion |
| CAGR | 16.6% |
The PD-1 segment reached USD 48.5 billion in 2025, reflecting its critical role in cancer immunotherapy. PD-1 inhibitors enhance the immune system's ability to attack tumor cells by blocking the PD-1 pathway, which is commonly used by cancer cells to escape immune recognition. The success of these therapies is supported by strong clinical outcomes, a growing list of approved indications, and widespread adoption in oncology practices, positioning PD-1 inhibitors as a cornerstone in modern cancer treatment. Their effectiveness across multiple cancer types reinforces their dominant share and solidifies their importance in clinical oncology.
The lung cancer segment held 25.4% share and is expected to grow at a CAGR of 16% through 2035. This dominance is attributed to the high prevalence of lung cancer and the demonstrated clinical benefits of immune checkpoint inhibitors in improving survival rates and patient outcomes. The increasing adoption of immunotherapy as a primary treatment, combined with ongoing clinical trials exploring its efficacy in different stages of lung cancer, continues to strengthen market presence. The high disease burden and unmet medical needs further drive demand for these therapies, sustaining robust growth in this segment.
North America Immune Checkpoint Inhibitors Market held 48.3% share in 2025. This leadership is supported by the presence of major pharmaceutical companies actively engaged in research, development, and commercialization of these therapies. The region benefits from a favorable regulatory environment, strong governmental and non-governmental support for cancer treatment initiatives, and rising demand driven by increasing cancer prevalence. Advanced healthcare infrastructure, widespread awareness of immunotherapy options, and access to innovative treatment solutions collectively strengthen North America's dominant position in the global market.
Key players in the Global Immune Checkpoint Inhibitors Market include Merck, Bristol-Myers Squibb Company, AstraZeneca, BeiGene, GlaxoSmithKline, Eli Lilly and Company, Sanofi, Immutep Limited, Incyte Corporation, Shanghai Junshi Biosciences, Zydus Lifesciences, Regeneron Pharmaceuticals, F. Hoffmann-La Roche, and Sun Pharmaceuticals. Companies in the immune checkpoint inhibitors market are leveraging multiple strategies to expand their footprint and reinforce their market presence. They are investing heavily in research and development to create next-generation inhibitors with improved safety profiles, broader efficacy, and expanded indications across cancer types. Strategic partnerships with biotechnology firms, hospitals, and academic institutions accelerate clinical trials and innovation. Geographic expansion into emerging markets allows firms to tap into growing patient populations. Additionally, mergers and acquisitions, licensing agreements, and collaborations with regulatory agencies streamline market entry and product approvals.
Table of Contents
Chapter 1 Methodology and Scope
- 1.1 Market scope and definition
- 1.2 Research approach
- 1.3 Quality commitments
- 1.3.1 GMI AI policy and data integrity commitment
- 1.3.1.1 Source consistency protocol
- 1.3.1 GMI AI policy and data integrity commitment
- 1.4 Research trail and confidence scoring
- 1.4.1 Research trail components
- 1.4.2 Scoring components
- 1.5 Data collection
- 1.5.1 Partial list of primary sources
- 1.6 Data mining sources
- 1.6.1 Paid sources
- 1.6.1.1 Sources, by region
- 1.6.1 Paid sources
- 1.7 Base estimates and calculations
- 1.7.1 Revenue share analysis
- 1.7.2 Base year calculation
- 1.8 Forecast model
- 1.9 Research transparency addendum
- 1.9.1 Source attribution framework
- 1.9.2 Quality assurance metrics
- 1.9.3 Our commitment to trust
Chapter 2 Executive Summary
- 2.1 Industry 360° synopsis
- 2.2 Key market trends
- 2.2.1 Regional trends
- 2.2.2 Type trends
- 2.2.3 Application trends
- 2.2.4 End use trends
- 2.3 CXO perspectives: Strategic imperatives
- 2.3.1 Key decision points for industry executives
- 2.3.2 Critical success factors for market players
- 2.4 Future outlook and strategic recommendations
Chapter 3 Industry Insights
- 3.1 Industry ecosystem analysis
- 3.2 Industry impact forces
- 3.2.1 Growth drivers
- 3.2.1.1 Rising prevalence of cancer worldwide
- 3.2.1.2 Expanding approvals across multiple cancer indications
- 3.2.1.3 Growing shift toward immunotherapy as standard of care
- 3.2.1.4 Increasing investments and partnerships
- 3.2.2 Industry pitfalls and challenges
- 3.2.2.1 Growing risk of immune-related adverse events
- 3.2.2.2 High treatment costs
- 3.2.3 Market opportunities
- 3.2.3.1 Growth of combination immunotherapy regimens
- 3.2.3.2 Development of next-generation immune checkpoint targets
- 3.2.1 Growth drivers
- 3.3 Growth potential analysis
- 3.4 Regulatory landscape
- 3.4.1 North America
- 3.4.2 Europe
- 3.4.3 Asia Pacific
- 3.4.4 Latin America
- 3.4.5 Middle East and Africa
- 3.5 Reimbursement scenario
- 3.6 Pipeline analysis
- 3.7 Pricing analysis
- 3.8 Future market trends
- 3.9 Porter's analysis
- 3.10 PESTEL analysis
Chapter 4 Competitive Landscape, 2025
- 4.1 Introduction
- 4.2 Company market share analysis
- 4.2.1 Global
- 4.2.2 North America
- 4.2.3 Europe
- 4.2.4 Asia Pacific
- 4.3 Company matrix analysis
- 4.4 Competitive analysis of major market players
- 4.5 Competitive positioning matrix
- 4.6 Key developments
- 4.6.1 Merger and acquisition
- 4.6.2 Partnership and collaboration
- 4.6.3 New product launches
- 4.6.4 Expansion plans
Chapter 5 Market Estimates and Forecast, By Type, 2022 - 2035 ($ Mn)
- 5.1 Key trends
- 5.2 PD-1
- 5.3 PD-L1
- 5.4 CTLA-4
- 5.5 Other types
Chapter 6 Market Estimates and Forecast, By Application, 2022 - 2035 ($ Mn)
- 6.1 Key trends
- 6.2 Lung cancer
- 6.3 Breast cancer
- 6.4 Bladder cancer
- 6.5 Melanoma
- 6.6 Cervical cancer
- 6.7 Hodgkin lymphoma
- 6.8 Colorectal cancer
- 6.9 Other applications
Chapter 7 Market Estimates and Forecast, By End Use, 2022 - 2035 ($ Mn)
- 7.1 Key trends
- 7.2 Hospitals and clinics
- 7.3 Cancer centers
- 7.4 Academic and research institutes
Chapter 8 Market Estimates and Forecast, By Region, 2022 - 2035 ($ Mn)
- 8.1 Key trends
- 8.2 North America
- 8.2.1 U.S.
- 8.2.2 Canada
- 8.3 Europe
- 8.3.1 Germany
- 8.3.2 UK
- 8.3.3 France
- 8.3.4 Spain
- 8.3.5 Italy
- 8.3.6 Netherlands
- 8.4 Asia Pacific
- 8.4.1 China
- 8.4.2 India
- 8.4.3 Japan
- 8.4.4 Australia
- 8.4.5 South Korea
- 8.5 Latin America
- 8.5.1 Brazil
- 8.5.2 Mexico
- 8.5.3 Argentina
- 8.6 Middle East and Africa
- 8.6.1 South Africa
- 8.6.2 Saudi Arabia
- 8.6.3 UAE
Chapter 9 Company Profiles
- 9.1 AstraZeneca
- 9.2 BeiGene
- 9.3 Bristol-Myers Squibb Company
- 9.4 Eli Lilly and Company
- 9.5 F. Hoffmann-La Roche
- 9.6 GlaxoSmithKline
- 9.7 Incyte Corporation
- 9.8 Immutep Limited
- 9.9 Merck
- 9.10 Regeneron Pharmaceuticals
- 9.11 Sanofi
- 9.12 Shanghai Junshi Biosciences
- 9.13 Sun Pharmaceuticals
- 9.14 Zydus Lifesciences




