PUBLISHER: Global Market Insights Inc. | PRODUCT CODE: 1982278

PUBLISHER: Global Market Insights Inc. | PRODUCT CODE: 1982278
Pre Analytical Automation Market Opportunity, Growth Drivers, Industry Trend Analysis, and Forecast 2026 - 2035
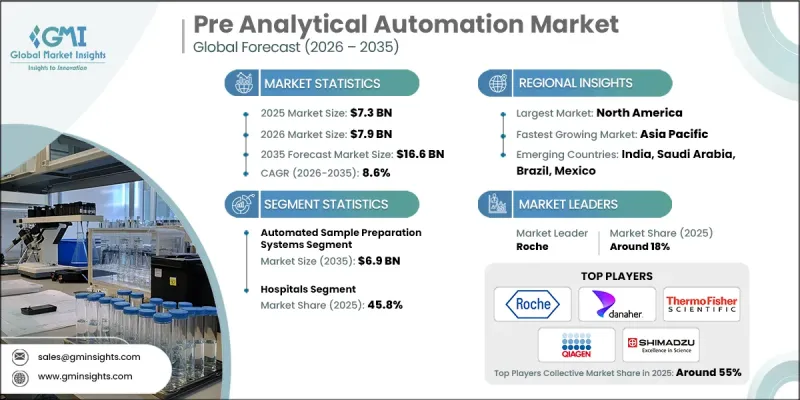
The Global Pre Analytical Automation Market was valued at USD 7.3 billion in 2025 and is estimated to grow at a CAGR of 8.6% to reach USD 16.6 billion by 2035.
Market growth is propelled by the steady rise in diagnostic test volumes, increasing demand for laboratory automation, and growing capital investments in advanced laboratory instrumentation aimed at accelerating result delivery. Clinical laboratories worldwide are under mounting pressure to enhance operational efficiency while maintaining accuracy and quality standards. The push for early disease detection, coupled with the rising burden of chronic and acute health conditions, has significantly increased the number of tests processed daily. To manage current and anticipated workloads, laboratories are accelerating digital transformation initiatives and implementing interconnected automation platforms within their operational frameworks. Modern pre-analytical automation systems streamline sample handling processes, improve workflow continuity, and reduce manual intervention. These solutions support higher throughput capacity, optimize sample routing, prioritize urgent cases, and enhance processing speed without proportionally increasing staffing requirements, thereby strengthening overall laboratory productivity.
| Market Scope | |
|---|---|
| Start Year | 2025 |
| Forecast Year | 2026-2035 |
| Start Value | $7.3 Billion |
| Forecast Value | $16.6 Billion |
| CAGR | 8.6% |
The automated sample preparation systems segment captured a 39.5% share in 2025. The demand for faster turnaround times continues to drive adoption across laboratories handling complex and high-volume testing environments. Automated systems simplify intricate pre-analytical procedures by executing essential preparation tasks through standardized and programmable workflows. By minimizing manual handling and reducing the risk of human error, these platforms enhance consistency and operational precision. Laboratories managing sophisticated testing protocols increasingly rely on automated sample preparation technologies to maintain efficiency, reproducibility, and quality control.
The hospitals segment accounted for 45.8% share in 2025. Rising diagnostic workloads within hospital-based laboratories are prompting institutions to automate key pre-analytical stages to preserve efficiency and accuracy. Large healthcare facilities processing high inpatient and outpatient volumes are integrating automation to sustain optimal turnaround times and alleviate workforce strain. Comprehensive automation lines that unify pre-analytical functions have strengthened operational standardization and minimized variability in sample processing. Automation deployment in hospital laboratories contributes to timely reporting, reinforced quality assurance measures, and reduced reliance on manual processes.
North America Pre Analytical Automation Market held a 38.6% share in 2025. The region's growth is supported by rising diagnostic demand and substantial investments in research, infrastructure, and laboratory modernization by both large-scale and mid-sized healthcare institutions. The increasing need for high-throughput testing capabilities, advanced molecular diagnostics, and AI-enhanced laboratory systems is reinforcing regional leadership. Ongoing technological adoption aimed at improving workflow efficiency and analytical accuracy continues to position North America at the forefront of laboratory automation innovation.
Key companies operating in the Global Pre Analytical Automation Market include Roche, Beckman Coulter (Danaher Corporation), Abbott, SIEMENS Healthineers, Thermo Fisher SCIENTIFIC, Becton Dickinson and Company, QIAGEN, Sysmex, HAMILTON, TECAN, SHIMADZU, SARSTEDT, Inpeco, Copan Diagnostics, and Greiner Bio-One. Companies in the Global Pre Analytical Automation Market are reinforcing their competitive positions through sustained investment in innovation, strategic collaborations, and global expansion initiatives. Market leaders are prioritizing the development of integrated and scalable automation platforms designed to accommodate rising test volumes and evolving laboratory requirements. Partnerships with healthcare institutions and diagnostic networks are enhancing system interoperability and long-term service contracts. Organizations are also incorporating artificial intelligence and advanced data analytics to optimize workflow management and predictive maintenance capabilities. Expansion into emerging healthcare markets and strengthening regional distribution networks remain key growth strategies.
Table of Contents
Chapter 1 Methodology and Scope
- 1.1 Market scope and definition
- 1.2 Research design
- 1.2.1 Research approach
- 1.2.2 Data collection methods
- 1.3 Data mining sources
- 1.3.1 Global
- 1.3.2 Regional/Country
- 1.4 Base estimates and calculations
- 1.4.1 Base year calculation
- 1.4.2 Key trends for market estimation
- 1.5 Primary research and validation
- 1.5.1 Primary sources
- 1.6 Forecast model
- 1.7 Research assumptions and limitations
Chapter 2 Executive Summary
- 2.1 Industry 360° synopsis
- 2.2 Key market trends
- 2.2.1 Regional trends
- 2.2.2 Product type trends
- 2.2.3 End use trends
- 2.3 CXO perspectives: Strategic imperatives
Chapter 3 Industry Insights
- 3.1 Industry ecosystem analysis
- 3.2 Industry impact forces
- 3.2.1 Growth drivers
- 3.2.1.1 Rising demand for laboratory automation
- 3.2.1.2 Increasing test volumes in clinical diagnostics
- 3.2.1.3 Growing investments in clinical laboratories for faster turnaround times
- 3.2.2 Industry pitfalls and challenges
- 3.2.2.1 High capital and installation costs
- 3.2.2.2 Integration challenges with existing LIS systems
- 3.2.3 Opportunities
- 3.2.3.1 Expansion of automation in emerging markets
- 3.2.1 Growth drivers
- 3.3 Growth potential analysis
- 3.4 Regulatory landscape (Driven by Primary Research)
- 3.4.1 North America
- 3.4.2 Europe
- 3.4.3 Asia Pacific
- 3.5 Technology and innovation landscape (Driven by Primary Research)
- 3.5.1 Current technological trends
- 3.5.2 Emerging technologies
- 3.6 Consumer insights
- 3.7 Pre analytical automation - Solution Architecture overview
- 3.8 Supply chain analysis
- 3.9 Investment landscape
- 3.10 Porter's analysis
- 3.11 PESTEL analysis
- 3.12 Gap analysis
- 3.13 Future market trends (Driven by Primary Research)
Chapter 4 Competitive Landscape, 2025
- 4.1 Introduction
- 4.2 Company matrix analysis
- 4.3 Company market share analysis
- 4.3.1 Global
- 4.3.2 North America
- 4.3.3 Europe
- 4.4 Competitive analysis of major market players
- 4.5 Competitive positioning matrix
- 4.6 Key developments
- 4.6.1 Mergers & acquisitions
- 4.6.2 Partnerships & collaborations
- 4.6.3 New product launches
- 4.6.4 Expansion plans
Chapter 5 Market Estimates and Forecast, By Product Type, 2022 - 2035 ($ Mn)
- 5.1 Key trends
- 5.2 Automated sample preparation systems
- 5.3 Automated sample transport systems
- 5.4 Automated sample sorting systems
- 5.5 Automated sample storage systems
- 5.6 Other product types
Chapter 6 Market Estimates and Forecast, By End Use, 2022 - 2035 ($ Mn)
- 6.1 Key trends
- 6.2 Hospitals
- 6.3 Clinical laboratories
- 6.4 Research institutes
- 6.5 Other end users
Chapter 7 Market Estimates and Forecast, By Region, 2022 - 2035 ($ Mn)
- 7.1 Key trends
- 7.2 North America
- 7.2.1 U.S.
- 7.2.2 Canada
- 7.3 Europe
- 7.3.1 Germany
- 7.3.2 UK
- 7.3.3 France
- 7.3.4 Spain
- 7.3.5 Italy
- 7.3.6 Netherlands
- 7.4 Asia Pacific
- 7.4.1 China
- 7.4.2 Japan
- 7.4.3 India
- 7.4.4 Australia
- 7.4.5 South Korea
- 7.5 Latin America
- 7.5.1 Brazil
- 7.5.2 Mexico
- 7.5.3 Argentina
- 7.6 MEA
- 7.6.1 South Africa
- 7.6.2 Saudi Arabia
- 7.6.3 UAE
Chapter 8 Company Profiles
- 8.1 Abbott
- 8.2 Beckman Coulter (Danaher Corporation)
- 8.3 Becton, Dickinson and Company
- 8.4 Copan Diagnostics
- 8.5 Greiner Bio-One
- 8.6 HAMILTON
- 8.7 Inpeco
- 8.8 QIAGEN
- 8.9 Roche
- 8.10 SARSTEDT
- 8.11 SHIMADZU
- 8.12 SIEMENS Healthineers
- 8.13 Sysmex
- 8.14 TECAN
- 8.15 Thermo Fisher SCIENTIFIC




