PUBLISHER: Global Market Insights Inc. | PRODUCT CODE: 1998664

PUBLISHER: Global Market Insights Inc. | PRODUCT CODE: 1998664
Cardiac Monitoring Devices Market Opportunity, Growth Drivers, Industry Trend Analysis, and Forecast 2026 - 2035
The Global Cardiac Monitoring Devices Market was valued at USD 15 billion in 2025 and is estimated to grow at a CAGR of 6.8% to reach USD 29 billion by 2035.
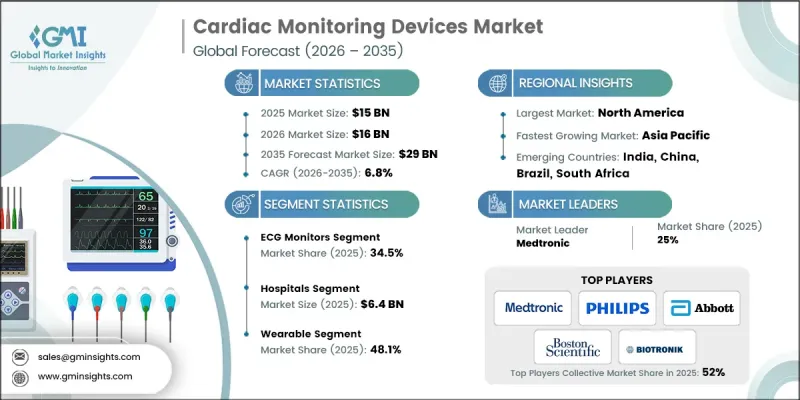
Growth across this market continues to accelerate as healthcare systems increasingly prioritize continuous cardiovascular surveillance and early disease detection. A key growth catalyst is the rapidly rising elderly population worldwide, which carries a greater probability of developing cardiovascular disorders. Age-related cardiac changes significantly increase the likelihood of rhythm disturbances, heart failure, and other cardiac complications, thereby increasing the need for reliable long-term monitoring technologies. At the same time, ongoing advancements in digital health technologies are enhancing the functionality and accessibility of monitoring systems, allowing healthcare providers to remotely observe cardiac performance, as well as in traditional care environments. These improvements support earlier clinical intervention, better patient management, and improved healthcare outcomes. Leading industry participants, such as Philips, Medtronic, Abbott Laboratories, Boston Scientific Corporation, and BIOTRONIK, continue to expand their product capabilities while investing heavily in advanced monitoring technologies, reinforcing their leadership in the global cardiac monitoring devices market and contributing to the steady growth of this industry.
| Market Scope | |
|---|---|
| Start Year | 2025 |
| Forecast Year | 2026-2035 |
| Start Value | $15 Billion |
| Forecast Value | $29 Billion |
| CAGR | 6.8% |
The number of individuals aged 65 years and above continues to increase globally due to longer life expectancy and declining fertility rates. This demographic trend is playing an important role in shaping the cardiac monitoring devices market because older adults face a higher probability of developing cardiovascular complications, particularly rhythm-related disorders and reduced cardiac efficiency. Biological aging contributes to structural and electrical changes within the heart, including tissue stiffening, slower electrical conduction, and alterations in autonomic control. These physiological changes elevate the risk of abnormal cardiac activity and circulatory complications, which increases the importance of reliable cardiac monitoring technologies.
In 2025, the ECG monitors segment held 34.5% share. ECG monitoring systems remain a critical tool for assessing cardiac electrical signals and identifying irregular heart activity. These systems support physicians in evaluating rhythm disturbances, electrical conduction abnormalities, and other changes in cardiac function. Modern ECG monitoring technologies now deliver highly accurate signal acquisition supported by improved noise reduction and digital filtering capabilities, which strengthen diagnostic accuracy and enable timely clinical decisions while maintaining patient safety across healthcare environments.
The wearable cardiac monitoring devices segment captured 48.1% share in 2025 and is expected to reach USD 14.2 billion by 2035. Wearable monitoring technologies consist of compact and convenient devices designed to continuously track cardiac rhythm during normal daily activities. Their growing adoption is largely attributed to improved comfort, extended monitoring duration, and the ability to detect intermittent cardiac irregularities that might remain unnoticed during short clinical evaluations. As healthcare continues to shift toward remote patient management, wearable monitoring solutions are becoming increasingly valuable for both patients and healthcare providers. These devices are lightweight, wireless, and designed for long-term use, enabling uninterrupted cardiac data collection while individuals maintain their normal routines.
North America Cardiac Monitoring Devices Market reached USD 5.6 billion in 2025. The region benefits from a sophisticated healthcare ecosystem, high adoption of advanced medical technologies, and a well-structured regulatory framework that ensures safety and performance standards for cardiac monitoring equipment. In addition, the region faces a substantial burden of cardiovascular conditions, which continues to generate strong demand for advanced monitoring technologies. Healthcare providers increasingly rely on continuous cardiac monitoring solutions to support early identification of rhythm abnormalities and other cardiovascular complications, enabling improved disease management and better patient outcomes.
Prominent organizations operating in the Global Cardiac Monitoring Devices Market include Abbott Laboratories, Medtronic, Philips, Boston Scientific Corporation, BIOTRONIK, GE Healthcare, Nihon Kohden, Schiller AG, MicroPort, AliveCor, BPL Medical, iRhythm, Neurosoft, and ACS Diagnostics. Companies competing in the Cardiac Monitoring Devices Market are focusing on multiple strategic initiatives to strengthen their market position and expand global reach. Major players are prioritizing continuous investment in research and development to introduce technologically advanced monitoring solutions with improved accuracy, connectivity, and patient comfort. Strategic collaborations with healthcare institutions and digital health companies are also becoming common as firms aim to integrate remote monitoring capabilities and data analytics into their product ecosystems. Many organizations are expanding their geographic footprint through distribution partnerships and regional market entry strategies.
Table of Contents
Chapter 1 Research Methodology
- 1.1 Research approach
- 1.2 Quality commitments
- 1.2.1 GMI ai policy & data integrity commitment
- 1.2.1.1 Source consistency protocol
- 1.2.1 GMI ai policy & data integrity commitment
- 1.3 Research trail & confidence scoring
- 1.3.1 Research trail components
- 1.3.2 Scoring components
- 1.4 Data collection
- 1.4.1 Partial list of primary sources
- 1.5 Data mining sources
- 1.5.1 Paid sources
- 1.5.1.1 Sources, by region
- 1.5.1 Paid sources
- 1.6 Base estimates and calculations
- 1.6.1 Base year calculation for any one approach
- 1.7 Forecast model
- 1.7.1 Quantified market impact analysis
- 1.7.1.1 Mathematical impact of growth parameters on forecast
- 1.7.1 Quantified market impact analysis
- 1.8 Research transparency addendum
- 1.8.1 Source attribution framework
- 1.8.2 Quality assurance metrics
- 1.8.3 Our commitment to trust
Chapter 2 Executive Summary
- 2.1 Industry 360° synopsis
- 2.2 Key market trends
- 2.2.1 Device type trends
- 2.2.2 Type trends
- 2.2.3 Application
- 2.2.4 End use trends
- 2.3 CXO perspectives: Strategic imperatives
Chapter 3 Industry Insights
- 3.1 Industry ecosystem analysis
- 3.2 Industry impact forces
- 3.2.1 Growth drivers
- 3.2.1.1 Rising aging population and increased risk of cardiovascular diseases
- 3.2.1.2 Lifestyle changes and increasing prevalence of heart diseases
- 3.2.1.3 Advancements in remote monitoring and wearable technology
- 3.2.1.4 Advancements in digital health and AI
- 3.2.2 Industry pitfalls and challenges
- 3.2.2.1 Data privacy and security concerns
- 3.2.2.2 High cost of advanced monitoring solutions
- 3.2.3 Market opportunity
- 3.2.3.1 Growing preference for home-based and ambulatory cardiac monitoring
- 3.2.1 Growth drivers
- 3.3 Growth potential analysis
- 3.4 Regulatory landscape
- 3.5 Technological landscape (Driven by Primary Research)
- 3.5.1 Current technologies
- 3.5.2 Emerging technologies
- 3.6 Future market trends (Driven by Primary Research)
- 3.7 Impact of AI and generative AI on the market
- 3.8 Porter's analysis
- 3.9 PESTEL analysis
- 3.10 Pricing analysis (Driven by Primary Research)
Chapter 4 Competitive Landscape, 2025
- 4.1 Introduction
- 4.2 Company market share analysis
- 4.3 Company matrix analysis
- 4.4 Competitive analysis of major market players
- 4.5 Competitive positioning matrix
- 4.6 Key developments
- 4.6.1 Merger and acquisition
- 4.6.2 Partnership and collaboration
- 4.6.3 New product launches
- 4.6.4 Expansion plans
Chapter 5 Market Estimates and Forecast, By Device Type, 2022 - 2035 ($ Mn)
- 5.1 Key trends
- 5.2 ECG monitors
- 5.3 Holter monitors
- 5.4 Event recorders/external loop recorders
- 5.5 Mobile cardiac telemetry devices
- 5.6 Implantable cardiac monitors
- 5.7 Other device types
Chapter 6 Market Estimates and Forecast, By Type, 2022 - 2035 ($ Mn)
- 6.1 Key trends
- 6.2 Wearable
- 6.3 Non wearable
Chapter 7 Market Estimates and Forecast, By Application, 2022 - 2035 ($ Mn)
- 7.1 Key trends
- 7.2 Arrhythmia detection
- 7.2.1 Atrial fibrillation
- 7.2.2 Tachycardia
- 7.2.3 Bradycardia
- 7.2.4 Premature atrial and ventricular contractions (PACs/PVCs)
- 7.3 Cardiac ischemia and myocardial infarction detection
- 7.4 Heart rate and rhythm monitoring
- 7.5 Syncope and palpitation monitoring
- 7.6 Pre/post-operative monitoring
- 7.7 Other applications
Chapter 8 Market Estimates and Forecast, By End Use, 2022 - 2035 ($ Mn)
- 8.1 Key trends
- 8.2 Hospitals
- 8.3 Diagnostic centers
- 8.4 Ambulatory surgery centers
- 8.5 Homecare settings
- 8.6 Other end users
Chapter 9 Market Estimates and Forecast, By Region, 2022 - 2035 ($ Mn)
- 9.1 Key trends
- 9.2 North America
- 9.2.1 U.S.
- 9.2.2 Canada
- 9.3 Europe
- 9.3.1 Germany
- 9.3.2 UK
- 9.3.3 France
- 9.3.4 Spain
- 9.3.5 Italy
- 9.3.6 Netherlands
- 9.4 Asia Pacific
- 9.4.1 China
- 9.4.2 Japan
- 9.4.3 India
- 9.4.4 Australia
- 9.4.5 South Korea
- 9.5 Latin America
- 9.5.1 Brazil
- 9.5.2 Mexico
- 9.5.3 Argentina
- 9.6 Middle East and Africa
- 9.6.1 South Africa
- 9.6.2 Saudi Arabia
- 9.6.3 UAE
Chapter 10 Company Profiles
- 10.1 Abbott Laboratories
- 10.2 ACS Diagnostics
- 10.3 AliveCor
- 10.4 BIOTRONIK
- 10.5 Boston Scientific Corporation
- 10.6 BPL Medical
- 10.7 GE Healthcare
- 10.8 iRhythm
- 10.9 Medtronic
- 10.10 MicroPort
- 10.11 Neurosoft
- 10.12 Nihon Kohden
- 10.13 Philips
- 10.14 Schiller AG




