PUBLISHER: Global Market Insights Inc. | PRODUCT CODE: 1998681

PUBLISHER: Global Market Insights Inc. | PRODUCT CODE: 1998681
Foot and Ankle Devices Market Opportunity, Growth Drivers, Industry Trend Analysis, and Forecast 2026 - 2035
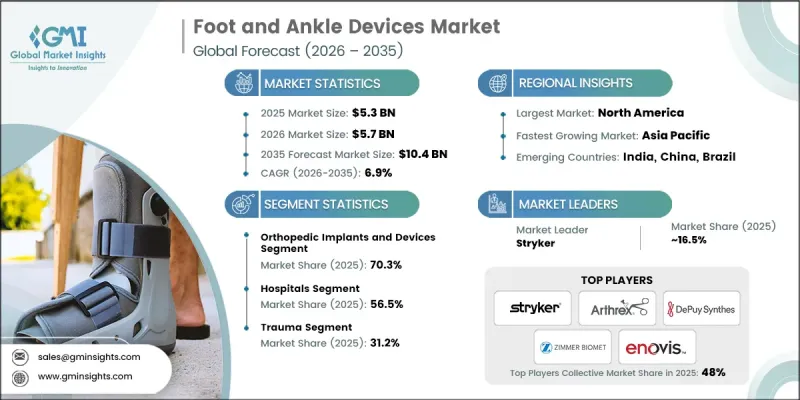
The Global Foot and Ankle Devices Market was valued at USD 5.3 billion in 2025 and is estimated to grow at a CAGR of 6.9% to reach USD 10.4 billion by 2035.
The market's growth is driven by the rising prevalence of orthopedic disorders, an increasing number of trauma cases, and advancements in foot and ankle device technologies. A growing diabetic population with related foot complications, coupled with the development of patient-specific implants and minimally invasive surgical innovations, is accelerating demand. Aging populations and lifestyle-related risk factors are also contributing to the rise in foot and ankle disorders. Furthermore, the global increase in road traffic accidents and high-impact trauma cases is boosting the need for devices that manage fractures, ligament tears, dislocations, and other lower extremity injuries. Technological innovation, including improvements in material science and device engineering, has significantly enhanced patient outcomes, further supporting market expansion.
| Market Scope | |
|---|---|
| Start Year | 2025 |
| Forecast Year | 2026-2035 |
| Start Value | $5.3 Billion |
| Forecast Value | $10.4 Billion |
| CAGR | 6.9% |
The orthopedic implants and devices segment held 70.3% share in 2025. Growth is being fueled by the adoption of 3D-printed orthopedic implants and the high procedural volume for trauma, deformity correction, and degenerative joint conditions. Orthopedic implants remain the primary treatment option for surgical management of foot and ankle conditions, including trauma fixation, joint reconstruction, and deformity correction. Rising incidences of lower extremity fractures, diabetes-related foot complications, age-related musculoskeletal degeneration, and sports injuries are driving demand globally.
The ambulatory surgical centers (ASCs) segment was valued at USD 1.3 billion in 2025 and is expected to grow at a CAGR of 6.7% during 2026-2035. ASCs are benefiting from the global shift toward outpatient orthopedic surgeries and minimally invasive procedures. Technological advancements in anesthesia, pain management, and surgical instrumentation have enabled ASCs to perform procedures such as bunion corrections, ligament reconstructions, and fixation surgeries safely in outpatient settings, supporting market growth.
U.S. Foot and Ankle Devices Market was valued at USD 2.6 billion in 2025. Growth is supported by the development of patient-specific implants, well-established healthcare infrastructure, high procedure volumes, comprehensive reimbursement frameworks, robust R&D activities, and accessible orthopedic specialists. The U.S. is also a leader in innovation, with new technologies such as total ankle replacement systems, advanced fixation screws, synthetic cartilage implants, and external fixation frames often launched and clinically validated domestically before global commercialization.
Prominent companies in the Global Foot and Ankle Devices Market include Acumed LLC, Embla Medical Corporation, Enovis Corporation, Arthrex, Inc., Medartis, Smith & Nephew plc, Stryker Corporation, VILEX, LLC, Zimmer Biomet Holdings, Inc., DePuy Synthes (Johnson & Johnson), CONMED Corporation, Ottobock SE & Co KGaA, Fillauer LLC, and aap implantate AG. Companies in the Global Foot and Ankle Devices Market strengthen their position by investing heavily in R&D for patient-specific implants, advanced fixation systems, and minimally invasive solutions. They pursue strategic partnerships with hospitals, surgical centers, and orthopedic clinics to integrate their devices into standard care pathways. Expanding geographically into emerging markets helps tap into growing patient populations. Firms also focus on product differentiation through innovative materials, 3D printing, and smart implant technologies. Comprehensive after-sales support, physician training programs, and collaborations with research institutions further solidify their market presence and enhance brand credibility.
Table of Contents
Chapter 1 Research Methodology
- 1.1 Research approach
- 1.2 Quality commitments
- 1.2.1 GMI AI policy & data integrity commitment
- 1.2.1.1 Source consistency protocol
- 1.2.1 GMI AI policy & data integrity commitment
- 1.3 Research trail & confidence scoring
- 1.3.1 Research trail components
- 1.3.2 Scoring components
- 1.4 Data collection
- 1.4.1 Partial list of primary sources
- 1.5 Data mining sources
- 1.5.1 Paid sources
- 1.5.1.1 Sources, by region
- 1.5.1 Paid sources
- 1.6 Base estimates and calculations
- 1.6.1 Base year calculation
- 1.7 Forecast model
- 1.7.1 Quantified market impact analysis
- 1.7.1.1 Mathematical impact of growth parameters on forecast
- 1.7.1 Quantified market impact analysis
- 1.8 Research transparency addendum
- 1.8.1 Source attribution framework
- 1.8.2 Quality assurance metrics
- 1.8.3 Our commitment to trust
Chapter 2 Executive Summary
- 2.1 Industry 360° synopsis
- 2.2 Key market trends
- 2.2.1 Regional trends
- 2.2.2 Product trends
- 2.2.3 Application trends
- 2.2.4 End use trends
- 2.3 CXO perspectives: Strategic imperatives
Chapter 3 Industry Insights
- 3.1 Industry ecosystem analysis
- 3.2 Industry impact forces
- 3.2.1 Growth drivers
- 3.2.1.1 Increasing prevalence of orthopedic disorders
- 3.2.1.2 Rising incidence of trauma and road accidents
- 3.2.1.3 Technological advancements in foot and ankle devices
- 3.2.2 Industry pitfalls and challenges
- 3.2.2.1 High cost of foot and ankle devices
- 3.2.2.2 Dearth of skilled healthcare professionals
- 3.2.3 Opportunities
- 3.2.3.1 Integration of digital health and smart orthotics
- 3.2.1 Growth drivers
- 3.3 Growth potential analysis
- 3.4 Regulatory landscape
- 3.4.1 North America
- 3.4.1.1 U.S.
- 3.4.1.2 Canada
- 3.4.2 Europe
- 3.4.3 Asia Pacific
- 3.4.1 North America
- 3.5 Technology and innovation landscape
- 3.5.1 Current technological trends
- 3.5.2 Emerging technologies
- 3.6 Future market trends
- 3.7 Reimbursement scenario
- 3.7.1 North America
- 3.7.2 Europe
- 3.7.3 Asia Pacific
- 3.8 Consumer behavior analysis
- 3.9 Ring fixator competitive mapping, by company
- 3.10 Hexapod categories and technology differentiation
- 3.11 Price trends, by products
- 3.12 Gap analysis
- 3.13 Porter's analysis
- 3.14 PESTEL analysis
Chapter 4 Competitive Landscape, 2025
- 4.1 Introduction
- 4.1.1 Stryker Corporation
- 4.1.2 DePuy Synthes (Johnson & Johnson)
- 4.1.3 Arthrex Inc.
- 4.2 Company matrix analysis
- 4.3 Company market share analysis
- 4.3.1 Global
- 4.3.2 North America
- 4.3.3 Europe
- 4.3.4 Asia Pacific
- 4.4 Competitive analysis of major market players
- 4.5 Competitive positioning matrix
- 4.6 Key developments
- 4.6.1 Mergers & acquisitions
- 4.6.2 Partnerships & collaborations
- 4.6.3 New product launches
- 4.6.4 Expansion plans
Chapter 5 Market Estimates and Forecast, By Product, 2022 - 2035 ($ Mn)
- 5.1 Key trends
- 5.2 Orthopedic implants and devices
- 5.2.1 Joint implants
- 5.2.1.1 Ankle implants
- 5.2.1.2 Other joint implants
- 5.2.2 Fixation devices
- 5.2.2.1 Internal fixation devices
- 5.2.2.1.1 Plates
- 5.2.2.1.2 Screws
- 5.2.2.1.3 Fusion nails
- 5.2.2.1.4 Other internal fixation devices
- 5.2.2.2 External fixation devices
- 5.2.2.2.1 Ring fixation systems
- 5.2.2.2.2 Pin-to-bar systems
- 5.2.2.2.3 Hexapod systems
- 5.2.2.2.4 Other external fixators
- 5.2.2.1 Internal fixation devices
- 5.2.3 Soft tissue orthopedic devices
- 5.2.1 Joint implants
- 5.3 Bracing and support devices
- 5.3.1 Soft bracing & support devices
- 5.3.2 Hard braces & support devices
- 5.3.3 Hinged braces & support devices
- 5.4 Prostheses
Chapter 6 Market Estimates and Forecast, By Application, 2022 - 2035 ($ Mn)
- 6.1 Key trends
- 6.2 Trauma
- 6.3 Hammertoe
- 6.4 Osteoarthritis
- 6.5 Rheumatoid Arthritis
- 6.6 Neurological Disorders
- 6.7 Osteoporosis
- 6.8 Other applications
Chapter 7 Market Estimates and Forecast, By End Use, 2022 - 2035 ($ Mn)
- 7.1 Key trends
- 7.2 Hospitals
- 7.3 Ambulatory surgical centers
- 7.4 Orthopedic clinics
- 7.5 Other end users
Chapter 8 Market Estimates and Forecast, By Region, 2022 - 2035 ($ Mn)
- 8.1 Key trends
- 8.2 North America
- 8.2.1 U.S.
- 8.2.2 Canada
- 8.3 Europe
- 8.3.1 Germany
- 8.3.2 UK
- 8.3.3 France
- 8.3.4 Spain
- 8.3.5 Italy
- 8.3.6 Netherlands
- 8.4 Asia Pacific
- 8.4.1 China
- 8.4.2 Japan
- 8.4.3 India
- 8.4.4 Australia
- 8.4.5 South Korea
- 8.5 Latin America
- 8.5.1 Brazil
- 8.5.2 Mexico
- 8.5.3 Argentina
- 8.6 MEA
- 8.6.1 South Africa
- 8.6.2 Saudi Arabia
- 8.6.3 UAE
Chapter 9 Company Profiles
- 9.1 aap implantate AG
- 9.2 Acumed LLC
- 9.3 Arthrex, Inc.
- 9.4 CONMED Corporation
- 9.5 DePuy Synthes (Johnson & Johnson)
- 9.6 Embla Medical Corporation
- 9.7 Enovis Corporation
- 9.8 Fillauer LLC
- 9.9 Medartis
- 9.10 Orthofix Medical Inc.
- 9.11 Ottobock SE & Co KGaA
- 9.12 Smith & Nephew plc
- 9.13 Stryker Corporation
- 9.14 VILEX, LLC
- 9.15 Zimmer Biomet Holdings, Inc.




