PUBLISHER: Global Market Insights Inc. | PRODUCT CODE: 1998711

PUBLISHER: Global Market Insights Inc. | PRODUCT CODE: 1998711
Fondaparinux Market Opportunity, Growth Drivers, Industry Trend Analysis, and Forecast 2026 - 2035
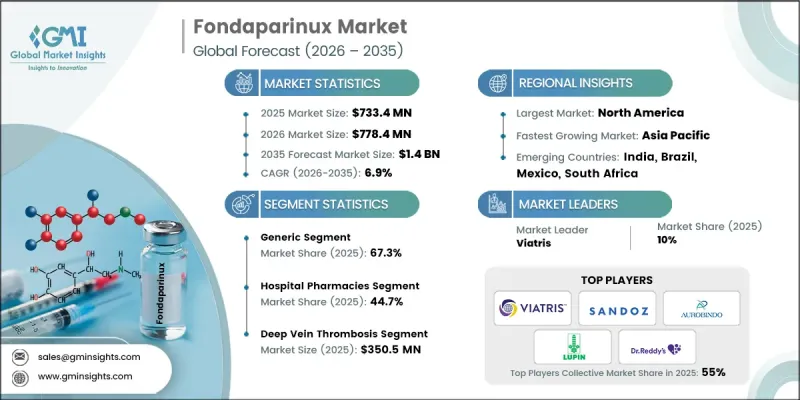
The Global Fondaparinux Market was valued at USD 733.4 million in 2025 and is estimated to grow at a CAGR of 6.9% to reach USD 1.4 billion by 2035.
The steady expansion of the fondaparinux market is primarily supported by the increasing prevalence of thromboembolic conditions, rising number of orthopedic surgical procedures, and the rapidly aging global population. As the incidence of clotting related disorders continues to increase, healthcare providers are focusing more on effective anticoagulation therapies that offer reliable safety and therapeutic outcomes. Fondaparinux, a synthetic anticoagulant, helps in managing and preventing blood clot-related complications by inhibiting specific coagulation pathways. Its consistent pharmacological profile, reduced likelihood of heparin-related adverse reactions, and suitability for use following major surgical procedures have contributed to its growing acceptance in clinical practice. Additionally, improvements in healthcare infrastructure and the expanding availability of cost-effective drug formulations are supporting wider patient access to anticoagulant treatments. Increased awareness regarding the early diagnosis and management of clotting disorders is further strengthening demand for reliable therapeutic options. These combined factors continue to support the long-term growth of the global fondaparinux market. The market is further supported by the rising burden of thromboembolic disorders, increasing surgical interventions, and global demographic changes that contribute to a higher demand for anticoagulant therapy. As healthcare systems continue to improve diagnostic capabilities, more patients are being identified with clotting related conditions that require preventive or therapeutic anticoagulation.
| Market Scope | |
|---|---|
| Start Year | 2025 |
| Forecast Year | 2026-2035 |
| Start Value | $733.4 Million |
| Forecast Value | $1.4 Billion |
| CAGR | 6.9% |
The generic segment accounted for 67.3% share in 2025. Generic fondaparinux consists of bioequivalent formulations of fondaparinux sodium that deliver the same therapeutic benefits as branded products while offering improved affordability. These formulations are widely used for the management and prevention of venous thromboembolism conditions. The dominance of this segment is primarily driven by the cost advantages associated with generic medications, which make them more accessible within healthcare systems that prioritize cost efficiency. In addition, fondaparinux has an established record of clinical safety and effectiveness, and the availability of generic versions has significantly expanded patient access to this treatment option.
The deep vein thrombosis segment generated USD 350.5 million in 2025. Deep vein thrombosis refers to the formation of blood clots within deep veins, typically affecting the lower extremities, and represents one of the primary medical conditions treated with fondaparinux therapy. The medication is widely utilized for both the prevention and treatment of clot formation due to its reliable anticoagulant action and convenient dosing schedule. This segment continues to maintain a strong market position as the global incidence of venous thromboembolism rises and healthcare providers increasingly implement pharmacological prevention strategies for at-risk patients. Fondaparinux remains a preferred therapeutic option because of its strong clinical performance, dependable safety characteristics, and proven ability to reduce clot progression and recurrence.
North America Fondaparinux Market held a 39% share in 2025. The region's leading position is largely attributed to the significant clinical burden of venous thromboembolism within its healthcare system. Medical facilities across the region continue to adopt fondaparinux as a reliable anticoagulant therapy due to its predictable pharmacological activity and reduced likelihood of certain drug-related complications. Furthermore, demographic changes, including an aging population, are expected to contribute to increasing demand for anticoagulant treatments throughout the forecast period. Strong healthcare infrastructure and access to advanced medical therapies also support the widespread use of fondaparinux across hospitals and specialized treatment centers in North America.
Key companies operating in the Global Fondaparinux Market include Viatris, Sandoz, Abbott Laboratories, Dr. Reddy's Laboratories, Lupin Pharmaceuticals, Aurobindo, Aspen Pharmacare Holdings, Jiangsu Hengrui Medicine, ScinoPharm Taiwan, BrightGene Health, and Shanghai Minbiotech. Companies participating in the Global Fondaparinux Market are implementing various strategic initiatives to strengthen their competitive presence and expand their global market reach. Leading pharmaceutical manufacturers are focusing on the development and distribution of cost-effective generic formulations to increase accessibility and capture a larger share of the anticoagulant therapy market. Many companies are also investing in research and development activities aimed at improving manufacturing processes and ensuring consistent product quality. Strategic collaborations with healthcare providers, distributors, and pharmaceutical partners are helping companies expand their geographic presence and strengthen supply chain capabilities. Additionally, firms are emphasizing regulatory approvals, portfolio diversification, and production capacity expansion to support growing global demand while maintaining strong competitiveness within the evolving fondaparinux market.
Table of Contents
Chapter 1 Research Methodology
- 1.1 Research approach
- 1.2 Quality commitments
- 1.2.1 GMI AI policy & data integrity commitment
- 1.2.1.1 Source consistency protocol
- 1.2.1 GMI AI policy & data integrity commitment
- 1.3 Research trail & confidence scoring
- 1.3.1 Research trail components
- 1.3.2 Scoring components
- 1.4 Data collection
- 1.4.1 Partial list of primary sources
- 1.5 Data mining sources
- 1.5.1 Paid sources
- 1.5.1.1 Sources, by region
- 1.5.1 Paid sources
- 1.6 Base estimates and calculations
- 1.6.1 Base year calculation for any one approach
- 1.7 Forecast model
- 1.7.1 Quantified market impact analysis
- 1.7.1.1 Mathematical impact of growth parameters on forecast
- 1.7.1 Quantified market impact analysis
- 1.8 Research transparency addendum
- 1.8.1 Source attribution framework
- 1.8.2 Quality assurance metrics
- 1.8.3 Our commitment to trust
Chapter 2 Executive Summary
- 2.1 Industry 360° synopsis
- 2.2 Key market trends
- 2.2.1 Product type trends
- 2.2.2 Application trends
- 2.2.3 Distribution channel trends
- 2.2.4 Regional trends
- 2.3 CXO perspectives: Strategic imperatives
Chapter 3 Industry Insights
- 3.1 Industry ecosystem analysis
- 3.2 Industry impact forces
- 3.2.1 Growth drivers
- 3.2.1.1 Rising incidence of thromboembolic diseases
- 3.2.1.2 Increase in hip and knee transplant surgeries
- 3.2.1.3 Surge in aging population worldwide
- 3.2.1.4 Greater awareness and early diagnosis of clotting disorders
- 3.2.2 Industry pitfalls and challenges
- 3.2.2.1 Side effects associated with fondaparinux
- 3.2.2.2 High cost of treatment
- 3.2.3 Market opportunity
- 3.2.3.1 Expansion in emerging markets
- 3.2.3.2 Increasing opportunities in orthopedic and trauma care
- 3.2.1 Growth drivers
- 3.3 Growth potential analysis
- 3.4 Regulatory landscape
- 3.4.1 North America
- 3.4.2 Europe
- 3.4.3 Asia Pacific
- 3.4.4 Latin America
- 3.4.5 Middle East and Africa
- 3.5 Technological landscape
- 3.5.1 Current technologies
- 3.5.2 Emerging technologies
- 3.6 Pricing analysis, 2025 (Driven by Primary Research)
- 3.7 Patent analysis
- 3.8 Future market trends
- 3.9 Porter's analysis
- 3.10 PESTEL analysis
Chapter 4 Competitive Landscape, 2025
- 4.1 Introduction
- 4.2 Company market share analysis
- 4.2.1 North America
- 4.2.2 Europe
- 4.2.3 Asia Pacific
- 4.3 Company matrix analysis
- 4.4 Competitive analysis of major market players
- 4.5 Competitive positioning matrix
- 4.6 Key developments
- 4.6.1 Merger and acquisition
- 4.6.2 Partnership and collaboration
- 4.6.3 New product launches
- 4.6.4 Expansion plans
Chapter 5 Market Estimates and Forecast, By Product Type, 2022 - 2035 ($ Mn)
- 5.1 Key trends
- 5.2 Branded
- 5.3 Generic
Chapter 6 Market Estimates and Forecast, By Application, 2022 - 2035 ($ Mn)
- 6.1 Key trends
- 6.2 Deep vein thrombosis
- 6.3 Pulmonary embolism
- 6.4 Acute coronary syndrome
- 6.5 Other applications
Chapter 7 Market Estimates and Forecast, By Distribution Channel, 2022 - 2035 ($ Mn)
- 7.1 Key trends
- 7.2 Hospital pharmacies
- 7.3 Retail pharmacies
- 7.4 Online pharmacies
Chapter 8 Market Estimates and Forecast, By Region, 2022 - 2035 ($ Mn)
- 8.1 Key trends
- 8.2 North America
- 8.2.1 U.S.
- 8.2.2 Canada
- 8.3 Europe
- 8.3.1 Germany
- 8.3.2 UK
- 8.3.3 France
- 8.3.4 Spain
- 8.3.5 Italy
- 8.3.6 Netherlands
- 8.4 Asia Pacific
- 8.4.1 China
- 8.4.2 Japan
- 8.4.3 India
- 8.4.4 Australia
- 8.4.5 South Korea
- 8.5 Latin America
- 8.5.1 Brazil
- 8.5.2 Mexico
- 8.5.3 Argentina
- 8.6 Middle East and Africa
- 8.6.1 South Africa
- 8.6.2 Saudi Arabia
- 8.6.3 UAE
Chapter 9 Company Profiles
- 9.1 Abbott Laboratories
- 9.2 Aspen Pharmacare Holdings
- 9.3 Aurobindo
- 9.4 BrightGene Health
- 9.5 Dr. Reddy's Laboratories
- 9.6 Jiangsu Hengrui Medicine
- 9.7 Lupin Pharmaceuticals
- 9.8 Sandoz
- 9.9 ScinoPharm Taiwan
- 9.10 Shanghai Minbiotech
- 9.11 Viatris




