PUBLISHER: Global Market Insights Inc. | PRODUCT CODE: 1998759

PUBLISHER: Global Market Insights Inc. | PRODUCT CODE: 1998759
Cyclosporine Market Opportunity, Growth Drivers, Industry Trend Analysis, and Forecast 2026 - 2035
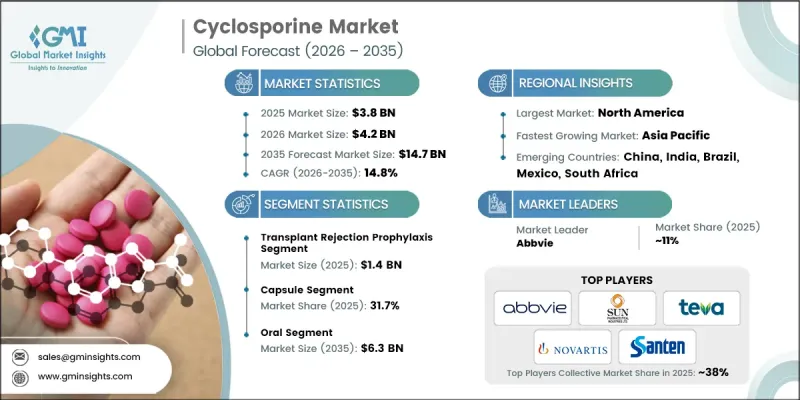
The Global Cyclosporine Market was valued at USD 3.8 billion in 2025 and is estimated to grow at a CAGR of 14.8% to reach USD 14.7 billion by 2035.
The rising prevalence of autoimmune disorders, including rheumatoid arthritis and psoriasis, is a primary driver for cyclosporine demand, while expanding healthcare access in emerging economies and growing investment in immunosuppressive therapy research support market growth. Additionally, heightened awareness of organ donation, advancements in transplant technology, and increasing adoption of immunosuppressive therapies contribute to the steady expansion of the global market. Cyclosporine is increasingly recognized for its effectiveness in preventing organ rejection and managing chronic autoimmune conditions, further enhancing its adoption across hospitals, outpatient clinics, and specialty care centers worldwide.
| Market Scope | |
|---|---|
| Start Year | 2025 |
| Forecast Year | 2026-2035 |
| Start Value | $3.8 Billion |
| Forecast Value | $14.7 Billion |
| CAGR | 14.8% |
Cyclosporine is an immunosuppressive medication that primarily prevents organ rejection in transplant recipients and manages autoimmune diseases. It works by inhibiting T-lymphocyte activity, a central component of the immune system, thereby preventing attacks on transplanted organs or healthy tissue. The drug is prescribed for patients undergoing kidney, liver, or heart transplants and for autoimmune conditions such as rheumatoid arthritis, psoriasis, and specific ophthalmic disorders. The growing incidence of autoimmune diseases, which result from the immune system attacking healthy cells and causing inflammation, has created a strong demand for effective therapies like cyclosporine.
The transplant rejection prophylaxis segment reached USD 1.4 billion in 2025. Its dominance is driven by the crucial role cyclosporine plays in preventing post-transplant organ rejection. High transplant volumes, including kidney, liver, and heart procedures, along with innovations in surgical techniques and post-operative care, have further boosted demand for reliable prophylactic treatments, making this segment a major revenue contributor.
The capsule segment held a share of 31.7% in 2025, as capsules offer superior patient compliance for long-term therapies required in transplant and autoimmune treatments. Advances in encapsulation technology enhance cyclosporine's bioavailability and stability, further strengthening this segment. The rising prevalence of chronic diseases requiring immunosuppressive therapy, coupled with the availability of generic formulations, has cemented the capsule segment's leadership position in the market.
North America Cyclosporine Market captured 36.8% share in 2025 and is expected to witness robust growth during the forecast period. This dominance is supported by strong clinical demand for transplant and ophthalmology indications, high rates of ocular surface disorders, and advanced healthcare infrastructure. Comprehensive reimbursement coverage and high procedure volumes further reinforce North America's leadership in the cyclosporine market, making the region a key revenue contributor globally.
Major players operating in the Global Cyclosporine Market include Amneal Pharmaceuticals, Avet Pharmaceuticals, AdvaCare Pharma, AbbVie, Apotex, Bora Pharmaceuticals, Cipla, Dr. Reddy's Laboratories, Novartis, Santen Pharmaceutical, Sun Pharmaceutical Industries, Strides Pharma Science Limited, Teva Pharmaceutical Industries, and Viatris. Companies in the Cyclosporine Market are adopting strategies such as expanding their product portfolios to include novel formulations with improved bioavailability, stability, and patient compliance. Firms are investing in research and development for new immunosuppressive therapies and exploring combination therapies to strengthen clinical efficacy. Geographic expansion into emerging markets with growing transplant programs and autoimmune disease prevalence is a key focus. Strategic partnerships with hospitals, clinics, and distribution networks enhance market penetration. Additionally, generic product launches, pricing strategies, and awareness campaigns for transplant and autoimmune care improve brand recognition and adoption rates, helping companies maintain a competitive edge and sustain long-term growth in the cyclosporine market.
Table of Contents
Chapter 1 Methodology and Scope
- 1.1 Market scope and definition
- 1.2 Research approach
- 1.3 Quality commitments
- 1.3.1 GMI AI policy and data integrity commitment
- 1.3.1.1 Source consistency protocol
- 1.3.1 GMI AI policy and data integrity commitment
- 1.4 Research trail and confidence scoring
- 1.4.1 Research trail components
- 1.4.2 Scoring components
- 1.5 Data collection
- 1.5.1 Partial list of primary sources
- 1.6 Data mining sources
- 1.6.1 Paid sources
- 1.6.1.1 Sources, by region
- 1.6.1 Paid sources
- 1.7 Base estimates and calculations
- 1.7.1 Revenue share analysis
- 1.7.2 Base year calculation
- 1.8 Forecast model
- 1.9 Research transparency addendum
- 1.9.1 Source attribution framework
- 1.9.2 Quality assurance metrics
- 1.9.3 Our commitment to trust
Chapter 2 Executive Summary
- 2.1 Industry 360° synopsis
- 2.2 Key market trends
- 2.2.1 Regional trends
- 2.2.2 Indication trends
- 2.2.3 Dosage form trends
- 2.2.4 Route of administration trends
- 2.2.5 End use trends
- 2.3 CXO perspectives: Strategic imperatives
Chapter 3 Industry Insights
- 3.1 Industry ecosystem analysis
- 3.2 Industry impact forces
- 3.2.1 Growth drivers
- 3.2.1.1 Growing prevalence of autoimmune diseases
- 3.2.1.2 Growing organ transplantation procedures
- 3.2.1.3 Expansion of ophthalmic indications
- 3.2.1.4 Growing access to generic products
- 3.2.2 Industry pitfalls and challenges
- 3.2.2.1 Adverse side effects
- 3.2.2.2 Availability of alternative immunosuppressants
- 3.2.3 Market opportunities
- 3.2.3.1 Expansion in the ophthalmology pipeline and indications
- 3.2.3.2 Emerging market penetration
- 3.2.1 Growth drivers
- 3.3 Growth potential analysis
- 3.4 Regulatory landscape
- 3.4.1 North America
- 3.4.1.1 U.S.
- 3.4.1.2 Canada
- 3.4.2 Europe
- 3.4.3 Asia Pacific
- 3.4.1 North America
- 3.5 Clinical trials and pipeline analysis (Driven by primary research)
- 3.6 Pricing analysis (Driven by primary research)
- 3.7 Reimbursement scenario (Driven by primary research)
- 3.8 Impact of AI and generative AI on the market
- 3.9 Porter's analysis
- 3.10 PESTEL analysis
Chapter 4 Competitive Landscape, 2025
- 4.1 Introduction
- 4.2 Company market share analysis
- 4.2.1 North America
- 4.2.2 Europe
- 4.2.3 Asia Pacific
- 4.3 Company matrix analysis
- 4.4 Competitive analysis of major market players
- 4.5 Competitive positioning matrix
- 4.6 Key developments
- 4.6.1 Mergers and acquisitions
- 4.6.2 Partnerships and collaborations
- 4.6.3 New product launches
- 4.6.4 Expansion plans
Chapter 5 Market Estimates and Forecast, By Indication, 2022 - 2035 ($ Mn)
- 5.1 Key trends
- 5.2 Transplant rejection prophylaxis
- 5.3 Rheumatoid arthritis
- 5.4 Psoriasis
- 5.5 Autoimmune myasthenia gravis
- 5.6 Dry eye
- 5.7 Other indications
Chapter 6 Market Estimates and Forecast, By Dosage Form, 2022 - 2035 ($ Mn)
- 6.1 Key trends
- 6.2 Capsule
- 6.3 Eye drops
- 6.4 Solutions
- 6.5 Creams and ointments
Chapter 7 Market Estimates and Forecast, By Route of Administration, 2022 - 2035 ($ Mn)
- 7.1 Key trends
- 7.2 Oral
- 7.3 Topical
- 7.4 Parenteral
Chapter 8 Market Estimates and Forecast, By Distribution Channel, 2022 - 2035 ($ Mn)
- 8.1 Key trends
- 8.2 Hospital pharmacies
- 8.3 Retail pharmacies
- 8.4 Online pharmacies
Chapter 9 Market Estimates and Forecast, By Region, 2022 - 2035 ($ Mn)
- 9.1 Key trends
- 9.2 North America
- 9.2.1 U.S.
- 9.2.2 Canada
- 9.3 Europe
- 9.3.1 Germany
- 9.3.2 UK
- 9.3.3 France
- 9.3.4 Spain
- 9.3.5 Italy
- 9.3.6 Netherlands
- 9.4 Asia Pacific
- 9.4.1 China
- 9.4.2 India
- 9.4.3 Japan
- 9.4.4 Australia
- 9.4.5 South Korea
- 9.5 Latin America
- 9.5.1 Brazil
- 9.5.2 Mexico
- 9.5.3 Argentina
- 9.6 Middle East and Africa
- 9.6.1 South Africa
- 9.6.2 Saudi Arabia
- 9.6.3 UAE
Chapter 10 Company Profiles
- 10.1 Amneal Pharmaceuticals
- 10.2 Avet Pharmaceuticals
- 10.3 AdvaCare Pharma
- 10.4 AbbVie
- 10.5 Apotex
- 10.6 Bora Pharmaceuticals
- 10.7 Cipla
- 10.8 Dr. Reddy's Laboratories
- 10.9 Novartis
- 10.10 Santen Pharmaceutical
- 10.11 Sun Pharmaceutical Industries
- 10.12 Strides Pharma Science Limited
- 10.13 Teva Pharmaceutical Industries
- 10.14 Viatris




