PUBLISHER: MarketsandMarkets | PRODUCT CODE: 2011929

PUBLISHER: MarketsandMarkets | PRODUCT CODE: 2011929
Peripheral Vascular Devices Market by Type (Angioplasty Balloon & Angioplasty Stent, Catheters (IVUS/OCT), Plaque Modification, Vascular Closure Devices, Balloon Inflation Devices, Hemodynamic Flow Alteration), End User - Global Forecast to 2031
The peripheral vascular devices market is projected to reach USD 13.22 billion by 2031 from USD 10.02 billion in 2026, at a CAGR of 5.7% from 2026 to 2031.
| Scope of the Report | |
|---|---|
| Years Considered for the Study | 2024-2031 |
| Base Year | 2025 |
| Forecast Period | 2026-2031 |
| Units Considered | Value (USD Billion), Volume (Thousand/Million Units) |
| Segments | Type, End user, and Region |
| Regions covered | North America, Europe, APAC, LATAM, MEA |
The peripheral vascular devices market is experiencing significant growth, driven by the rising prevalence of peripheral artery disease (PAD) and other vascular disorders. These conditions are largely linked to the aging population, along with rising rates of diabetes, obesity, and smoking. Innovations in minimally invasive endovascular devices, such as drug-coated balloons, next-generation stents, atherectomy systems, and embolic protection devices, are greatly improving clinical outcomes and expanding the range of patients who can be treated. Additionally, the growing preference for outpatient and ambulatory surgical center-based procedures, supported by favorable reimbursement policies in many regions, is accelerating the adoption of these devices.
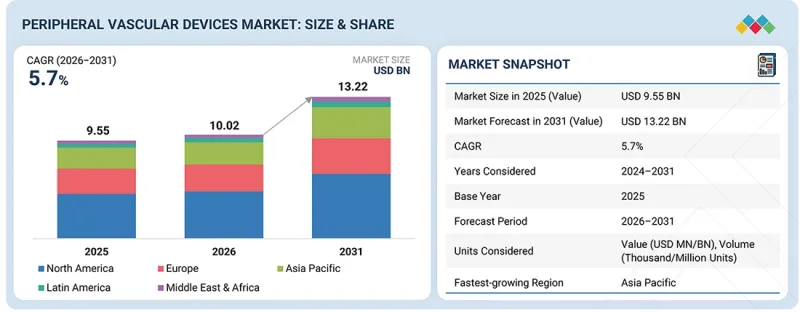
Furthermore, increasing awareness of early diagnosis and treatment, improvements in healthcare infrastructure in developing countries, and ongoing investments by major manufacturers in innovation and clinical research are all contributing to market growth.
"The old/normal balloons segment is expected to grow at the highest CAGR in the angioplasty balloons market during the forecast period."
The angioplasty balloons market is experiencing increased demand for plain (standard) balloons due to their cost-effectiveness, availability of clinical expertise, and versatility across peripheral and coronary interventions. These balloons remain popular for lesion preparation, as well as for pre- and post-dilatation procedures, particularly when drug-coated options are either unnecessary or not covered by insurance. Despite the emergence of advanced balloon technologies, standard balloons continue to see strong sales in less demanding, high-volume markets, especially in developing countries, due to their ease of use, reliability, and compatibility with different stent systems.
"The embolic protection devices segment is expected to grow at the highest CAGR in the hemodynamic flow alteration devices market during the forecast period."
Embolic protection devices are expected to see increased demand in the hemodynamic flow alteration devices market. This is due to their growing use in complex endovascular and structural heart procedures, where the risk of distal embolization is notably high. The rising awareness of procedure-related complications, such as stroke and organ ischemia-especially during carotid, peripheral, and transcatheter interventions-has accelerated their clinical adoption. Additionally, improvements in device design, which enhance capture efficiency, deliverability, and vessel compatibility, have significantly supported market growth. This trend is further bolstered by the introduction of established guidelines and the expansion of minimally invasive vascular procedures.
"Based on end users, the ambulatory surgical centers segment is expected to grow at the highest CAGR in the peripheral vascular devices market during the forecast period."
The peripheral vascular devices market is experiencing significant growth, largely driven by the positive impact of ambulatory surgery centers (ASCs). These centers facilitate the shift toward minimally invasive procedures that prioritize the comfort and safety of outpatient care. ASCs offer numerous benefits, including lower procedural costs, faster patient turnover, shorter hospital stays, and improved operational efficiency, making them appealing to payers, providers, and patients alike. Innovations in peripheral vascular devices, such as miniaturized imaging systems, low-profile catheters, balloons, and atherectomy devices, have enabled even the most complex procedures to be performed in ASC settings. Additionally, favorable reimbursement policies, physician ownership models, increasing patient preference for same-day discharge, and the rising incidence of peripheral artery disease (PAD) are all contributing to the accelerated adoption of ASCs in this market.
"The Asia Pacific is estimated to register the highest CAGR during the forecast period."
The primary driver of the peripheral vascular devices market in the APAC region is the rapidly increasing incidence of peripheral artery disease (PAD) and diabetes. This trend is supported by a large aging population and a global shift towards healthier lifestyles. Countries like China, India, and Japan are leading the way in adopting these devices, driven by developing healthcare infrastructure, rising healthcare expenditures, and improved access to advanced endovascular procedures. Additionally, growing awareness of the benefits of minimally invasive interventions, an increasing number of skilled interventional specialists, and a gradual but steady expansion of reimbursement coverage in key markets are also contributing to market growth. Furthermore, low-cost manufacturing, the presence of local companies, and the strategic expansion of global medical device firms are significant drivers of this market's growth in the APAC region.
Key Players
Some of the major players in this market are Boston Scientific (US), Medtronic Plc (US), Abbott (US), Becton, Dickinson and Company (US), Terumo Corporation (Japan), B. Braun Melsungen AG (Germany), Merit Medical Systems (US), Penumbra, Inc. (US), Koninklijke Philips (Netherlands), Stryker (US), Cordis (US), W.L. Gore & Associates (US), Biotronik Stryker (US), Endologix LLC (US), and Cook Medical (US), among others.
Research Coverage
The report analyzes the peripheral vascular devices market, estimating market size and future growth potential across various segments. These segments include angioplasty stents, endovascular aneurysm repair stent grafts, inferior vena cava filters, catheters, angioplasty balloons, plaque modification devices, hemodynamic flow alteration devices, other devices, and end users. Additionally, the report includes a product portfolio matrix of various peripheral vascular devices available in the market. It also provides a competitive analysis of the key players in this market, detailing their company profiles, product offerings, and key market strategies.
Reasons to Buy the Report
This report will provide market leaders and new entrants with valuable revenue estimates for the overall peripheral vascular devices market and its subsegments. It will help stakeholders understand the competitive landscape, enabling them to better position their businesses and develop effective go-to-market strategies. Additionally, the report will offer insights into the current market dynamics, highlighting key drivers, restraints, challenges, and opportunities.
This report provides insights into the following pointers:
- Analysis of key drivers (rapid growth in geriatric population and resulting increase in disease prevalence and favorable reimbursement scenario for peripheral vascular procedures), restraints (availability of alternative treatment options and shortage of skilled professionals with expertise in peripheral vascular devices), opportunities (high growth potential in emerging markets and growing incidence of obesity & diabetes), and challenges (product failures and device recalls).
- Product Developments/Enhancements: Comprehensive details about new product launches and anticipated trends in the global peripheral vascular devices market.
- Product Innovation: Detailed insights on upcoming trends, research & development activities, and new product launches in the global peripheral vascular devices market.
- Market Development: Comprehensive information on the lucrative emerging markets by angioplasty stents, structural heart devices, catheters, angioplasty balloons, plaque modification devices, hemodynamic flow alteration devices, other devices, and end users.
- Market Diversification: Exhaustive information about new products and services or product and service enhancements, growing geographies, recent developments, and investments in the global peripheral vascular devices market.
- Competitive Assessment: Thorough evaluation of the market shares, growth plans, offerings of products and services, and capacities of the major competitors in the global peripheral vascular devices market.
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 STUDY OBJECTIVES
- 1.2 MARKET DEFINITION
- 1.3 MARKET SCOPE
- 1.3.1 MARKET SEGMENTATION
- 1.3.2 INCLUSIONS & EXCLUSIONS
- 1.3.3 YEARS CONSIDERED
- 1.3.4 CURRENCY CONSIDERED
- 1.4 MARKET STAKEHOLDERS
- 1.5 SUMMARY OF CHANGES
2 EXECUTIVE SUMMARY
- 2.1 MARKET HIGHLIGHTS AND KEY INSIGHTS
- 2.2 KEY MARKET PARTICIPANTS: MAPPING OF STRATEGIC DEVELOPMENTS
- 2.3 DISRUPTIVE TRENDS IN PERIPHERAL VASCULAR DEVICES MARKET
- 2.4 HIGH GROWTH SEGMENTS
- 2.5 REGIONAL SNAPSHOT: MARKET SIZE, GROWTH RATE, AND FORECAST
3 PREMIUM INSIGHTS
- 3.1 PERIPHERAL VASCULAR DEVICES MARKET OVERVIEW
- 3.2 ASIA PACIFIC: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE AND COUNTRY
- 3.3 PERIPHERAL VASCULAR DEVICES MARKET: GEOGRAPHIC GROWTH OPPORTUNITIES
- 3.4 PERIPHERAL VASCULAR DEVICES MARKET: DEVELOPED VS. EMERGING ECONOMIES
4 MARKET OVERVIEW
- 4.1 INTRODUCTION
- 4.2 MARKET DYNAMICS
- 4.2.1 DRIVERS
- 4.2.1.1 Rapid growth in geriatric population
- 4.2.1.2 Favorable reimbursement scenario for procedures
- 4.2.1.3 Increasing number of product approvals
- 4.2.1.4 Rising prevalence of diabetes
- 4.2.1.5 Growth in tobacco consumption
- 4.2.1.6 Rising obesity rates
- 4.2.2 RESTRAINTS
- 4.2.2.1 Increasing number of product failures and recalls
- 4.2.2.2 Regulatory complexity and approval delays
- 4.2.3 OPPORTUNITIES
- 4.2.3.1 High growth potential in emerging markets
- 4.2.3.2 Increasing awareness and early diagnosis of PAD
- 4.2.4 CHALLENGES
- 4.2.4.1 Limited availability of alternative treatments
- 4.2.4.2 Dearth of skilled professionals with expertise in peripheral vascular devices
- 4.2.1 DRIVERS
- 4.3 UNMET NEEDS AND WHITE SPACES
- 4.4 INTERCONNECTED MARKETS AND CROSS-SECTOR OPPORTUNITIES
- 4.5 STRATEGIC MOVES BY TIER-1/2/3 PLAYERS
5 INDUSTRY TRENDS
- 5.1 PORTER'S FIVE FORCES ANALYSIS
- 5.1.1 THREAT OF NEW ENTRANTS
- 5.1.2 THREAT OF SUBSTITUTES
- 5.1.3 BARGAINING POWER OF SUPPLIERS
- 5.1.4 BARGAINING POWER OF BUYERS
- 5.1.5 INTENSITY OF COMPETITIVE RIVALRY
- 5.2 MACROECONOMICS INDICATORS
- 5.2.1 INTRODUCTION
- 5.2.2 GDP TRENDS AND FORECAST
- 5.2.3 TRENDS IN GLOBAL HEALTHCARE INDUSTRY
- 5.2.4 TRENDS IN GLOBAL MEDICAL DEVICE INDUSTRY
- 5.3 SUPPLY CHAIN ANALYSIS
- 5.4 VALUE CHAIN ANALYSIS
- 5.5 ECOSYSTEM ANALYSIS
- 5.6 PRICING ANALYSIS
- 5.6.1 AVERAGE SELLING PRICE TREND OF PRODUCTS, BY KEY PLAYERS, 2023-2025
- 5.6.2 AVERAGE SELLING PRICE TREND OF PRODUCTS, BY REGION, 2023-2025
- 5.7 TRADE ANALYSIS
- 5.7.1 IMPORT DATA FOR HS CODE 901839
- 5.7.2 EXPORT DATA FOR HS CODE 901839
- 5.8 KEY CONFERENCES AND EVENTS, 2026-2027
- 5.9 TRENDS/DISRUPTIONS IMPACTING CUSTOMER BUSINESS
- 5.10 INVESTMENT AND FUNDING SCENARIO
- 5.11 CASE STUDY ANALYSIS
- 5.11.1 MEDTRONIC
- 5.11.2 TERUMO
- 5.11.3 BOSTON SCIENTIFIC CORPORATION
- 5.12 IMPACT OF 2025 US TARIFFS - PERIPHERAL VASCULAR DEVICES MARKET
- 5.12.1 INTRODUCTION
- 5.12.2 KEY TARIFF RATES
- 5.12.3 PRICE IMPACT ANALYSIS
- 5.12.4 IMPACT ON COUNTRIES/REGIONS
- 5.12.4.1 US
- 5.12.4.2 Europe
- 5.12.4.3 Asia Pacific
- 5.12.5 IMPACT ON END-USE INDUSTRIES
- 5.12.5.1 Hospitals
- 5.12.5.1.1 Large chain hospitals
- 5.12.5.1.2 Standalone hospital facilities
- 5.12.5.2 Ambulatory care settings
- 5.12.5.3 Cardiac care centers
- 5.12.5.4 Other end users
- 5.12.5.1 Hospitals
6 TECHNOLOGICAL ADVANCEMENTS, AI-DRIVEN IMPACT, PATENTS, INNOVATIONS, AND FUTURE APPLICATIONS
- 6.1 TECHNOLOGY ANALYSIS
- 6.1.1 KEY EMERGING TECHNOLOGIES
- 6.1.1.1 Drug-eluting stents
- 6.1.1.2 Atherectomy devices
- 6.1.2 COMPLEMENTARY TECHNOLOGIES
- 6.1.2.1 Intravascular ultrasound
- 6.1.2.2 Optical coherence tomography
- 6.1.3 ADJACENT TECHNOLOGIES
- 6.1.3.1 Remote patient monitoring
- 6.1.3.2 3D printing and patient-specific modeling
- 6.1.1 KEY EMERGING TECHNOLOGIES
- 6.2 TECHNOLOGY/PRODUCT ROADMAP
- 6.3 PATENT ANALYSIS
- 6.3.1 INSIGHTS ON PATENT PUBLICATION TRENDS, TOP APPLICANTS AND JURISDICTION FOR PERIPHERAL VASCULAR DEVICES MARKET, JANUARY 2015-DECEMBER 2025
- 6.3.2 LIST OF MAJOR PATENTS, 2023-2026
- 6.4 FUTURE APPLICATIONS
- 6.4.1 INTRAVASCULAR LITHOTRIPSY FOR CALCIFIED PAD LESIONS
- 6.4.2 ADVANCED IMAGE-GUIDED INTERVENTIONS
- 6.4.3 SMART SENSOR-INTEGRATED VASCULAR IMPLANTS
- 6.4.4 INTEGRATED MULTI-FUNCTION ENDOVASCULAR DEVICES
- 6.5 IMPACT OF AI ON PERIPHERAL VASCULAR DEVICES MARKET
7 REGULATORY LANDSCAPE AND SUSTAINABILITY INITIATIVES
- 7.1 REGULATIONS AND COMPLIANCE
- 7.1.1 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- 7.1.2 INDUSTRY STANDARDS
- 7.1.2.1 Medical Device Quality Management Standard (ISO 13485)
- 7.1.2.2 Biological Evaluation and Biocompatibility of Medical Devices (ISO 10993)
- 7.1.2.3 Vascular Stent Performance Standard (ISO 25539-2)
- 7.1.2.4 Intravascular Access Device Standard (ISO 11070)
- 7.2 SUSTAINABILITY INITIATIVES
- 7.2.1 SUSTAINABLE PACKAGING AND DIGITAL INSTRUCTIONS (BOSTON SCIENTIFIC CASE STUDY)
- 7.2.2 SUSTAINABLE PRODUCT LIFECYCLE MANAGEMENT (ABBOTT CASE STUDY)
- 7.2.3 PACKAGING WASTE REDUCTION INITIATIVE BY MEDTRONIC
- 7.3 CERTIFICATIONS, LABELING, ECO-STANDARDS
8 CUSTOMER LANDSCAPE AND BUYER BEHAVIOR
- 8.1 DECISION-MAKING PROCESS
- 8.2 KEY STAKEHOLDERS INVOLVED IN BUYING PROCESS AND THEIR EVALUATION CRITERIA
- 8.2.1 KEY STAKEHOLDERS IN BUYING PROCESS
- 8.2.2 BUYING CRITERIA
- 8.3 ADOPTION BARRIERS AND INTERNAL CHALLENGES
- 8.4 UNMET NEEDS IN VARIOUS END-USE SETTINGS
- 8.5 MARKET PROFITABILITY
- 8.5.1 SHIFT TOWARD HIGH-VALUE, DRUG-ELUTING, AND SPECIALTY DEVICES
- 8.5.2 PROCEDURE VOLUME GROWTH DRIVEN BY AGING POPULATION AND PAD PREVALENCE
- 8.5.3 COST OPTIMIZATION THROUGH SUPPLY CHAIN EFFICIENCY AND PORTFOLIO BREADTH
9 PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE
- 9.1 INTRODUCTION
- 9.2 ANGIOPLASTY STENTS MARKET, BY TYPE
- 9.2.1 VOLUME ANALYSIS: ANGIOPLASTY STENTS MARKET, BY TYPE
- 9.2.2 DRUG-ELUTING STENTS
- 9.2.2.1 Drug-eluting stents to witness higher adoption due to technological advancements leading to lower restenosis rate
- 9.2.3 BARE-METAL STENTS
- 9.2.3.1 Bare-metal stents, by delivery platform
- 9.2.3.1.1 Balloon-expandable stents
- 9.2.3.1.1.1 Better radial strength and ease of deployment to drive usage
- 9.2.3.1.2 Self-expandable stents
- 9.2.3.1.2.1 Flexibility, tolerance, and support for vessel movement and compression to propel market
- 9.2.3.1.1 Balloon-expandable stents
- 9.2.3.1 Bare-metal stents, by delivery platform
- 9.3 ENDOVASCULAR ANEURYSM REPAIR STENT GRAFTS MARKET, BY TYPE
- 9.3.1 ABDOMINAL AORTIC ANEURYSM STENT GRAFTS
- 9.3.1.1 Rising prevalence of AAA and need for less invasive treatment to drive market
- 9.3.2 THORACIC AORTIC ANEURYSM STENT GRAFTS
- 9.3.2.1 Increasing incidence of thoracic aortic aneurysms to propel adoption
- 9.3.1 ABDOMINAL AORTIC ANEURYSM STENT GRAFTS
- 9.4 CATHETERS MARKET, BY TYPE
- 9.4.1 ANGIOGRAPHY CATHETERS
- 9.4.1.1 Angiography catheters to dominate catheters market
- 9.4.2 GUIDING CATHETERS
- 9.4.2.1 Growing number of target procedures to drive demand for guiding catheters
- 9.4.3 IVUS/OCT CATHETERS
- 9.4.3.1 IVUS/OCT catheters segment to register highest growth
- 9.4.1 ANGIOGRAPHY CATHETERS
- 9.5 INFERIOR VENA CAVA FILTERS MARKET, BY TYPE
- 9.5.1 RETRIEVABLE FILTERS
- 9.5.1.1 Retrievable filters segment to register higher growth due to increase in use as temporary measure to block pulmonary embolism
- 9.5.2 PERMANENT FILTERS
- 9.5.2.1 Advances in other technologies and risks associated with permanent filters to hamper sales
- 9.5.1 RETRIEVABLE FILTERS
- 9.6 ANGIOPLASTY BALLOONS MARKET, BY TYPE
- 9.6.1 OLD/NORMAL BALLOONS
- 9.6.1.1 Old/normal balloons to dominate angioplasty balloons market as they remain effective and widely used
- 9.6.2 DRUG-ELUTING BALLOONS
- 9.6.2.1 Drug-eluting balloons to witness highest growth with increasing demand for angioplasty procedures and rising regulatory approvals for DEBs
- 9.6.3 CUTTING & SCORING BALLOONS
- 9.6.3.1 Preference for medication as primary treatment for atherosclerosis to restrain adoption
- 9.6.1 OLD/NORMAL BALLOONS
- 9.7 PLAQUE MODIFICATION DEVICES MARKET, BY TYPE
- 9.7.1 ATHERECTOMY DEVICES
- 9.7.1.1 Increase in incidence of atherosclerosis to drive market
- 9.7.2 THROMBECTOMY DEVICES
- 9.7.2.1 Growing DVT incidence to drive adoption of thrombectomy devices
- 9.7.1 ATHERECTOMY DEVICES
- 9.8 HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE
- 9.8.1 EMBOLIC PROTECTION DEVICES
- 9.8.1.1 Investments in technological advancements and growing regulatory approvals to drive market
- 9.8.2 CHRONIC TOTAL OCCLUSION DEVICES
- 9.8.2.1 Rise in support for device development especially in addressing longstanding plaque buildup to drive market
- 9.8.1 EMBOLIC PROTECTION DEVICES
- 9.9 OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE
- 9.9.1 GUIDEWIRES
- 9.9.1.1 Higher success rate during stenting and endovascular aneurysm repair contribute to largest market share
- 9.9.2 VASCULAR CLOSURE DEVICES
- 9.9.2.1 Technological advancements and minimized duration of hospital stay to support demand
- 9.9.3 INTRODUCER SHEATHS
- 9.9.3.1 Potential for reducing arterial damage during interventional procedures to drive adoption
- 9.9.4 BALLOON INFLATION DEVICES
- 9.9.4.1 Integrated technologies and rising number of target procedures to propel adoption
- 9.9.1 GUIDEWIRES
10 PERIPHERAL VASCULAR DEVICES MARKET, BY END USER
- 10.1 INTRODUCTION
- 10.2 HOSPITALS
- 10.2.1 ADVANCED INFRASTRUCTURE AND SPECIALIZED EQUIPMENT TO DRIVE SEGMENT GROWTH
- 10.2.2 LARGE CHAIN HOSPITALS
- 10.2.2.1 Strong infrastructure and integrated networks driving dominance in complex peripheral vascular procedures
- 10.2.3 STANDALONE HOSPITAL FACILITIES
- 10.2.3.1 Expanding accessibility and cost-effective care to support peripheral vascular procedure volumes
- 10.3 AMBULATORY SURGICAL CENTERS
- 10.3.1 FASTER PROCEDURE TIME AND LOWER INFECTION RATES TO DRIVE SEGMENT GROWTH
- 10.4 OTHER END USERS
11 PERIPHERAL VASCULAR DEVICES MARKET, BY REGION
- 11.1 INTRODUCTION
- 11.2 NORTH AMERICA
- 11.2.1 MACROECONOMIC OUTLOOK FOR NORTH AMERICA
- 11.2.2 NORTH AMERICA ANGIOPLASTY STENTS MARKET: VOLUME ANALYSIS, BY TYPE
- 11.2.3 US
- 11.2.3.1 Favorable reimbursement scenario to contribute to market growth
- 11.2.4 CANADA
- 11.2.4.1 Increasing cases of chronic conditions to fuel market
- 11.3 EUROPE
- 11.3.1 MACROECONOMIC OUTLOOK FOR EUROPE
- 11.3.2 EUROPE ANGIOPLASTY STENTS MARKET: VOLUME ANALYSIS, BY TYPE
- 11.3.3 GERMANY
- 11.3.3.1 Statutory health insurance policy to support market growth
- 11.3.4 FRANCE
- 11.3.4.1 Growing geriatric population to drive market
- 11.3.5 UK
- 11.3.5.1 Increasing volume of coronary angioplasty procedures to sustain market growth
- 11.3.6 SPAIN
- 11.3.6.1 Growing cases of diabetes and obesity to boost market
- 11.3.7 ITALY
- 11.3.7.1 Increasing prevalence of atherosclerotic cardiovascular disease to spur market growth
- 11.3.8 REST OF EUROPE
- 11.4 ASIA PACIFIC
- 11.4.1 MACROECONOMIC OUTLOOK FOR ASIA PACIFIC
- 11.4.2 ASIA PACIFIC ANGIOPLASTY STENTS MARKET: VOLUME ANALYSIS, BY TYPE
- 11.4.3 JAPAN
- 11.4.3.1 Large geriatric population to ensure sustained demand
- 11.4.4 CHINA
- 11.4.4.1 Growing incidence of lifestyle diseases to fuel market
- 11.4.5 INDIA
- 11.4.5.1 Rising prevalence of target diseases and growing healthcare expenditure to propel market
- 11.4.6 AUSTRALIA
- 11.4.6.1 High prevalence of cardiovascular and peripheral artery diseases to drive market
- 11.4.7 SOUTH KOREA
- 11.4.7.1 Rising rates of chronic diseases to boost market
- 11.4.8 REST OF ASIA PACIFIC
- 11.5 LATIN AMERICA
- 11.5.1 MACROECONOMIC OUTLOOK FOR LATIN AMERICA
- 11.5.2 LATIN AMERICA ANGIOPLASTY STENTS MARKET: VOLUME ANALYSIS, BY TYPE
- 11.5.3 BRAZIL
- 11.5.3.1 Rising healthcare expenditure to aid market growth
- 11.5.4 MEXICO
- 11.5.4.1 Increasing prevalence of cardiovascular diseases to spur market growth
- 11.5.5 COLOMBIA
- 11.5.5.1 Growing incidence of peripheral artery diseases to expedite market growth
- 11.5.6 ARGENTINA
- 11.5.6.1 Increasing prevalence of peripheral artery diseases to support market growth
- 11.5.7 REST OF LATIN AMERICA
- 11.6 MIDDLE EAST & AFRICA
- 11.6.1 MACROECONOMIC OUTLOOK FOR MIDDLE EAST & AFRICA
- 11.6.2 MIDDLE EAST & AFRICA ANGIOPLASTY STENTS MARKET: VOLUME ANALYSIS, BY TYPE
- 11.6.3 GCC COUNTRIES
- 11.6.3.1 Kingdom of Saudi Arabia (KSA)
- 11.6.3.1.1 Rising diabetes prevalence and strong government healthcare investments to drive market
- 11.6.3.2 United Arab Emirates (UAE)
- 11.6.3.2.1 Advanced healthcare infrastructure and growing medical tourism to accelerate peripheral vascular devices adoption
- 11.6.3.1 Kingdom of Saudi Arabia (KSA)
- 11.6.4 REST OF GCC COUNTRIES
- 11.6.5 REST OF MIDDLE EAST & AFRICA
12 COMPETITIVE LANDSCAPE
- 12.1 INTRODUCTION
- 12.2 KEY PLAYER STRATEGIES/RIGHT TO WIN
- 12.2.1 OVERVIEW OF STRATEGIES ADOPTED BY KEY PLAYERS
- 12.3 REVENUE ANALYSIS
- 12.4 MARKET SHARE ANALYSIS
- 12.4.1 GLOBAL: PERIPHERAL VASCULAR DEVICES MARKET SHARE ANALYSIS, 2025
- 12.4.2 US: PERIPHERAL VASCULAR DEVICES MARKET SHARE ANALYSIS, 2025
- 12.5 COMPANY EVALUATION MATRIX: KEY PLAYERS, 2025
- 12.5.1 STARS
- 12.5.2 EMERGING LEADERS
- 12.5.3 PERVASIVE PLAYERS
- 12.5.4 PARTICIPANTS
- 12.5.5 COMPANY FOOTPRINT: KEY PLAYERS, 2025
- 12.5.5.1 Company footprint
- 12.5.5.2 Regional footprint
- 12.5.5.3 Product type footprint
- 12.5.5.4 End user footprint
- 12.6 COMPANY EVALUATION MATRIX: STARTUPS/SMES, 2025
- 12.6.1 PROGRESSIVE COMPANIES
- 12.6.2 DYNAMIC COMPANIES
- 12.6.3 RESPONSIVE COMPANIES
- 12.6.4 STARTING BLOCKS
- 12.6.5 COMPETITIVE BENCHMARKING: STARTUPS/SMES, 2025
- 12.7 BRAND/PRODUCT COMPARISON
- 12.8 COMPANY VALUATION AND FINANCIAL METRICS
- 12.8.1 FINANCIAL METRICS
- 12.8.2 COMPANY VALUATION
- 12.9 COMPETITIVE SCENARIO
- 12.9.1 PRODUCT LAUNCHES AND APPROVALS
- 12.9.2 DEALS
13 COMPANY PROFILES
- 13.1 KEY PLAYERS
- 13.1.1 BOSTON SCIENTIFIC CORPORATION
- 13.1.1.1 Business overview
- 13.1.1.2 Products offered
- 13.1.1.3 Recent developments
- 13.1.1.3.1 Product approvals
- 13.1.1.3.2 Deals
- 13.1.1.3.3 Other developments
- 13.1.1.4 MnM view
- 13.1.1.4.1 Right to win
- 13.1.1.4.2 Strategic choices
- 13.1.1.4.3 Weaknesses and competitive threats
- 13.1.2 BECTON, DICKINSON AND COMPANY
- 13.1.2.1 Business overview
- 13.1.2.2 Products offered
- 13.1.2.3 Recent developments
- 13.1.2.3.1 Product launches and approvals
- 13.1.2.3.2 Deals
- 13.1.2.3.3 Expansions
- 13.1.2.3.4 Other developments
- 13.1.2.4 MnM view
- 13.1.2.4.1 Right to win
- 13.1.2.4.2 Strategic choices
- 13.1.2.4.3 Weaknesses and competitive threats
- 13.1.3 ABBOTT LABORATORIES
- 13.1.3.1 Business overview
- 13.1.3.2 Products offered
- 13.1.3.3 Recent developments
- 13.1.3.3.1 Product launches
- 13.1.3.3.2 Deals
- 13.1.3.3.3 Expansions
- 13.1.3.4 MnM view
- 13.1.3.4.1 Right to win
- 13.1.3.4.2 Strategic choices made
- 13.1.3.4.3 Weaknesses and competitive threats
- 13.1.4 CARDINAL HEALTH, INC.
- 13.1.4.1 Business overview
- 13.1.4.2 Products offered
- 13.1.4.3 Recent developments
- 13.1.4.3.1 Product launches
- 13.1.4.3.2 Deals
- 13.1.4.3.3 Expansions
- 13.1.4.4 MnM view
- 13.1.4.4.1 Right to win
- 13.1.4.4.2 Strategic choices
- 13.1.4.4.3 Weaknesses and competitive threats
- 13.1.5 KONINKLIJKE PHILIPS N.V.
- 13.1.5.1 Business overview
- 13.1.5.2 Products offered
- 13.1.5.3 Recent developments
- 13.1.5.3.1 Product approvals
- 13.1.5.3.2 Deals
- 13.1.5.3.3 Expansions
- 13.1.5.4 MnM view
- 13.1.5.4.1 Right to win
- 13.1.5.4.2 Strategic choices
- 13.1.5.4.3 Weaknesses and competitive threats
- 13.1.6 TERUMO CORPORATION
- 13.1.6.1 Business overview
- 13.1.6.2 Products offered
- 13.1.6.3 Recent developments
- 13.1.6.3.1 Product launches
- 13.1.6.3.2 Deals
- 13.1.6.3.3 Expansions
- 13.1.7 MERIT MEDICAL SYSTEMS, INC.
- 13.1.7.1 Business overview
- 13.1.7.2 Products offered
- 13.1.7.3 Recent developments
- 13.1.7.3.1 Product launches
- 13.1.7.3.2 Deals
- 13.1.8 B. BRAUN SE
- 13.1.8.1 Business overview
- 13.1.8.2 Products offered
- 13.1.8.3 Recent developments
- 13.1.8.3.1 Product approvals
- 13.1.9 MEDTRONIC
- 13.1.9.1 Business overview
- 13.1.9.2 Products offered
- 13.1.9.3 Recent developments
- 13.1.9.3.1 Product launches and approvals
- 13.1.9.3.2 Deals
- 13.1.10 PENUMBRA, INC.
- 13.1.10.1 Business overview
- 13.1.10.2 Products offered
- 13.1.10.3 Recent developments
- 13.1.10.3.1 Product launches and approvals
- 13.1.11 STRYKER CORPORATION
- 13.1.11.1 Business overview
- 13.1.11.2 Products offered
- 13.1.11.3 Recent developments
- 13.1.11.3.1 Deals
- 13.1.12 CORDIS
- 13.1.12.1 Business overview
- 13.1.12.2 Products offered
- 13.1.12.3 Recent developments
- 13.1.12.3.1 Product approvals
- 13.1.12.3.2 Deals
- 13.1.13 COOK
- 13.1.13.1 Business overview
- 13.1.13.2 Products offered
- 13.1.13.3 Recent developments
- 13.1.13.3.1 Deals
- 13.1.14 W. L. GORE & ASSOCIATES, INC.
- 13.1.14.1 Business overview
- 13.1.14.2 Products offered
- 13.1.14.3 Recent developments
- 13.1.14.3.1 Product approvals
- 13.1.15 BIOSENSORS INTERNATIONAL GROUP, LTD.
- 13.1.15.1 Business overview
- 13.1.15.2 Products offered
- 13.1.16 IVASCULAR
- 13.1.16.1 Business overview
- 13.1.16.2 Products offered
- 13.1.17 BIOTRONIK
- 13.1.17.1 Business overview
- 13.1.17.2 Products offered
- 13.1.17.3 Recent developments
- 13.1.17.3.1 Product launches
- 13.1.18 ENDOLOGIX
- 13.1.18.1 Business overview
- 13.1.18.2 Products offered
- 13.1.19 ENDOCOR GMBH & CO., KG
- 13.1.19.1 Business overview
- 13.1.19.2 Products offered
- 13.1.1 BOSTON SCIENTIFIC CORPORATION
- 13.2 OTHER PLAYERS
- 13.2.1 MERIL LIFE SCIENCES PVT. LTD.
- 13.2.2 ALVIMEDICA
- 13.2.3 CARDIONOVUM GMBH
- 13.2.4 SMT
- 13.2.5 MEDINOL
- 13.2.6 ANDRAMED GMBH
- 13.2.7 REX MEDICAL
- 13.2.8 QMD
- 13.2.9 BROSMED MEDICAL CO., LTD.
- 13.2.10 ELIXIR MEDICAL
14 RESEARCH METHODOLOGY
- 14.1 RESEARCH DATA
- 14.1.1 SECONDARY DATA
- 14.1.1.1 Key data from secondary sources
- 14.1.2 PRIMARY DATA
- 14.1.2.1 Key data from primary sources
- 14.1.2.2 Key industry insights
- 14.1.2.3 Key industry insights
- 14.1.1 SECONDARY DATA
- 14.2 MARKET SIZE ESTIMATION
- 14.3 GROWTH FORECAST
- 14.3.1 KEY INDUSTRY INSIGHTS
- 14.4 MARKET BREAKDOWN AND DATA TRIANGULATION
- 14.5 RESEARCH ASSUMPTIONS
- 14.6 RISK ASSESSMENT
- 14.7 RESEARCH LIMITATIONS
15 APPENDIX
- 15.1 DISCUSSION GUIDE
- 15.2 KNOWLEDGESTORE: MARKETSANDMARKETS' SUBSCRIPTION PORTAL
- 15.3 CUSTOMIZATION OPTIONS
- 15.3.1 PRODUCT ANALYSIS
- 15.3.2 US: END-USER ANALYSIS
- 15.3.3 GEOGRAPHIC ANALYSIS
- 15.3.4 COMPANY INFORMATION
- 15.3.5 REGIONAL/COUNTRY-LEVEL MARKET SHARE ANALYSIS
- 15.3.6 COUNTRY-LEVEL VOLUME ANALYSIS BY PRODUCT TYPE
- 15.3.7 MARKET SHARE ANALYSIS, BY PRODUCT TYPE (TOP FIVE PLAYERS)
- 15.3.8 ANY CONSULTS/CUSTOM REQUIREMENTS AS PER CLIENT REQUEST
- 15.4 RELATED REPORTS
- 15.5 AUTHOR DETAILS
List of Tables
- TABLE 1 INCLUSIONS & EXCLUSIONS
- TABLE 2 STANDARD CURRENCY CONVERSION RATES
- TABLE 3 GLOBAL GERIATRIC POPULATION TRENDS (2015 VS. 2030): REGIONAL BREAKDOWN
- TABLE 4 US: REIMBURSEMENT FOR PERIPHERAL VASCULAR PROCEDURES, 2026 (USD)
- TABLE 5 PRODUCT APPROVALS IN PERIPHERAL VASCULAR DEVICES MARKET, 2023-2024
- TABLE 6 DIABETES POPULATION, BY REGION, 2024 VS. 2050
- TABLE 7 TOP 10 COUNTRIES WITH HIGHEST SMOKING RATE, 2025
- TABLE 8 OBESITY RATE IN DEVELOPING COUNTRIES, 2026
- TABLE 9 PRODUCT FAILURES AND RECALLS
- TABLE 10 UNMET NEEDS AND WHITE SPACES
- TABLE 11 INTERCONNECTED MARKETS
- TABLE 12 CROSS-SECTOR OPPORTUNITIES
- TABLE 13 STRATEGIC MOVES BY TIER-1/2/3 PLAYERS
- TABLE 14 IMPACT OF PORTER'S FIVE FORCES ON PERIPHERAL VASCULAR DEVICES MARKET
- TABLE 15 AVERAGE SELLING PRICE TREND OF PRODUCTS, BY KEY PLAYER, 2023-2025 (USD)
- TABLE 16 AVERAGE SELLING PRICE TREND OF PRODUCTS, BY REGION, 2023-2025 (USD)
- TABLE 17 IMPORT DATA FOR HS CODE 901839-COMPLIANT PRODUCTS, BY COUNTRY, 2021-2025 (USD MILLION)
- TABLE 18 EXPORT DATA FOR HS CODE 901839-COMPLIANT PRODUCTS, BY COUNTRY, 2021-2025 (USD MILLION)
- TABLE 19 PERIPHERAL VASCULAR DEVICES MARKET: LIST OF KEY CONFERENCES AND EVENTS, 2026-2027
- TABLE 20 US ADJUSTED RECIPROCAL TARIFF RATES
- TABLE 21 PERIPHERAL VASCULAR DEVICES MARKET: TECHNOLOGY/PRODUCT ROADMAP (2026-2035)
- TABLE 22 PERIPHERAL VASCULAR DEVICES MARKET: LIST OF MAJOR PATENTS, JANUARY 2023-FEBRUARY 2026
- TABLE 23 AI INTEGRATION: PERIPHERAL VASCULAR DEVICES MARKET
- TABLE 24 NORTH AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 25 EUROPE: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 26 ASIA PACIFIC: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 27 LATIN AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 28 MIDDLE EAST & AFRICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 29 INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS FOR PERIPHERAL VASCULAR DEVICES
- TABLE 30 KEY BUYING CRITERIA FOR TOP THREE END USERS
- TABLE 31 PERIPHERAL VASCULAR DEVICES MARKET: UNMET NEEDS IN KEY END-USE SETTINGS
- TABLE 32 PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 33 ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (THOUSAND UNITS)
- TABLE 34 ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 35 ANGIOPLASTY STENTS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 36 DRUG-ELUTING STENTS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 37 BARE-METAL STENTS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 38 BARE-METAL STENTS MARKET, BY DELIVERY PLATFORM, 2024-2031 (USD MILLION)
- TABLE 39 BALLOON-EXPANDABLE STENTS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 40 SELF-EXPANDABLE STENTS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 41 EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 42 EVAR STENT GRAFTS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 43 AAA STENT GRAFTS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 44 TAA STENT GRAFTS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 45 CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 46 CATHETERS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 47 ANGIOGRAPHY CATHETERS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 48 GUIDING CATHETERS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 49 IVUS/OCT CATHETERS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 50 INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 51 INFERIOR VENA CAVA FILTERS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 52 RETRIEVABLE INFERIOR VENA CAVA FILTERS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 53 PERMANENT INFERIOR VENA CAVA FILTERS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 54 ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 55 ANGIOPLASTY BALLOONS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 56 OLD/NORMAL BALLOONS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 57 DRUG-ELUTING BALLOONS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 58 CUTTING & SCORING BALLOONS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 59 PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 60 PLAQUE MODIFICATION DEVICES MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 61 ATHERECTOMY DEVICES MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 62 THROMBECTOMY DEVICES MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 63 HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 64 HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 65 EMBOLIC PROTECTION DEVICES MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 66 CHRONIC TOTAL OCCLUSION DEVICES MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 67 OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 68 OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 69 GUIDEWIRES MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 70 VASCULAR CLOSURE DEVICES MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 71 INTRODUCER SHEATHS MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 72 BALLOON INFLATION DEVICES MARKET, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 73 PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 74 PERIPHERAL VASCULAR DEVICES MARKET FOR HOSPITALS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 75 PERIPHERAL VASCULAR DEVICES MARKET FOR HOSPITALS, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 76 PERIPHERAL VASCULAR DEVICES MARKET FOR LARGE CHAIN HOSPITALS, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 77 PERIPHERAL VASCULAR DEVICES MARKET FOR STANDALONE HOSPITAL FACILITIES SEGMENT, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 78 PERIPHERAL VASCULAR DEVICES MARKET FOR AMBULATORY SURGICAL CENTERS, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 79 PERIPHERAL VASCULAR DEVICES MARKET FOR OTHER END USERS, BY COUNTRY/REGION, 2024-2031 (USD MILLION)
- TABLE 80 PERIPHERAL VASCULAR DEVICES MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 81 NORTH AMERICA: PERIPHERAL VASCULAR DEVICES MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 82 NORTH AMERICA: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 83 NORTH AMERICA: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 84 NORTH AMERICA: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (THOUSAND UNITS)
- TABLE 85 NORTH AMERICA: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 86 NORTH AMERICA: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 87 NORTH AMERICA: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 88 NORTH AMERICA: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 89 NORTH AMERICA: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 90 NORTH AMERICA: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 91 NORTH AMERICA: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 92 NORTH AMERICA: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 93 NORTH AMERICA: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 94 NORTH AMERICA: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 95 US: MACROECONOMIC INDICATORS
- TABLE 96 US: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 97 US: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 98 US: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 99 US: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 100 US: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 101 US: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 102 US: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 103 US: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 104 US: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 105 US: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 106 US: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 107 US: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 108 CANADA: MACROECONOMIC INDICATORS
- TABLE 109 CANADA: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 110 CANADA: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 111 CANADA: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 112 CANADA: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 113 CANADA: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 114 CANADA: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 115 CANADA: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 116 CANADA: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 117 CANADA: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 118 CANADA: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 119 CANADA: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 120 CANADA: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 121 EUROPE: PERIPHERAL VASCULAR DEVICES MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 122 EUROPE: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 123 EUROPE: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 124 EUROPE: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (THOUSAND UNITS)
- TABLE 125 EUROPE: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 126 EUROPE: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 127 EUROPE: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 128 EUROPE: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 129 EUROPE: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 130 EUROPE: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 131 EUROPE: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 132 EUROPE: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 133 EUROPE: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 134 EUROPE: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 135 GERMANY: MACROECONOMIC INDICATORS
- TABLE 136 GERMANY: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 137 GERMANY: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 138 GERMANY: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 139 GERMANY: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 140 GERMANY: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 141 GERMANY: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 142 GERMANY: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 143 GERMANY: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 144 GERMANY: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 145 GERMANY: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 146 GERMANY: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 147 GERMANY: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 148 FRANCE: MACROECONOMIC INDICATORS
- TABLE 149 FRANCE: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 150 FRANCE: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 151 FRANCE: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 152 FRANCE: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 153 FRANCE: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 154 FRANCE: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 155 FRANCE: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 156 FRANCE: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 157 FRANCE: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 158 FRANCE: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 159 FRANCE: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 160 FRANCE: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 161 UK: MACROECONOMIC INDICATORS
- TABLE 162 UK: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 163 UK: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 164 UK: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 165 UK: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 166 UK: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 167 UK: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 168 UK: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 169 UK: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 170 UK: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 171 UK: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 172 UK: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 173 UK: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 174 SPAIN: MACROECONOMIC INDICATORS
- TABLE 175 SPAIN: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 176 SPAIN: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 177 SPAIN: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 178 SPAIN: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 179 SPAIN: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 180 SPAIN: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 181 SPAIN: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 182 SPAIN: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 183 SPAIN: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 184 SPAIN: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 185 SPAIN: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 186 SPAIN: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 187 ITALY: MACROECONOMIC INDICATORS
- TABLE 188 ITALY: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 189 ITALY: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 190 ITALY: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 191 ITALY: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 192 ITALY: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 193 ITALY: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 194 ITALY: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 195 ITALY: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 196 ITALY: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 197 ITALY: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 198 ITALY: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 199 ITALY: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 200 REST OF EUROPE: PERCENTAGE OF GERIATRIC POPULATION TO TOTAL POPULATION, BY COUNTRY, 2015 VS. 2030
- TABLE 201 REST OF EUROPE: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 202 REST OF EUROPE: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 203 REST OF EUROPE: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 204 REST OF EUROPE: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 205 REST OF EUROPE: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 206 REST OF EUROPE: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 207 REST OF EUROPE: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 208 REST OF EUROPE: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 209 REST OF EUROPE: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 210 REST OF EUROPE: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 211 REST OF EUROPE: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 212 REST OF EUROPE: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 213 ASIA PACIFIC: PERIPHERAL VASCULAR DEVICES MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 214 ASIA PACIFIC: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 215 ASIA PACIFIC: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 216 ASIA PACIFIC: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (THOUSAND UNITS)
- TABLE 217 ASIA PACIFIC: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 218 ASIA PACIFIC: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 219 ASIA PACIFIC: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 220 ASIA PACIFIC: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 221 ASIA PACIFIC: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 222 ASIA PACIFIC: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 223 ASIA PACIFIC: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 224 ASIA PACIFIC: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 225 ASIA PACIFIC: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 226 ASIA PACIFIC: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 227 JAPAN: MACROECONOMIC INDICATORS
- TABLE 228 JAPAN: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 229 JAPAN: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 230 JAPAN: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 231 JAPAN: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 232 JAPAN: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 233 JAPAN: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 234 JAPAN: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 235 JAPAN: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 236 JAPAN: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 237 JAPAN: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 238 JAPAN: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 239 JAPAN: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 240 CHINA: MACROECONOMIC INDICATORS
- TABLE 241 CHINA: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 242 CHINA: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 243 CHINA: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 244 CHINA: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 245 CHINA: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 246 CHINA: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 247 CHINA: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 248 CHINA: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 249 CHINA: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 250 CHINA: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 251 CHINA: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 252 CHINA: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 253 INDIA: MACROECONOMIC INDICATORS
- TABLE 254 INDIA: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 255 INDIA: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 256 INDIA: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 257 INDIA: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 258 INDIA: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 259 INDIA: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 260 INDIA: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 261 INDIA: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 262 INDIA: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 263 INDIA: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 264 INDIA: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 265 INDIA: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 266 AUSTRALIA: MACROECONOMIC INDICATORS
- TABLE 267 AUSTRALIA: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 268 AUSTRALIA: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 269 AUSTRALIA: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 270 AUSTRALIA: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 271 AUSTRALIA: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 272 AUSTRALIA: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 273 AUSTRALIA: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 274 AUSTRALIA: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 275 AUSTRALIA: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 276 AUSTRALIA: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 277 AUSTRALIA: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 278 AUSTRALIA: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 279 SOUTH KOREA: MACROECONOMIC INDICATORS
- TABLE 280 SOUTH KOREA: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 281 SOUTH KOREA: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 282 SOUTH KOREA: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 283 SOUTH KOREA: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 284 SOUTH KOREA: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 285 SOUTH KOREA: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 286 SOUTH KOREA: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 287 SOUTH KOREA: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 288 SOUTH KOREA: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 289 SOUTH KOREA: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 290 SOUTH KOREA: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 291 SOUTH KOREA: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 292 REST OF ASIA PACIFIC: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 293 REST OF ASIA PACIFIC: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 294 REST OF ASIA PACIFIC: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 295 REST OF ASIA PACIFIC: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 296 REST OF ASIA PACIFIC: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 297 REST OF ASIA PACIFIC: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 298 REST OF ASIA PACIFIC: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 299 REST OF ASIA PACIFIC: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 300 REST OF ASIA PACIFIC: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 301 REST OF ASIA PACIFIC: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 302 REST OF ASIA PACIFIC: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 303 REST OF ASIA PACIFIC: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 304 LATIN AMERICA: PERIPHERAL VASCULAR DEVICES MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 305 LATIN AMERICA: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 306 LATIN AMERICA: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 307 LATIN AMERICA: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (THOUSAND UNITS)
- TABLE 308 LATIN AMERICA: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 309 LATIN AMERICA: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 310 LATIN AMERICA: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 311 LATIN AMERICA: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 312 LATIN AMERICA: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 313 LATIN AMERICA: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 314 LATIN AMERICA: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 315 LATIN AMERICA: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 316 LATIN AMERICA: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 317 LATIN AMERICA: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 318 BRAZIL: MACROECONOMIC INDICATORS
- TABLE 319 BRAZIL: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 320 BRAZIL: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 321 BRAZIL: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 322 BRAZIL: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 323 BRAZIL: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 324 BRAZIL: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 325 BRAZIL: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 326 BRAZIL: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 327 BRAZIL: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 328 BRAZIL: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 329 BRAZIL: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 330 BRAZIL: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 331 MEXICO: MACROECONOMIC INDICATORS
- TABLE 332 MEXICO: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 333 MEXICO: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 334 MEXICO: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 335 MEXICO: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 336 MEXICO: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 337 MEXICO: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 338 MEXICO: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 339 MEXICO: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 340 MEXICO: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 341 MEXICO: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 342 MEXICO: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 343 MEXICO: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 344 COLOMBIA: MACROECONOMIC INDICATORS
- TABLE 345 COLOMBIA: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 346 COLOMBIA: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 347 COLOMBIA: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 348 COLOMBIA: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 349 COLOMBIA: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 350 COLOMBIA: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 351 COLOMBIA: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 352 COLOMBIA: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 353 COLOMBIA: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 354 COLOMBIA: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 355 COLOMBIA: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 356 COLOMBIA: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 357 ARGENTINA: MACROECONOMIC INDICATORS
- TABLE 358 ARGENTINA: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 359 ARGENTINA: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 360 ARGENTINA: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 361 ARGENTINA: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 362 ARGENTINA: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 363 ARGENTINA: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 364 ARGENTINA: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 365 ARGENTINA: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 366 ARGENTINA: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 367 ARGENTINA: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 368 ARGENTINA: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 369 ARGENTINA: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 370 REST OF LATIN AMERICA: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 371 REST OF LATIN AMERICA: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 372 REST OF LATIN AMERICA: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 373 REST OF LATIN AMERICA: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 374 REST OF LATIN AMERICA: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 375 REST OF LATIN AMERICA: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 376 REST OF LATIN AMERICA: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 377 REST OF LATIN AMERICA: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 378 REST OF LATIN AMERICA: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 379 REST OF LATIN AMERICA: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 380 REST OF LATIN AMERICA: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 381 REST OF LATIN AMERICA: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 382 MIDDLE EAST & AFRICA: PERIPHERAL VASCULAR DEVICES MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 383 MIDDLE EAST & AFRICA: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 384 MIDDLE EAST & AFRICA: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 385 MIDDLE EAST & AFRICA: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (THOUSAND UNITS)
- TABLE 386 MIDDLE EAST & AFRICA: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 387 MIDDLE EAST & AFRICA: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 388 MIDDLE EAST & AFRICA: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 389 MIDDLE EAST & AFRICA: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 390 MIDDLE EAST & AFRICA: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 391 MIDDLE EAST & AFRICA: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 392 MIDDLE EAST & AFRICA: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 393 MIDDLE EAST & AFRICA: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 394 MIDDLE EAST & AFRICA: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 395 MIDDLE EAST & AFRICA: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 396 GCC COUNTRIES: PERIPHERAL VASCULAR DEVICES MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 397 GCC COUNTRIES: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 398 GCC COUNTRIES: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 399 GCC COUNTRIES: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 400 GCC COUNTRIES: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 401 GCC COUNTRIES: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 402 GCC COUNTRIES: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 403 GCC COUNTRIES: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 404 GCC COUNTRIES: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 405 GCC COUNTRIES: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 406 GCC COUNTRIES: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 407 GCC COUNTRIES: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 408 GCC COUNTRIES: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 409 KSA: MACROECONOMIC INDICATORS
- TABLE 410 KSA: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 411 KSA: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 412 KSA: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 413 KSA: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 414 KSA: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 415 KSA: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 416 KSA: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 417 KSA: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 418 KSA: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 419 KSA: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 420 KSA: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 421 KSA: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 422 UAE: MACROECONOMIC INDICATORS
- TABLE 423 UAE: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 424 UAE: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 425 UAE: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 426 UAE: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 427 UAE: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 428 UAE: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 429 UAE: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 430 UAE: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 431 UAE: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 432 UAE: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 433 UAE: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 434 UAE: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 435 REST OF GCC COUNTRIES: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 436 REST OF GCC COUNTRIES: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 437 REST OF GCC COUNTRIES: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 438 REST OF GCC COUNTRIES: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 439 REST OF GCC COUNTRIES: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 440 REST OF GCC COUNTRIES: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 441 REST OF GCC COUNTRIES: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 442 REST OF GCC COUNTRIES: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 443 REST OF GCC COUNTRIES: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 444 REST OF GCC COUNTRIES: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 445 REST OF GCC COUNTRIES: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 446 REST OF GCC COUNTRIES: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 447 REST OF MIDDLE EAST & AFRICA: PERIPHERAL VASCULAR DEVICES MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 448 REST OF MIDDLE EAST & AFRICA: ANGIOPLASTY BALLOONS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 449 REST OF MIDDLE EAST & AFRICA: ANGIOPLASTY STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 450 REST OF MIDDLE EAST & AFRICA: BARE-METAL STENTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 451 REST OF MIDDLE EAST & AFRICA: CATHETERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 452 REST OF MIDDLE EAST & AFRICA: EVAR STENT GRAFTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 453 REST OF MIDDLE EAST & AFRICA: INFERIOR VENA CAVA FILTERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 454 REST OF MIDDLE EAST & AFRICA: PLAQUE MODIFICATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 455 REST OF MIDDLE EAST & AFRICA: HEMODYNAMIC FLOW ALTERATION DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 456 REST OF MIDDLE EAST & AFRICA: OTHER PERIPHERAL VASCULAR DEVICES MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 457 REST OF MIDDLE EAST & AFRICA: PERIPHERAL VASCULAR DEVICES MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 458 REST OF MIDDLE EAST & AFRICA: HOSPITALS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 459 OVERVIEW OF STRATEGIES ADOPTED BY PLAYERS IN PERIPHERAL VASCULAR DEVICES MARKET
- TABLE 460 PERIPHERAL VASCULAR DEVICES MARKET: DEGREE OF COMPETITION
- TABLE 461 PERIPHERAL VASCULAR DEVICES MARKET: REGIONAL FOOTPRINT, 2025
- TABLE 462 PERIPHERAL VASCULAR DEVICES MARKET: PRODUCT TYPE FOOTPRINT, 2025
- TABLE 463 PERIPHERAL VASCULAR DEVICES MARKET: END USER FOOTPRINT, 2025
- TABLE 464 PERIPHERAL VASCULAR DEVICES MARKET: KEY STARTUPS/SMES, 2025
- TABLE 465 PERIPHERAL VASCULAR DEVICES MARKET: COMPETITIVE BENCHMARKING OF STARTUPS/SMES, 2025
- TABLE 466 PERIPHERAL VASCULAR DEVICES MARKET: PRODUCT LAUNCHES AND APPROVALS, JANUARY 2023-MARCH 2026
- TABLE 467 PERIPHERAL VASCULAR DEVICES MARKET: DEALS, JANUARY 2023-MARCH 2026
- TABLE 468 PERIPHERAL VASCULAR DEVICES MARKET: EXPANSIONS, JANUARY 2023-MARCH 2026
- TABLE 469 PERIPHERAL VASCULAR DEVICES MARKET: OTHER DEVELOPMENTS, JANUARY 2023-MARCH 2026
- TABLE 470 BOSTON SCIENTIFIC CORPORATION: COMPANY OVERVIEW
- TABLE 471 BOSTON SCIENTIFIC CORPORATION: PRODUCTS OFFERED
- TABLE 472 BOSTON SCIENTIFIC CORPORATION: PRODUCT APPROVALS
- TABLE 473 BOSTON SCIENTIFIC CORPORATION: DEALS
- TABLE 474 BOSTON SCIENTIFIC CORPORATION: OTHER DEVELOPMENTS
- TABLE 475 BECTON, DICKINSON AND COMPANY: COMPANY OVERVIEW
- TABLE 476 BECTON, DICKINSON AND COMPANY: PRODUCTS OFFERED
- TABLE 477 BECTON, DICKINSON AND COMPANY: PRODUCT LAUNCHES AND APPROVALS
- TABLE 478 BECTON, DICKINSON AND COMPANY: DEALS
- TABLE 479 BECTON, DICKINSON AND COMPANY: EXPANSIONS
- TABLE 480 BECTON, DICKINSON AND COMPANY: OTHER DEVELOPMENTS
- TABLE 481 ABBOTT LABORATORIES: COMPANY OVERVIEW
- TABLE 482 ABBOTT LABORATORIES: PRODUCTS OFFERED
- TABLE 483 ABBOTT LABORATORIES: PRODUCT LAUNCHES
- TABLE 484 ABBOTT LABORATORIES: DEALS
- TABLE 485 ABBOTT LABORATORIES: EXPANSIONS
- TABLE 486 CARDINAL HEALTH, INC.: COMPANY OVERVIEW
- TABLE 487 CARDINAL HEALTH, INC.: PRODUCTS OFFERED
- TABLE 488 CARDINAL HEALTH, INC.: PRODUCT LAUNCHES
- TABLE 489 CARDINAL HEALTH, INC.: DEALS
- TABLE 490 CARDINAL HEALTH, INC.: EXPANSIONS
- TABLE 491 KONINKLIJKE PHILIPS N.V.: COMPANY OVERVIEW
- TABLE 492 KONINKLIJKE PHILIPS N.V.: PRODUCTS OFFERED
- TABLE 493 KONINKLIJKE PHILIPS N.V.: PRODUCT APPROVALS
- TABLE 494 KONINKLIJKE PHILIPS N.V.: DEALS
- TABLE 495 KONINKLIJKE PHILIPS N.V.: EXPANSIONS
- TABLE 496 TERUMO CORPORATION: COMPANY OVERVIEW
- TABLE 497 TERUMO CORPORATION: PRODUCTS OFFERED
- TABLE 498 TERUMO CORPORATION: PRODUCT LAUNCHES
- TABLE 499 TERUMO CORPORATION: DEALS
- TABLE 500 TERUMO CORPORATION: EXPANSIONS
- TABLE 501 MERIT MEDICAL SYSTEMS, INC.: COMPANY OVERVIEW
- TABLE 502 MERIT MEDICAL SYSTEMS, INC.: PRODUCTS OFFERED
- TABLE 503 MERIT MEDICAL SYSTEMS, INC.: PRODUCT LAUNCHES
- TABLE 504 MERIT MEDICAL SYSTEMS, INC.: DEALS
- TABLE 505 B. BRAUN SE: COMPANY OVERVIEW
- TABLE 506 B. BRAUN SE: PRODUCTS OFFERED
- TABLE 507 B. BRAUN SE: PRODUCT APPROVALS
- TABLE 508 MEDTRONIC: COMPANY OVERVIEW
- TABLE 509 MEDTRONIC: PRODUCTS OFFERED
- TABLE 510 MEDTRONIC: PRODUCT LAUNCHES AND APPROVALS
- TABLE 511 MEDTRONIC: DEALS
- TABLE 512 PENUMBRA, INC.: COMPANY OVERVIEW
- TABLE 513 PENUMBRA, INC.: PRODUCTS OFFERED
- TABLE 514 PENUMBRA, INC.: PRODUCT LAUNCHES AND APPROVALS
- TABLE 515 STRYKER CORPORATION: COMPANY OVERVIEW
- TABLE 516 STRYKER CORPORATION: PRODUCTS OFFERED
- TABLE 517 STRYKER CORPORATION: DEALS
- TABLE 518 CORDIS: COMPANY OVERVIEW
- TABLE 519 CORDIS: PRODUCTS OFFERED
- TABLE 520 CORDIS: PRODUCT APPROVALS
- TABLE 521 CORDIS: DEALS
- TABLE 522 COOK: COMPANY OVERVIEW
- TABLE 523 COOK: PRODUCTS OFFERED
- TABLE 524 COOK: DEALS
- TABLE 525 W. L. GORE & ASSOCIATES, INC.: COMPANY OVERVIEW
- TABLE 526 W. L. GORE & ASSOCIATES, INC.: PRODUCTS OFFERED
- TABLE 527 W. L. GORE & ASSOCIATES, INC: PRODUCT APPROVALS
- TABLE 528 BIOSENSORS INTERNATIONAL GROUP, LTD.: COMPANY OVERVIEW
- TABLE 529 BIOSENSORS INTERNATIONAL GROUP, LTD.: PRODUCTS OFFERED
- TABLE 530 IVASCULAR: COMPANY OVERVIEW
- TABLE 531 IVASCULAR: PRODUCTS OFFERED
- TABLE 532 BIOTRONIK: COMPANY OVERVIEW
- TABLE 533 BIOTRONIK: PRODUCTS OFFERED
- TABLE 534 BIOTRONIK: PRODUCT LAUNCHES
- TABLE 535 ENDOLOGIX: COMPANY OVERVIEW
- TABLE 536 ENDOLOGIX: PRODUCTS OFFERED
- TABLE 537 ENDOCOR GMBH & CO., KG: COMPANY OVERVIEW
- TABLE 538 ENDOCOR GMBH & CO., KG: PRODUCTS OFFERED
- TABLE 539 MERIL LIFE SCIENCES PVT. LTD.: COMPANY OVERVIEW
- TABLE 540 ALVIMEDICA: COMPANY OVERVIEW
- TABLE 541 CARDIONOVUM GMBH: COMPANY OVERVIEW
- TABLE 542 SMT: COMPANY OVERVIEW
- TABLE 543 MEDINOL: COMPANY OVERVIEW
- TABLE 544 ANDRAMED GMBH: COMPANY OVERVIEW
- TABLE 545 REX MEDICAL: COMPANY OVERVIEW
- TABLE 546 QMD: COMPANY OVERVIEW
- TABLE 547 BROSMED MEDICAL CO., LTD.: COMPANY OVERVIEW
- TABLE 548 ELIXIR MEDICAL: COMPANY OVERVIEW
List of Figures
- FIGURE 1 MARKETS COVERED
- FIGURE 2 STUDY YEARS
- FIGURE 3 MARKET SCENARIO
- FIGURE 4 GLOBAL PERIPHERAL VASCULAR DEVICES MARKET, 2024-2031
- FIGURE 5 PERIPHERAL VASCULAR DEVICES MARKET: MAJOR STRATEGIES ADOPTED BY KEY PLAYERS, 2022-2025
- FIGURE 6 DISRUPTIONS INFLUENCING GROWTH OF PERIPHERAL VASCULAR DEVICES MARKET
- FIGURE 7 PERIPHERAL VASCULAR DEVICES MARKET: HIGH-GROWTH SEGMENTS, 2026-2031
- FIGURE 8 ASIA PACIFIC TO REGISTER HIGHEST CAGR IN PERIPHERAL VASCULAR DEVICES MARKET DURING FORECAST PERIOD
- FIGURE 9 RISING PREVALENCE OF PERIPHERAL ARTERY DISEASE (PAD) TO DRIVE MARKET
- FIGURE 10 ANGIOPLASTY STENT PRODUCTS AND CHINA LED RESPECTIVE MARKET SEGMENTS IN 2025
- FIGURE 11 INDIA TO REGISTER HIGHEST GROWTH DURING FORECAST PERIOD
- FIGURE 12 INDIA TO REGISTER HIGHER GROWTH RATES THAN EMERGING ECONOMIES DURING FORECAST PERIOD
- FIGURE 13 PERIPHERAL VASCULAR DEVICES MARKET: DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES
- FIGURE 14 PERIPHERAL VASCULAR DEVICES MARKET: PORTER'S FIVE FORCES ANALYSIS
- FIGURE 15 PERIPHERAL VASCULAR DEVICES MARKET: SUPPLY CHAIN
- FIGURE 16 PERIPHERAL VASCULAR DEVICES MARKET: VALUE CHAIN ANALYSIS (2025)
- FIGURE 17 PERIPHERAL VASCULAR DEVICES MARKET: ECOSYSTEM ANALYSIS
- FIGURE 18 IMPORT DATA FOR HS CODE 901839-COMPLIANT PRODUCTS, BY COUNTRY, 2021-2025 (USD MILLION)
- FIGURE 19 EXPORT DATA FOR HS CODE 901839-COMPLIANT PRODUCTS, BY COUNTRY, 2021-2025 (USD MILLION)
- FIGURE 20 TRENDS AND DISRUPTIONS IMPACTING CUSTOMER BUSINESS
- FIGURE 21 INVESTMENT/VENTURE CAPITAL SCENARIO IN PERIPHERAL VASCULAR DEVICES MARKET, 2021-2025
- FIGURE 22 PATENTS APPLIED AND GRANTED, 2015-2025
- FIGURE 23 TOP APPLICANT COUNTRIES/REGIONS FOR PERIPHERAL VASCULAR PATENTS, 2015-2025
- FIGURE 24 PERIPHERAL VASCULAR DEVICES MARKET: DECISION-MAKING FACTORS
- FIGURE 25 INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS FOR PERIPHERAL VASCULAR DEVICES
- FIGURE 26 KEY BUYING CRITERIA FOR TOP THREE END USERS
- FIGURE 27 ADOPTION BARRIERS AND INTERNAL CHALLENGES
- FIGURE 28 NORTH AMERICA: PERIPHERAL VASCULAR DEVICES MARKET SNAPSHOT
- FIGURE 29 ASIA PACIFIC: PERIPHERAL VASCULAR DEVICES MARKET SNAPSHOT
- FIGURE 30 REVENUE ANALYSIS FOR KEY PLAYERS IN PERIPHERAL VASCULAR DEVICES MARKET, 2023-2025 (USD MILLION)
- FIGURE 31 GLOBAL SHARE ANALYSIS OF KEY PLAYERS IN PERIPHERAL VASCULAR DEVICES MARKET, 2025
- FIGURE 32 US: PERIPHERAL VASCULAR DEVICES MARKET SHARE ANALYSIS, 2025
- FIGURE 33 PERIPHERAL VASCULAR DEVICES MARKET: COMPANY EVALUATION MATRIX (KEY PLAYERS), 2025
- FIGURE 34 PERIPHERAL VASCULAR DEVICES MARKET: COMPANY FOOTPRINT, 2025
- FIGURE 35 PERIPHERAL VASCULAR DEVICES MARKET: COMPANY EVALUATION MATRIX (STARTUPS/SMES), 2025
- FIGURE 36 PERIPHERAL VASCULAR DEVICES MARKET: BRAND/PRODUCT COMPARATIVE ANALYSIS
- FIGURE 37 EV/EBITDA OF KEY VENDORS, 2024
- FIGURE 38 YEAR-TO-DATE (YTD) PRICE TOTAL RETURN AND 5-YEAR STOCK BETA OF KEY VENDORS, 2024
- FIGURE 39 BOSTON SCIENTIFIC CORPORATION: COMPANY SNAPSHOT, 2025
- FIGURE 40 BECTON, DICKINSON AND COMPANY: COMPANY SNAPSHOT, 2025
- FIGURE 41 ABBOTT LABORATORIES: COMPANY SNAPSHOT, 2025
- FIGURE 42 CARDINAL HEALTH, INC.: COMPANY SNAPSHOT, 2025
- FIGURE 43 KONINKLIJKE PHILIPS N.V.: COMPANY SNAPSHOT, 2025
- FIGURE 44 TERUMO CORPORATION: COMPANY SNAPSHOT, 2025
- FIGURE 45 MERIT MEDICAL SYSTEMS, INC.: COMPANY SNAPSHOT, 2025
- FIGURE 46 B. BRAUN SE: COMPANY SNAPSHOT, 2025
- FIGURE 47 MEDTRONIC: COMPANY SNAPSHOT, 2025
- FIGURE 48 PENUMBRA, INC.: COMPANY SNAPSHOT, 2025
- FIGURE 49 STRYKER CORPORATION: COMPANY SNAPSHOT, 2025
- FIGURE 50 RESEARCH DESIGN
- FIGURE 51 PRIMARY SOURCES
- FIGURE 52 BREAKDOWN OF PRIMARY INTERVIEWS: SUPPLY-SIDE PARTICIPANTS
- FIGURE 53 BREAKDOWN OF PRIMARY INTERVIEWS: DEMAND-SIDE PARTICIPANTS
- FIGURE 54 BREAKDOWN OF PRIMARY INTERVIEWS: SUPPLY-SIDE AND DEMAND-SIDE PARTICIPANTS
- FIGURE 55 MARKET SIZE ESTIMATION: REVENUE SHARE ANALYSIS
- FIGURE 56 BOSTON SCIENTIFIC CORPORATION: REVENUE SHARE ANALYSIS (2025)
- FIGURE 57 PERIPHERAL VASCULAR DEVICES MARKET: SUPPLY-SIDE ANALYSIS
- FIGURE 58 TOP-DOWN APPROACH
- FIGURE 59 CAGR PROJECTIONS FROM ANALYSIS OF PERIPHERAL VASCULAR DEVICES MARKET DYNAMICS, 2026-2031
- FIGURE 60 CAGR PROJECTIONS: SUPPLY-SIDE ANALYSIS OF PERIPHERAL VASCULAR DEVICES MARKET, 2026-2031
- FIGURE 61 DATA TRIANGULATION




