PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1537704

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1537704
Pupillometer - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts (2024 - 2029)
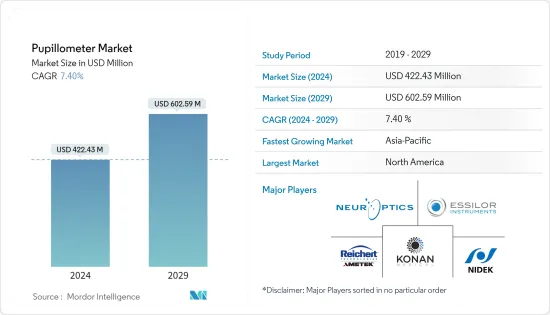
The Pupillometer Market size is estimated at USD 422.43 million in 2024, and is expected to reach USD 602.59 million by 2029, growing at a CAGR of 7.40% during the forecast period (2024-2029).
The major factor leading to the growth of the market studied includes the rise in awareness associated with pupillometers over manual pupillary examination, coupled with technological advancements. Other factors that contribute to the growth of the market include the surge in the prevalence of chronic disorders requiring the use of a pupillometer.
People suffering from depression, anxiety, Parkinson's disease, and autism spectrum disorder had abnormal pupillometry responses to stimuli. Pupillometry aids in understanding an individual's normal eye performance and brain activity disrupted by sickness. Hence, the growing burden of neurological diseases is expected to increase the screening rate and demand for pupilometers, thereby boosting market growth. For instance, according to the World Alzheimer's Report 2023, an estimated 6.7 million Americans aged 65 and older had Alzheimer's in 2023. Seventy-three percent were aged 75 years or older. This disease led to an abnormal effect on the eye. The demand for pupillometers is likely to increase during the forecast period.
Afferent pupillary defect (APD) is a measure of asymmetry that may occur with the optic nerve, brain tumors, retinal or cerebral vascular disease, and amblyopia. It occurs specifically when the disease or disorder presents asymmetrically. Hence, a surge in the prevalence of diseases such as brain tumors leads to a rise in demand for pupillometers, which are used to detect APD in patients suffering from brain tumors. For instance, according to an estimate by the American Society of Clinical Oncology (ASCO), 25,050 adults were estimated to be diagnosed with primary cancerous tumors of the brain and spinal cord in the United States in 2022.
Over the years, manual pupil assessment has remained a major way of obtaining pupillary information about a patient. The various initiatives taken by private and public organizations to increase the adoption of pupillometer in developing and underdeveloped countries are expected to boost market growth during the forecast period. For instance, in February 2023, the SBI Foundation donated INR 1.41 crore (USD 0.16 million) to a charitable medical institute to upgrade its facilities, including rare equipment such as India's first Danish pupillometer. The timely diagnosis, advanced medical equipment, and personalized follow-up care available to people with communication, speech, and hearing impairments can significantly improve their quality of life and reduce the impact of their disabilities. The institute unveiled several highly unusual pieces of equipment, including the pupilometer and stroboscope, which are not typically found in India.
Therefore, owing to the aforementioned factors, the market studied is anticipated to witness growth during the forecast period. However, the high cost of pupillometers and the lack of specialized clinicians impact their penetration in emerging countries. This, in turn, affects the growth of the pupillometer market.
Pupillometer Market Trends
Video Pupillometer is Expected to Witness Significant Growth During the Forecast Period
- Video pupillometers are more advanced compared to digital pupillometers. A video pupillometer is favored as it captures the entire pupil dilation procedure with more clarity. The video pupillometer aids in the capture of pupil characteristics and response across time, which is predicted to improve diagnosis. The segment is driven by the rising demand for video pupillometry and technological advancements in pupillometry. The pupillometer helps in the diagnosis of various neurological diseases, such as dementia and brain tumors.
- The availability of several players offering video pupillometers on the market gives other players an enormous opportunity to research and develop technologically advanced video pupillometers. For example, the EyeKinetix, NeuroOptics, IDMED, and others. Further, the technological advances and benefits associated with video pupillometers are expected to increase the use of video pupillometers, which is likely to boost market growth. For instance, according to the study published in Clinical Ophthalmology in February 2023, high-quality infrared video pupillography was successfully captured for light and near pupillary reflexes and video documentation of pupillary examination at the bedside and in the clinic with accurate and reproducible measurements of the pupillary size in light and dark environments. Hence, the effectiveness of video pupillometry is expected to increase the adoption, thereby contributing to market growth.
- Hence, the presence of market players and the adoption of video pupillometry are expected to propel the growth of the segment during the forecast period.
North America is Expected to Hold a Significant Market Share in the Pupillometer Market During the Forecast Period
- North America is expected to dominate the market due to the presence of a large number of key players offering pupillometers in the region. A rising number of chronic diseases, such as neurological and ophthalmic diseases, is expected to boost the market in the region.
- According to a study published in the Journal of the American College of Emergency Physicians in June 2023, NeurOptics' automated pupillometry and the neurological pupil index (NPi) were unaffected by clinical intoxication. Thus, they can be used for rapidly screening emergency room patients for brain injury or neurological injuries regardless of intoxication.
- Accurate neurological and pupillary assessments are critical for the detection of neurological deterioration in patients with traumatic brain injury (TBI). Hence, the surge in cases of traumatic brain injury in the region is another factor that drives the growth of the market. For instance, according to a StatPearls article published in January 2022, traumatic brain injury affects around 1.7 million people in the United States each year, with adolescents aged 15 to 19 years and adults aged 65 years and older being the most likely to develop TBI. In addition, as per the same source, there are 500 traumatic brain injuries per 100,000 people in the United States each year. Furthermore, pupillometers are employed to check pupil response in patients with retinal diseases such as macular degeneration, diabetic retinopathy, and intraocular tumors. Retinal diseases such as macular degeneration are more prevalent in the geriatric population. For instance, according to the CDC Report in November 2022, glaucoma cases are expected to rise to 6.3 million by 2050 in the United States. Further, the Statistics Canada report published in March 2023 estimated that glaucoma impacted more than 728,000 Canadians in 2022.
- Strategic activities by market players, such as partnerships, mergers, and acquisitions are expected to boost the market's growth during the forecast period. For instance, in January 2023, Essilor Instruments, a division of EssilorLuxottica and Espansione Group, announced a long-term exclusive partnership in North America for the distribution of ophthalmic instrumentation and expansion into the region.
- Therefore, owing to the aforesaid factors, the market studied is anticipated to grow in North America.
Pupillometer Industry Overview
The pupillometer market is competitive due to the presence of several companies operating globally as well as regionally. The market players are adopting various strategies such as product launches, mergers and acquisitions, and collaboration to stay competitive in the pupillometer market. The key companies operating in the market are NeurOptics Inc., Essilor Group (Essilor Instruments), Visionix, US Ophthalmic, NIDEK SA, HAAG-STREIT GROUP, Reichert Inc., Konan Medical USA Inc., Good-Lite Company, and Grobet USA (Vigor Optical).
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope of the Study
2 RESEARCH METHODOLOGY
3 EXECUTIVE SUMMARY
4 MARKET DYNAMICS
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Rise in Awareness Associated with Pupillometers Coupled with Technological Advancements
- 4.2.2 Surge in Prevalence of Disorders Requiring the Use of Pupillometer
- 4.3 Market Restraints
- 4.3.1 High Cost of Pupillometers and Lack of Specialized Clinicians
- 4.4 Porter's Five Forces Analysis
- 4.4.1 Threat of New Entrants
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Bargaining Power of Suppliers
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5 MARKET SEGMENTATION (Market Size by Value - USD)
- 5.1 By Modality
- 5.1.1 Handheld Pupillometer
- 5.1.2 Table Top Pupillometer
- 5.2 By Type
- 5.2.1 Digital Pupillometer
- 5.2.2 Video Pupillometer
- 5.3 By Application
- 5.3.1 Ophthalmology
- 5.3.2 Neurology
- 5.3.3 Other Applications
- 5.4 By End User
- 5.4.1 Hospitals
- 5.4.2 Eye Clinics
- 5.4.3 Other End Users
- 5.5 By Geography
- 5.5.1 North America
- 5.5.1.1 United States
- 5.5.1.2 Canada
- 5.5.1.3 Mexico
- 5.5.2 Europe
- 5.5.2.1 Germany
- 5.5.2.2 United Kingdom
- 5.5.2.3 France
- 5.5.2.4 Italy
- 5.5.2.5 Spain
- 5.5.2.6 Rest of Europe
- 5.5.3 Asia-Pacific
- 5.5.3.1 China
- 5.5.3.2 Japan
- 5.5.3.3 India
- 5.5.3.4 Australia
- 5.5.3.5 South Korea
- 5.5.3.6 Rest of Asia-Pacific
- 5.5.4 Rest of the World
- 5.5.1 North America
6 COMPETITIVE LANDSCAPE
- 6.1 Company Profiles
- 6.1.1 NeurOptics Inc.
- 6.1.2 Essilor Instruments USA
- 6.1.3 Visionix
- 6.1.4 Advancing Eyecare (US Ophthalmic)
- 6.1.5 NIDEK SA
- 6.1.6 HAAG-STREIT GROUP
- 6.1.7 Reichert Inc.
- 6.1.8 Konan Medical USA Inc.
- 6.1.9 Good-Lite Company
- 6.1.10 Grobet USA (Vigor Optical)
7 MARKET OPPORTUNITIES AND FUTURE TRENDS




