PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1443911

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1443911
Global Biosimilars - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts (2024 - 2029)
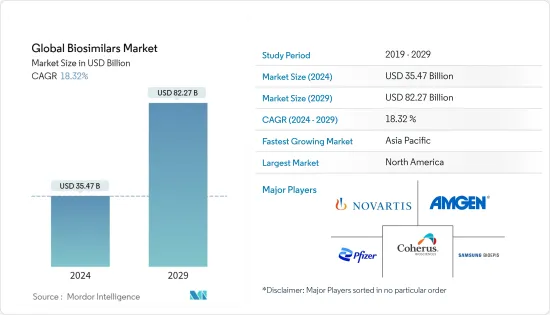
The Global Biosimilars Market size is estimated at USD 35.47 billion in 2024, and is expected to reach USD 82.27 billion by 2029, growing at a CAGR of 18.32% during the forecast period (2024-2029).
The impact of COVID-19 on the biosimilars market may be significant, and it has imposed a great challenge on the pharmaceutical companies focused on biosimilar development. The reduction in the approval by regulatory bodies of non-COVID therapeutics during the current pandemic is expected to delay the process of product approvals and launches, hindering the market's growth. Additionally, as per the research article titled "Impact of Covid-19 on Cancer Care: A Global Collaborative Study" published in September 2020, nearly 88% of the cancer care centers faced challenges in delivering usual cancer care for many reasons, including preventive measures, lack of personal protective equipment, and staff shortages due to COVID-19. As biosimilars are injections that need experts to be administered and also as the supply chain was greatly impaired in the early pandemic phase, it has significantly impacted the market growth. Therefore, COVID-19 impacted the growth of the biosimilars market in its initial phase, however, the resumption of supply chain activities and clinical trials for drug development globally will boost the market growth rate over the coming years.
Moreover, several blockbuster biologic drugs of major pharmaceutical companies, such as Truvada, Chantix, Forteo, Ciprodex, Afinitor, and many other drugs, lost United States exclusivity in 2020. In the coming decade, there will be a rise in the patent expiration of several existing biological drugs, such as Erbitux, Avastin, and Orencia, which would provide an opportunity for many innovator companies as well as generic manufacturers to offer services specially tailored toward biosimilars. Besides, factors such as the cost-effective nature of biosimilars, rising acceptance and adoption by various stakeholders, the need for diversification in technology and business models, and the growing prevalence of chronic diseases are expected to drive the global biosimilar market.
Furthermore, the growing burden of cancer and increasing deaths due to it create the need for affordable treatment and thus boost the growth of the biosimilar market. For instance, according to GLOBOCAN 2020, in the United States, there were 2,281,658 estimated new cases of cancer and nearly 612,390 deaths in 2020. Additionally, the key market players anticipated market growth over the forecast period through various strategic activities, such as product launches, mergers, and acquisitions. For instance, in July 2021, the Food and Drug Administration (FDA) approved Viatris Inc.'s (formerly Mylan Pharmaceuticals Inc.) SEMGLEE (insulin-glargine-yfgn), a biosimilar to LANTUS (insulin glargine).
On the other hand, factors such as lack of definitive standards for approval and adequate profitability, given the greater risk with concerns regarding substitutability and interchangeability, discourage many companies from investing in this market, thus impeding the growth of the market studied.
Biosimilars Market Trends
The Oncology Segment Holds the Major Share in the Global Biosimilars Market
The oncology segment is expected to hold the major share in the biosimilars market, and the high incidence of cancers worldwide is the major factor driving the growth of the studied segment over the forecast period. According to the International Agency for Research on Cancer 2020, the estimated number of new leukemia cases in 2020 was 474,519 globally. According to the same source, the disease also had significantly high mortality, with a total global death toll of 311,594. Additionally, according to estimates from the International Agency for Research on Cancer (IARC), by 2040, the global burden of cancer is expected to grow to 27.5 million new cancer cases and 16.3 million deaths worldwide. The increasing incidence of cancer cases is expected to drive the need for advanced cancer drugs for the effective treatment of patients.
Additionally, the increasing research and development activities by key players focused on oncology, along with rising approvals by different regulatory bodies, are expected to drive the studied market's growth. For instance, in December 2020, Amgen received United States Food and Drug Administration (USFDA) approval for its RIABNI (rituximab-arrx), a biosimilar to Rituxan (rituximab), for the treatment of adult patients with non-Hodgkin's lymphoma, chronic lymphocytic leukemia, granulomatosis with polyangiitis, and microscopic polyangiitis. Moreover, in May 2022, Biocon Biologics Ltd. and Viatris Inc. received approval from Health Canada across four oncology indications for Abevmy (Bevacizumab), a biosimilar to Roche's Avastin (Bevacizumab).
Thus, because of the factors above, the oncology segment is anticipated to witness significant growth over the forecast period.
North America Holds the Major Share and is Expected to Dominate the Biosimilars Market Over the Forecast Period
North America is anticipated to observe a significant CAGR over the forecast period in the biosimilars market. The major factors driving the growth of the studied market in the region include the high incidence of chronic diseases, such as cancers, along with the increased investment in research and development activities by the major players. According to GLOBOCAN 2020, the number of new cancer cases diagnosed was 2,281,658 in the United States in 2020, with 612,390 deaths. Among all cancers, breast cancer had the highest incidence with 253,465 cases, followed by lung (227,875), prostate (209,512), and colon (101,809).
Moreover, North America is the hub for many key players in the market, such as Pfizer Inc., Mylan NV, Amgen Inc., and Coherus Biosciences Inc., among others. The increasing pipeline of products and new product launches increase the market growth in the region. For instance, in June 2020, Pfizer Inc. received United States approval for its pegfilgrastim biosimilar, Nyvepria, indicated for lowering infection incidence. Additionally, in February 2021, Coherus BioSciences, Inc. reported that the United States Food and Drug Administration ("FDA") has accepted for review the 351(k) Biologics License Application ("BLA") for CHS-1420, a Humira (adalimumab) biosimilar product candidate.
Thus, the market is expected to propel significantly over the forecast period in North America due to the high incidence of chronic diseases, such as cancer, and increasing R&D activities.
Biosimilars Industry Overview
The biosimilars market is highly competitive, with many key players dominating the market. The major players in the studied market comprise Novartis AG, Pfizer Inc., Amgen Inc., Coherus Biosciences Inc., Mylan NV (Viatris Inc.), and Samsung Bioepis Co., Ltd., among others. Most market players are adopting various growth strategies, such as acquisitions, partnerships, and new product launches, to secure their position in the global market.
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope of the Study
2 RESEARCH METHODOLOGY
3 EXECUTIVE SUMMARY
4 MARKET DYNAMICS
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Several Blockbuster Biopharmaceuticals Going Off-patent over the Next Five to Ten Years
- 4.2.2 Growing Burden of Chronic Diseases and Increasing R&D Investments
- 4.2.3 Increasing Demand for Biosimilar Drugs Due to Their Cost Effectiveness
- 4.3 Market Restraints
- 4.3.1 Concerns Regarding Substitutability and Interchangeability
- 4.3.2 Regulatory Uncertainty and Growing Competition from Biobetters
- 4.3.3 High Cost Involvement and Complexities in Manufacturing
- 4.4 Porter's Five Forces Analysis
- 4.4.1 Threat of New Entrants
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Bargaining Power of Suppliers
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5 MARKET SEGMENTATION (Market Size by Value - USD million)
- 5.1 By Product Class
- 5.1.1 Monoclonal Antibodies
- 5.1.2 Recombinant Hormones
- 5.1.3 Immunomodulators
- 5.1.4 Anti-Inflammatory Agents
- 5.1.5 Other Product Classes
- 5.2 By Application
- 5.2.1 Blood Disorders
- 5.2.2 Growth Hormonal Deficiency
- 5.2.3 Chronic and Autoimmune Disorders
- 5.2.4 Oncology
- 5.2.5 Other Applications
- 5.3 Geography
- 5.3.1 North America
- 5.3.1.1 United States
- 5.3.1.2 Canada
- 5.3.1.3 Mexico
- 5.3.2 Europe
- 5.3.2.1 Germany
- 5.3.2.2 United Kingdom
- 5.3.2.3 France
- 5.3.2.4 Italy
- 5.3.2.5 Spain
- 5.3.2.6 Rest of Europe
- 5.3.3 Asia-Pacific
- 5.3.3.1 China
- 5.3.3.2 Japan
- 5.3.3.3 India
- 5.3.3.4 Australia
- 5.3.3.5 South Korea
- 5.3.3.6 Rest of Asia-Pacific
- 5.3.4 Middle East and Africa
- 5.3.4.1 GCC
- 5.3.4.2 South Africa
- 5.3.4.3 Rest of Middle East and Africa
- 5.3.5 South America
- 5.3.5.1 Brazil
- 5.3.5.2 Argentina
- 5.3.5.3 Rest of South America
- 5.3.1 North America
6 COMPETITIVE LANDSCAPE
- 6.1 Company Profiles
- 6.1.1 Pfizer Inc.
- 6.1.2 Eli Lilly and Company
- 6.1.3 Celltrion Healthcare
- 6.1.4 Viatris Inc. (Mylan)
- 6.1.5 Novartis AG
- 6.1.6 Samsung Bioepis Co. Ltd
- 6.1.7 Stada Arzneimittel AG
- 6.1.8 Teva Pharmaceutical Industries Ltd
- 6.1.9 Intas Pharmaceutical Ltd
- 6.1.10 LG Life Sciences (LG Chem)
- 6.1.11 Biocon Limited
- 6.1.12 Amgen Inc.
- 6.1.13 Dr. Reddy's Laboratories
- 6.1.14 Coherus Biosciences Inc.
- 6.1.15 Biocad
7 MARKET OPPORTUNITIES AND FUTURE TRENDS




