PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1437875

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1437875
Defibrillator - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts (2024 - 2029)
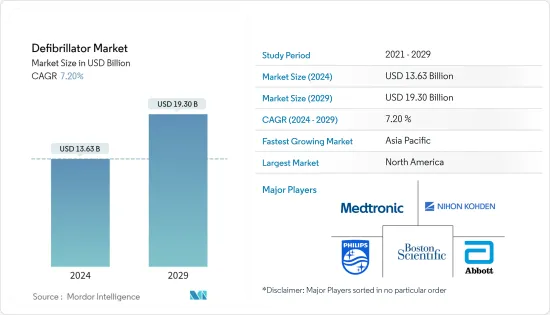
The Defibrillator Market size is estimated at USD 13.63 billion in 2024, and is expected to reach USD 19.30 billion by 2029, growing at a CAGR of 7.20% during the forecast period (2024-2029).
The COVID-19 epidemic has altered several facets of the healthcare system, including how people are treated for cardiac arrests both outside and within hospitals. There was a reduction in cardiac procedures during the pandemic period due to the strict regulation by various governments across the world. For instance, the EHRA article published in March 2021 mentioned that during the pandemic period, cardiac resynchronization therapy procedures with defibrillation devices decreased by 45%, and procedures involving implantable cardioverter defibrillators were 45% in England. Such reductions in the cardiovascular procedures involving defibrillator devices during the pandemic period had a notable impact on the growth of the market. However, the resumption of all cardiac surgical procedures after the relaxation of strict regulations during the post-pandemic period is expected to contribute to the growth of the market over the forecast period.
The increasing prevalence of cardiovascular diseases and technological advancements in the field of defibrillators are the major drivers for the market. For instance, the British Heart Foundation (BHF) data published in January 2022 reported that the most common heart conditions affected globally were coronary (ischemic) heart disease (global prevalence estimated at 200 million), peripheral arterial (vascular) disease (110 million), stroke (100 million) and atrial fibrillation (60 million). The report also mentioned that the prevalence of heart and circulatory diseases in North America was 46 million, in Europe 99 million, in Africa 58 million, in South America 32 million, in Asia and Australia 310 million. Therefore, the high burden of cardiovascular diseases is expected to contribute to the growing demand for defibrillators as they are widely used in reviving patients from sudden cardiac arrest by providing sufficient oxygen and blood to the required organs via a sudden shock, thereby fueling the growth of the market.
Additionally, the increasing product launches by various governments and various market players are also expected to contribute to the growth of the market. For instance, in February 2022, Infrastructure Minister Nichola Mallon unveiled one of the first life-saving defibrillators that will be installed across the public transport network in Northern Ireland. Additionally, in July 2021, Jeevtronics Pvt Ltd developed SanMitra 100 HCT, the hand-cranked dual-powered defibrillator. The device is affordable, low-weight, and considered more reliable than traditional defibrillators by experts as it can be used even in areas where electricity is unavailable.
However, the stringent regulatory framework is likely to hinder market growth over the forecast period.
Defibrillator Market Trends
Fully Automated External Defibrillator is Expected to Witness Growth Over the Forecast Period
A fully automated external defibrillator (AED) is designed to automatically deliver a shock when needed without the rescuer having to press a button to deliver the shock. The device communicates step-by-step instructions to let rescuers know when the victim is about to receive a shock. Fully automatic models are designed to assist responders in cardiac arrest emergencies. Many studies have shown that fully automatic AEDs are safe, and effective, and can reduce extended delays associated with hesitating to press the shock button during a rescue.
The launch of various programs, awareness campaigns, product launches, and AED management platforms to promote the importance of fully automated external defibrillators are the major factors driving the segment's growth. For instance, in August 2022, Portage Health Foundation (PHF) announced a one-time automated external defibrillator (AED) grant program to address the foundation's long-term sustainability goal of building safer communities. Additionally, in March 2021, the government of Australia launched an automated external defibrillator at the Government house of Australia. The AED was a joint venture between St. John Ambulance Australia and Zoll Medical Australia.
Also, in August 2022, the Great Western Air Ambulance Charity team launched an automated external defibrillator (AED) usage campaign during CPR in the United States. Likewise, in June 2021, WhaleTeq launched the first automated external defibrillator management platform with their latest defibrillator test, DSF2000 to fill in the AED testing and management gap. Thus, the rising awareness programs for AED and increasing product launches by various government organizations are expected to contribute to the growth of the market.
North America is Expected to Hold Significant Share in the Defibrillator Market Over the Forecast Period
North America is expected to hold a significant share of the market over the forecast period owing to the high prevalence of cardiovascular diseases, increasing adoption of technologically advanced defibrillator products, and the presence of major market players. For instance, the ACC article published in May 2022 mentioned that about 377,000 adults and 23,000 children experience out-of-hospital cardiac arrest each year in the United States.
Also, as per the February 2022 report from the Heart and Stroke Foundation of Canada, 750,000 people are living with heart failure, and 100,000 people are diagnosed with this incurable condition each year in the country and it is projected that by the year 2030, the healthcare costs associated with heart failure in Canada will reach up to USD 2.8 billion per year. Hence, with this high number of heart patients along with the increasing economic burden due to heart failure in the country, the market is expected to witness high growth over the forecast period in this region.
The presence of a key market in this region and the development of advanced products to meet the growing demand for defibrillators is expected to contribute to the growth of the market. For instance, in December 2021, Boston Scientific launched the MODULAR ATP clinical trial to evaluate the safety, performance, and effectiveness of the mCRM Modular Therapy System. The mCRM system consists of two cardiac rhythm management devices, the EMBLEM MRI subcutaneous implantable defibrillator (S-ICD) system and the other.
Therefore, owing to the aforesaid factors, such as the high burden of cardiovascular diseases, innovative product launches, are expected to contribute to the growth of the studied market in the North America Region.
Defibrillator Industry Overview
The defibrillator market is fragmented in nature due to the presence of several companies operating globally as well as regionally. The competitive landscape includes an analysis of a few international as well as local companies which hold the market shares and are well known including Boston Scientific Corporation, Koninklijke Philips NV, Medtronic PLC, Nihon Kohden Corporation, and Abbott Laboratories among others.
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 Study Assumptions and Market Defination
- 1.2 Scope of the Study
2 RESEARCH METHODOLOGY
3 EXECUTIVE SUMMARY
4 MARKET DYNAMICS
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Increasing Prevalence of Cardiovascular Diseases
- 4.2.2 Technological Advancements in the Field of Defibrillators
- 4.3 Market Restraints
- 4.3.1 Stringent Regulatory Framework
- 4.4 Porter's Five Force Analysis
- 4.4.1 Threat of New Entrants
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Bargaining Power of Suppliers
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5 MARKET SEGMENTATION (Market Size by Value - USD Million)
- 5.1 By Product
- 5.1.1 Implantable Cardioverter Defibrillator (ICD)
- 5.1.1.1 Transvenous Implantable Cardioverter Defibrillator (T-ICDs)
- 5.1.1.2 Subcutaneous Implantable Cardioverter Defibrillator (S-ICDs)
- 5.1.1.3 CRT with Pacemaker and ICD Function (CRT-D)
- 5.1.2 External Defibrillator (ED)
- 5.1.2.1 Automated External Defibrillator (AEDs)
- 5.1.2.1.1 Semi-automated External Defibrillators
- 5.1.2.1.2 Fully-automated External Defibrillators
- 5.1.3 Manual External Defibrillator
- 5.1.3.1 Wearable Cardioverter Defibrillators (WCDs)
- 5.1.1 Implantable Cardioverter Defibrillator (ICD)
- 5.2 By End User
- 5.2.1 Hospitals
- 5.2.2 Emergency Care
- 5.2.3 Home Care
- 5.2.4 Other End Users
- 5.3 Geography
- 5.3.1 North America
- 5.3.1.1 United States
- 5.3.1.2 Canada
- 5.3.1.3 Mexico
- 5.3.2 Europe
- 5.3.2.1 Germany
- 5.3.2.2 United Kingdom
- 5.3.2.3 France
- 5.3.2.4 Italy
- 5.3.2.5 Spain
- 5.3.2.6 Rest of Europe
- 5.3.3 Asia-Pacific
- 5.3.3.1 China
- 5.3.3.2 Japan
- 5.3.3.3 India
- 5.3.3.4 Australia
- 5.3.3.5 South Korea
- 5.3.3.6 Rest of Asia-Pacific
- 5.3.4 Middle East and Africa
- 5.3.4.1 GCC
- 5.3.4.2 South Africa
- 5.3.4.3 Rest of Middle East and Africa
- 5.3.5 South America
- 5.3.5.1 Brazil
- 5.3.5.2 Argentina
- 5.3.5.3 Rest of South America
- 5.3.1 North America
6 COMPETITIVE LANDSCAPE
- 6.1 Company Profiles
- 6.1.1 Abbott Laboratories
- 6.1.2 Boston Scientific Corporation
- 6.1.3 Cardiac Science Corporation
- 6.1.4 Defibtech LLC
- 6.1.5 Koninklijke Philips NV
- 6.1.6 LivaNova PLC
- 6.1.7 Medtronic PLC
- 6.1.8 Nihon Kohden Corporation
- 6.1.9 Physio-Control Inc.
- 6.1.10 ZOLL Medical Corporation
- 6.1.11 Bexen Cardio
- 6.1.12 Mindray Medical International Ltd.
7 MARKET OPPORTUNITIES AND FUTURE TRENDS




