PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1407061

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1407061
Pharmaceutical Granulation Equipment - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts 2024 - 2029
The pharmaceutical granulation equipment market is anticipated to register a CAGR of 5.5% over the forecast period.
During the COVID-19 pandemic, many countries faced challenges in manufacturing pharmaceuticals, impacting the study market. The difficulties faced by pharmaceutical makers were primarily due to the inability of most products' production and supply to be interrupted or reduced, regardless of organizational or outbreak-related factors. Hence, the challenges faced by the companies reduced the demand for pharmaceutical granulation equipment, adversely impacting the market during the pandemic. However, the pharmaceutical granulation equipment market may grow further over the coming years due to resumed supply chain activities.
The increasing research and development spending on healthcare, specifically pharmaceuticals, is expected to boost market growth. For instance, according to the data published by the Congressional Budget Office 2021, the pharmaceutical industries spent nearly USD 200 billion on research and development globally in 2021, compared to USD 83.0 million in previous years. Hence, the increasing healthcare R&D expenditure raises the need for various pharmaceutical products that need granulation procedures using various equipment and is expected to propel the market over the study period.
The increasing number of clinical trials by some pharmaceutical and biotechnology companies contributes to market growth. For instance, in May 2021, Evotec reported a phase 1 clinical trial on a new anticancer molecule. The candidate was created in partnership with Exscientia, a company based in Oxford, United Kingdom, that applies artificial intelligence (AI) techniques to small-molecule drug discovery. Thus, the market is expected to grow significantly over the forecast period due to the abovementioned factors.
However, the high equipment cost will likely hinder the market growth over the forecast period.
Pharmaceutical Granulation Equipment Market Trends
Pharmaceutical Manufacturing Companies Segment is Expected to Hold a Significant Market Share Over the Forecast Period
Advancements in science and technology are augmenting the growth of R&D activities by pharmaceutical companies globally. With the rise in R&D spending and increasing sales of pharmaceuticals in developed countries like the United States and Canada, the pharmaceutical industry is entering a new era of advanced medical development. For instance, in April 2022, Ferring Pharmaceuticals opened its integrated R&D and manufacturing facility in Hyderabad and established it with a total investment of EUR 30 million (USD 32.9 million). The R&D capability includes formulation development, and the manufacturing facility is mainly designed for the oral solid dosage form, where the granulation of active ingredients is a crucial step. Hence, with new manufacturing facilities, the market is expected to grow significantly over the study period.
Furthermore, the initiation of the manufacturing of powders is expected to significantly impact the growth of the market. For instance, in November 2022, Hosokawa Micron Powder Systems took the initial steps to begin more in-house manufacturing. This initiative will result in better quality control and lower costs over time. Hence, the new manufacturing of pharmaceuticals helps to increase the demand for granulation and is expected to grow significantly in the market over the forecast period.
Thus, establishing new manufacturing facilities with new investments and initiation of manufacturing services is expected to increase the need for granulation in pharmaceutical manufacturing, which will likely propel the market to grow over the forecast period.
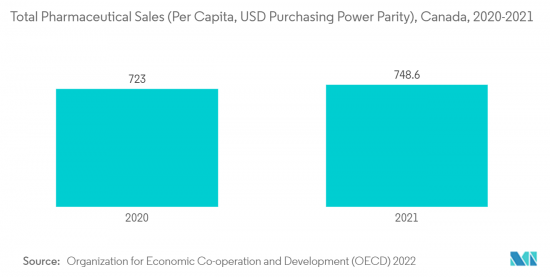
North America is Expected to Hold Significant Share in the Market Over the Forecast Period
North America is expected to hold a significant market share due to increased investments and initiatives by public and private organizations. The United States pharmaceutical industry is one of the leaders in developing new medicines. Pharmaceutical Research and Manufacturers of America (PhRMA) member companies remained at the forefront. The increasing funding from the United States government for supporting healthcare is an attributing factor to the growth of the market in the United States. For instance, the Congressional Budget Office 2021 published that federal funding for the National Institute of Health (NIH) in the past few decades totaled over USD 700 billion. This significant funding in the pharmaceutical field is expected to drive the market over the study period.
Furthermore, in July 2021, the United States Pharmacopeia (USP), an independent, scientific nonprofit organization, acquired Pharmatech Associates, Inc. With the addition of Pharmatech Associates' expertise, USP further advanced its 200-year mission. It is to ensure access to quality medicines by extending services that assist manufacturers in meeting global quality standards across the drug and product lifecycle.
Rising healthcare expenditure is fueling the market growth. For instance, as per the data published by the OECD in 2021, Mexico's healthcare expenditure represented 6.2% of its GDP. Therefore, pharmaceutical companies are shifting to discover novel therapies required during development. This approach is expected to affect pharmaceutical production positively in Mexico, thus driving market growth.
Pharmaceutical Granulation Equipment Industry Overview
The market is partially fragmented and consists of several major players. Some of the major players in the market are Alexanderwerk, Anchor Mark Pvt Ltd, Syntegon Technology GmbH, GEA Group, and HOSOKAWA MICRON B.V., among others.
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope of the Study
2 RESEARCH METHODOLOGY
3 EXECUTIVE SUMMARY
4 MARKET DYNAMICS
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Increasing Number of Research Laboratories
- 4.2.2 Growing Healthcare R&D Expenditure
- 4.3 Market Restraints
- 4.3.1 High Cost of Equipment
- 4.4 Porter's Five Forces Analysis
- 4.4.1 Bargaining Power of Suppliers
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Threat of New Entrants
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5 MARKET SEGMENTATION (Market Size by Value - USD)
- 5.1 By Type
- 5.1.1 Single Pot Granulators
- 5.1.2 High Sheer Granulators
- 5.1.3 Oscillating Granulators
- 5.1.4 Others
- 5.2 By Mechanism
- 5.2.1 Dry Granulators
- 5.2.2 Wet Granulators
- 5.2.3 Mixture Granulators
- 5.3 By End User
- 5.3.1 Pharmaceutical Manufacturing Companies
- 5.3.2 Contract Manufacturing
- 5.3.3 Others
- 5.4 Geography
- 5.4.1 North America
- 5.4.1.1 United States
- 5.4.1.2 Canada
- 5.4.1.3 Mexico
- 5.4.2 Europe
- 5.4.2.1 Germany
- 5.4.2.2 United Kingdom
- 5.4.2.3 France
- 5.4.2.4 Italy
- 5.4.2.5 Spain
- 5.4.2.6 Rest of Europe
- 5.4.3 Asia-Pacific
- 5.4.3.1 China
- 5.4.3.2 Japan
- 5.4.3.3 India
- 5.4.3.4 Australia
- 5.4.3.5 South Korea
- 5.4.3.6 Rest of Asia-Pacific
- 5.4.4 Middle East and Africa
- 5.4.4.1 GCC
- 5.4.4.2 South Africa
- 5.4.4.3 Rest of Middle East and Africa
- 5.4.5 South America
- 5.4.5.1 Brazil
- 5.4.5.2 Argentina
- 5.4.5.3 Rest of South America
- 5.4.1 North America
6 COMPETITIVE LANDSCAPE
- 6.1 Company Profiles
- 6.1.1 Alexanderwerk
- 6.1.2 Anchor Mark Pvt Ltd
- 6.1.3 Fluid Air (Spraying Systems Co.)
- 6.1.4 HOSOKAWA MICRON B.V.
- 6.1.5 GEA Group
- 6.1.6 I.M.A. INDUSTRIA MACCHINE AUTOMATICHE S.P.A.
- 6.1.7 L.B. Bohle Maschinen und Verfahren GmbH
- 6.1.8 LFA Machines Oxford LTD
- 6.1.9 PTK-GB Limited
- 6.1.10 Romaco Group
- 6.1.11 Syntegon Technology GmbH
7 MARKET OPPORTUNITIES AND FUTURE TRENDS




