PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1851178

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1851178
Prosthetic Heart Valve - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts (2025 - 2030)
The prosthetic heart valve market reached USD 13.35 billion in 2025 and is forecast to grow to USD 23.32 billion by 2030, registering an 11.8% CAGR over the period.
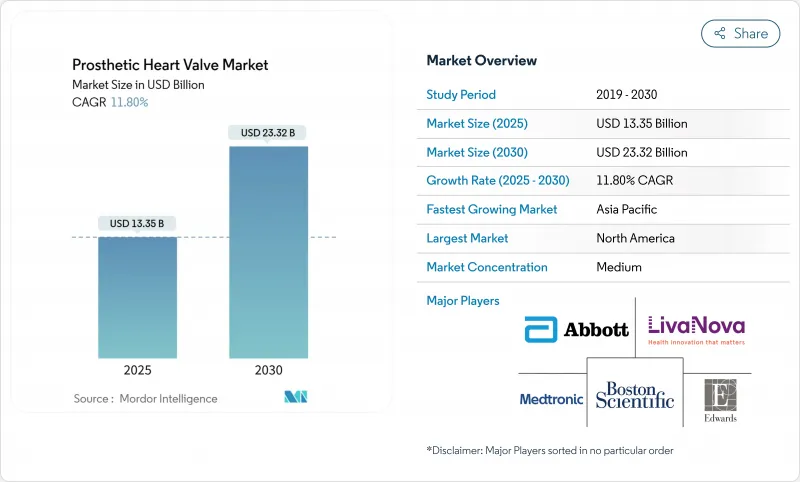
Demographic ageing, expanded indications for transcatheter aortic valve replacement (TAVR), and faster regulatory reviews position transcatheter innovation as the primary growth engine of the prosthetic heart valve market. Edwards Lifesciences obtained United States Food and Drug Administration (FDA) approval in May 2025 for the SAPIEN 3 platform in asymptomatic severe aortic stenosis, enlarging the treatable population beyond symptomatic patients. Transcatheter heart valves held 45.55% of revenue in 2024, while tricuspid systems such as Edwards' EVOQUE and Abbott's TriClip have accelerated into double-digit growth after first-in-class clearances. Hospitals continue to dominate procedure volumes, yet ambulatory surgical centers (ASCs) are expanding fastest as same-day discharge protocols reduce inpatient reliance. North America generates the largest share, but Asia-Pacific is the high-growth frontier thanks to local approvals like MicroPort's VitaFlow Liberty Flex in China. Portfolio consolidation-exemplified by Edwards' USD 300 million Innovalve purchase and Johnson & Johnson's USD 1.7 billion V-Wave deal-further intensifies competition.
Global Prosthetic Heart Valve Market Trends and Insights
Expanding Indications for TAVR/TAVI
The May 2025 FDA approval of Edwards' SAPIEN 3 platform for asymptomatic severe aortic stenosis removes the "watchful waiting" mindset, allowing clinicians to intervene before symptom onset. EARLY TAVR data showed 26.8% adverse events with early treatment versus 45.3% under surveillance during 3.8-year follow-up, validating proactive therapy and effectively doubling the addressable pool. Edwards forecasts TAVR sales of USD 4.1-4.4 billion in 2025, and rivals such as Abbott have launched the ENVISION trial to capture lower-risk patients. Centers for Medicare & Medicaid Services (CMS) coverage decisions will shape adoption because private payers typically mirror Medicare precedent. As coverage broadens, procedure volumes rise, reinforcing the prosthetic heart valve market's shift toward transcatheter dominance.
Regulatory Fast-Tracks & Breakthrough Designations
Breakthrough device designations hit a record 1,041 by September 2024; 128 authorized products demonstrate how the pathway compresses approvals from 3-5 years to around 18-24 months. Edwards' EVOQUE tricuspid valve capitalized on breakthrough status to secure February 2024 clearance, while 4C Medical's polymer AltaValve holds two breakthrough labels. Europe parallels this urgency, granting the world's first transfemoral mitral CE mark to SAPIEN M3 in April 2025. Companies that lock in early designations gain speed-to-market advantages, boosting revenue and brand positioning within the prosthetic heart valve market.
High TAVR Device & Procedure Costs
Commercial US payers reimburse a median USD 71,312 for TAVR, versus Medicare's USD 37,865; Aetna tops at USD 84,190, and prices vary two-fold between New England and Pacific regions. Europe and North America together spend over USD 2 billion annually on TAVR, pressuring budgets. Emerging markets face larger hurdles, as devices can exceed annual per-capita healthcare outlays. Spain's incremental cost-effectiveness ratio of EUR 6,952 per QALY is below threshold, but payer constraints limit immediate uptake. Manufacturers are exploring value-based contracts, yet high prices remain a brake on the prosthetic heart valve market.
Other drivers and restraints analyzed in the detailed report include:
- Polymeric Valve R&D Breakthroughs
- AI-Guided Patient Selection & Sizing Tools
- Durability Concerns in Younger Cohorts
For complete list of drivers and restraints, kindly check the Table Of Contents.
Segment Analysis
Transcatheter heart valves accounted for 45.55% of 2024 revenue, underlining their rapid shift from salvage therapy to first-line option across risk profiles. This segment anchors the prosthetic heart valve market through streamlined procedures that shorten hospital stays and widen eligibility. Polymeric valves represent the fastest-growing niche, delivering an 18.25% CAGR through 2030 because materials resist calcification without anticoagulation, appealing to active patients. Tissue valves retain relevance for conventional surgery, while mechanical devices remain the choice for select younger users who accept lifelong anticoagulation in exchange for durability. Regulatory wins-such as Edwards' SAPIEN 3 for asymptomatic patients and the EVOQUE tricuspid system-keep transcatheter solutions at the forefront. Polymeric innovators Foldax and 4C Medical are reshaping durability expectations, catalyzing competitive differentiation. Clinical acceptance broadens as device platforms address multiple positions, reinforcing the prosthetic heart valve market's direction toward catheter-based therapy.
Sutureless platforms blur the line between open surgery and catheter techniques by offering shorter cross-clamp times and facilitating future valve-in-valve interventions. This hybrid evolution attracts surgeons who seek faster procedures without relinquishing operative control. Rapid-deployment valves, such as LivaNova's Perceval Plus, appeal to institutions balancing throughput and outcomes, prompting incremental share gains within the broader prosthetic heart valve market.
Aortic valves represented 56.53% of revenue in 2024, buttressed by a mature evidence base and streamlined reimbursement pathways. Patient demand remains strong because aortic stenosis prevalence rises with age, yet growth moderates as penetration in high-income markets stabilizes. Tricuspid interventions recorded a 15.15% forecast CAGR, the fastest among all positions, buoyed by Edwards' EVOQUE approval and trial success for Abbott's TriClip. Mitral programs gain momentum as the SAPIEN M3 CE mark unlocks the transfemoral approach. Specialized firms address pulmonary needs with devices such as the Venus P-valve for enlarged outflow tracts.
The prosthetic heart valve market share for aortic products is expected to narrow modestly as tricuspid and mitral growth outpaces traditional volumes. CMS coverage under Evidence Development for transcatheter tricuspid procedures will accelerate US uptake. Simultaneously, Asia-Pacific companies craft position-specific devices suited to local anatomies, such as Venus Medtech's mitral platform, diversifying competitive dynamics. As position specialization deepens, manufacturers secure regulatory timelines and clinical trials tailored to anatomical nuance, anchoring durable growth within the prosthetic heart valve market.
The Prosthetic Heart Valve Market Report is Segmented by Valve Type (Mechanical Heart Valve, Bioprosthetic Heart Valve, and More), Position (Mitral, Aortic, Tricuspid, and Pulmonary), Delivery Method (Surgical, Transcatheter and Sutureless), End-User (Tertiary-Care Hospitals, Cardiac Specialty Centers, and More), and Geography (North America, Europe, Asia Pacific and More). The Market Forecasts are Provided in Terms of Value (USD).
Geography Analysis
North America accounted for 42.52% of 2024 revenue, cementing its leadership through established reimbursement, robust clinical infrastructure, and early adopter mindsets. CMS coverage expansions drive procedure growth, and private insurers generally mirror Medicare's stance, ensuring broad access. The United States faces a looming 8,650-cardiologist shortfall by 2037, a constraint that may dampen procedure growth if training pipelines do not accelerate. Canada benefits from integrated provincial health systems and rising TAVR volumes. Mexico's modernizing private sector and cross-border medical tourism represent niche growth contributors.
Asia-Pacific delivers the fastest 14.12% CAGR through 2030, propelled by infrastructure investment, regulatory harmonization, and domestic innovation. China's National Medical Products Administration (NMPA) approval of MicroPort's VitaFlow Liberty Flex expands home-grown transcatheter options. Japan and South Korea leverage ageing populations and universal coverage to accelerate uptake. India exhibits long-term potential as cardiac programmes expand beyond metro hubs. Anatomical differences, notably smaller aortic annuli in East Asian patients, necessitate region-specific valve sizing and reinforce local R&D. Australia functions as a clinical trial nucleus, supporting regional skill transfer and evidence generation.
Europe sustains a balanced growth outlook, underpinned by coordinated regulation and strong clinician networks. Germany, United Kingdom, France, Italy, and Spain anchor procedural volumes, supported by long-standing TAVR programmes and standardized curricula. Edwards' SAPIEN M3 CE mark emphasises Europe's role as a launch pad for transfemoral mitral solutions. Eastern Europe lags but offers catch-up potential as economies converge. Meanwhile, the Middle East, Africa, and South America grow from a small base; selective investments in centres of excellence create regional hubs that train physicians and demonstrate outcomes, progressively widening access to the prosthetic heart valve market.
- Edward Lifesciences
- Medtronic
- Abbott Laboratories
- Boston Scientific
- Artivion (CryoLife)
- LivaNova
- MicroPort CardioFlow
- Jenavalve Technology
- Foldax Inc.
- Lepu Medical
- On-X Life Technologies
- Terumo Corp.
- Colibri Heart Valve
- TTK Healthcare
- Meril Life Sciences
- Teleflex (Chordis)
- NaviGate Cardiac Structures
- Peijia Medical
- Xeltis NV
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 Introduction
- 1.1 Study Assumptions & Market Definition
- 1.2 Scope of the Study
2 Research Methodology
3 Executive Summary
4 Market Landscape
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Aging Population & Rising VHD Prevalence
- 4.2.2 Expanding Indications For TAVR/TAVI
- 4.2.3 Regulatory Fast-Tracks & Breakthrough Designations
- 4.2.4 Reimbursement Expansion In Middle-Income Countries
- 4.2.5 Polymeric Valve R&D Breakthroughs
- 4.2.6 AI-Guided Patient Selection & Sizing Tools
- 4.3 Market Restraints
- 4.3.1 High TAVR Device & Procedure Costs
- 4.3.2 Durability Concerns In Younger Cohorts
- 4.3.3 Limited Cath-Lab Capacity Outside Tier-1 Cities
- 4.3.4 Surge In Valve-In-Valve Re-Interventions
- 4.4 Technological Outlook
- 4.5 Porter's Five Forces
- 4.5.1 Threat of New Entrants
- 4.5.2 Bargaining Power of Buyers
- 4.5.3 Bargaining Power of Suppliers
- 4.5.4 Threat of Substitutes
- 4.5.5 Intensity of Competitive Rivalry
5 Market Size & Growth Forecasts (Value, USD)
- 5.1 By Valve Type
- 5.1.1 Mechanical Heart Valve
- 5.1.2 Tissue/Bioprosthetic Heart Valve
- 5.1.3 Transcatheter Heart Valve (TAVR/TMVR/TTVR/TPVR)
- 5.1.4 Polymeric/Next-Gen Heart Valve
- 5.2 By Position
- 5.2.1 Aortic
- 5.2.2 Mitral
- 5.2.3 Tricuspid
- 5.2.4 Pulmonary
- 5.3 By Delivery Method
- 5.3.1 Surgical (SAVR/SMVR)
- 5.3.2 Transcatheter
- 5.3.3 Sutureless/Rapid-deployment
- 5.4 By End-user
- 5.4.1 Tertiary-care Hospitals
- 5.4.2 Cardiac Specialty Centers
- 5.4.3 Ambulatory Surgical Centers
- 5.5 Geography
- 5.5.1 North America
- 5.5.1.1 United States
- 5.5.1.2 Canada
- 5.5.1.3 Mexico
- 5.5.2 Europe
- 5.5.2.1 Germany
- 5.5.2.2 United Kingdom
- 5.5.2.3 France
- 5.5.2.4 Italy
- 5.5.2.5 Spain
- 5.5.2.6 Rest of Europe
- 5.5.3 Asia-Pacific
- 5.5.3.1 China
- 5.5.3.2 Japan
- 5.5.3.3 India
- 5.5.3.4 South Korea
- 5.5.3.5 Australia
- 5.5.3.6 Rest of Asia-Pacific
- 5.5.4 Middle East and Africa
- 5.5.4.1 GCC
- 5.5.4.2 South Africa
- 5.5.4.3 Rest of Middle East and Africa
- 5.5.5 South America
- 5.5.5.1 Brazil
- 5.5.5.2 Argentina
- 5.5.5.3 Rest of South America
- 5.5.1 North America
6 Competitive Landscape
- 6.1 Market Concentration
- 6.2 Market Share Analysis
- 6.3 Company Profiles (includes Global level Overview, Market level overview, Core Segments, Financials as available, Strategic Information, Market Rank/Share for key companies, Products & Services, and Recent Developments)
- 6.3.1 Edwards Lifesciences
- 6.3.2 Medtronic plc
- 6.3.3 Abbott Laboratories
- 6.3.4 Boston Scientific Corp.
- 6.3.5 Artivion (CryoLife)
- 6.3.6 LivaNova PLC
- 6.3.7 MicroPort CardioFlow
- 6.3.8 Jenavalve Technology
- 6.3.9 Foldax Inc.
- 6.3.10 Lepu Medical
- 6.3.11 On-X Life Technologies
- 6.3.12 Terumo Corp.
- 6.3.13 Colibri Heart Valve
- 6.3.14 TTK Healthcare
- 6.3.15 Meril Life Sciences
- 6.3.16 Teleflex (Chordis)
- 6.3.17 NaviGate Cardiac Structures
- 6.3.18 Peijia Medical
- 6.3.19 Xeltis NV
7 Market Opportunities & Future Outlook
- 7.1 White-space & Unmet-need Assessment




