PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1439845

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1439845
Transcutaneous Monitors - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts (2024 - 2029)
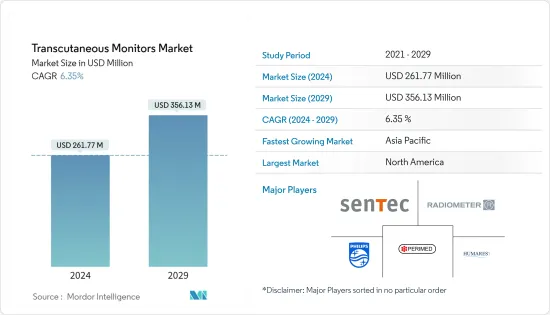
The Transcutaneous Monitors Market size is estimated at USD 261.77 million in 2024, and is expected to reach USD 356.13 million by 2029, growing at a CAGR of 6.35% during the forecast period (2024-2029).
The COVID-19 pandemic had a significant positive impact on the market studied. This was majorly attributed to the increasing number of patients being hospitalized for COVID-19, and the majority of them needed ventilation or continuous monitoring of oxygen levels. According to the European Respiratory Journal data published in March 2021, approximately 62.5% of the severe cases of COVID-19 in Europe had a history of COPD. Moreover, COVID-19 has also increased the burden on respiratory disease patients. This has led to the increasing demand for patient monitoring devices, such as transcutaneous monitors among critically ill patients, for better management of diseases. Hence, during the pandemic, the market studied witnessed significant growth. However, it has reached its pre-pandemic nature in terms of demand for transcutaneous monitors and is expected to witness healthy growth in the coming years.
The market studied is majorly driven by the factors such as the increasing prevalence of respiratory diseases, technological advancements in transcutaneous monitors, and the rising prevalence of chronic diseases.
Transcutaneous monitors play a crucial role in patient monitoring. The rising respiratory disease patient population often requires constant carbon dioxide and oxygen supervision with limited blood flow. Hence, transcutaneous monitors help monitor the oxygenation and ventilation in the patient's body and provide real-time information that is expected to help improve patient safety. As per the 2022 update by the Asthma and Allergy Foundation of America, approximately 25 million people in the United States have asthma. This equals about 1 in 13 people.
Additionally, as per the data from CDC published in October 2022, the percentage of adults who have ever been diagnosed with COPD, emphysema, or chronic bronchitis in the United States was 4.6% as of the year 2021. The CDC data also reported that the burden of respiratory diseases is increasing annually. Therefore, with the high prevalence of such diseases, there is a growing demand for transcutaneous monitors, which is believed to propel market growth in the coming years.
Furthermore, technological advancements in transcutaneous monitors contribute largely to market growth. For instance, as per the data from the National Institute of Health published in May 2021, wearable technology has emerged significantly and is widely practiced in transcutaneous monitors. The wearable transcutaneous CO2 monitor displays a linear response to CO2 concentration and a well-behaved response curve to humid CO2. Therefore, researchers are focusing on the development of a CO2 Monitor based on a Miniaturized Nondispersive Infrared Sensor. Hence, with such technologies, it is believed that there will be a wide range of transcutaneous monitors available in the market in the coming years, which will boost the market growth.
However, the stringent regulatory framework is expected to hinder market growth over the forecast period of study.
Transcutaneous Monitors Market Trends
Hospitals are Expected to Hold a Significant Share in the End User Segment
Hospitals are expected to hold a significant share in the end-user segment of the transcutaneous monitors market, which is majorly attributed to several services provided by the hospitals, including blood gas monitoring, wound healing, respiratory care, and sleep diagnostics.
Asthma continues to be a serious public concern globally. The United States Environmental Protection Agency and the National Health Interview Surveys (NHIS), in April 2021, estimated that almost 25.1 million people, comprising 5.1 million children, had asthma in the country as of the year 2021. Moreover, as per the data from the Australian Bureau of Statistics published in March 2022, around 10.7% of Australian had Asthma in the year 2021. Furthermore, the Indian Study on Epidemiology of Asthma, Respiratory Symptoms and Chronic Bronchitis in Adults (INSEARCH) published in April 2022 estimated the national burden of asthma at 17.23 million with an overall prevalence of 2.05% during the year 2021. Therefore, the rising cases of asthma across various countries are increasing the demand for transcutaneous monitors in hospitals.
Furthermore, as per Tommys.org, in 2021, around 8% of births in the United Kingdom were preterm. That was around 60,000 babies each year. Transcutaneous monitoring in neonates provides a real-time overview of the patient's often fluctuating oxygenation (tcpO2) and ventilation (tcpCO2) status. Hence, the demand for transcutaneous oximetry systems in hospitals is also expected to rise due to the increasing number of preterm births.
Thus, hospitals are expected to hold a significant share of the end-user segment in the transcutaneous monitor market due to the aforementioned factors.
North America is Expected to Hold a Considerable Share in the Market and Expected to do Same in the Forecast Period
North America is expected to hold a major market share in the Transcutaneous Monitors Market. This can be attributed to the rising number of respiratory disorders leading to increasing demand for non-invasive monitoring, a growing number of hospitals, increasing healthcare expenditure, and well-established healthcare infrastructure.
According to National Center for Chronic Disease Prevention and Health Promotion update in 2021, preterm birth affected 1 of every ten infants born in the United States in 2021. The rise in preterm birth is expected to boost the demand for neonatal monitoring systems in the United States, as preterm birth babies are more susceptible to diseases like sepsis, pneumonia, etc. Moreover, non-invasive monitoring of oxygenation has become a standard procedure in neonatal critical care in the United States, which is expected to boost the market growth.
Furthermore, strategic collaborations, acquisitions, expansion, product launches, agreements, and research sponsorship are the key strategies adopted by manufacturers to increase product sales in the United States. For instance, in December 2021, Royal Philips announced that its Philips Patient Monitors MX750 and MX850 received clearance from the United States Food and Drug Administration. Initially cleared for Emergency Use Authorization, the MX750 and MX850 were Philips' most advanced patient monitors uniquely designed to support scalability, alarm management, cybersecurity, and infection prevention within the hospital. Therefore, with such new launches from the key companies, the market studied is believed to witness substantial growth over the forecast period.
Transcutaneous Monitors Industry Overview
The Transcutaneous Monitors Market is moderately competitive and consists of several major players. Key companies are implementing strategies such as partnerships, collaborations, and new product launch to sustain in the global market. Some of the companies which are currently operating in the market are Sentec AG, Radiometer, HUMARES GMBH, Primed AB, and Koninklijke Philips N.V.
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 Study Assumptions
- 1.2 Scope of the Study
2 RESEARCH METHODOLOGY
3 EXECUTIVE SUMMARY
4 MARKET DYNAMICS
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Increasing Prevalence of Respiratory Diseases
- 4.2.2 Technological Advancements in Transcutaneous Monitors
- 4.2.3 Rising Prevalence of Chronic Disorders
- 4.3 Market Restraints
- 4.3.1 Stringent Regulatory Framework
- 4.4 Porter's Five Force Analysis
- 4.4.1 Threat of New Entrants
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Bargaining Power of Suppliers
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5 MARKET SEGMENTATION
- 5.1 By Application Type
- 5.1.1 Wound Healing
- 5.1.2 Blood Gas Monitoring
- 5.1.3 Respiratory Care
- 5.1.4 Sleep Diagnostics
- 5.1.5 Others
- 5.2 By End User
- 5.2.1 Hospitals
- 5.2.2 Ambulatory Surgical centers
- 5.2.3 Speciality Clinics
- 5.3 Geography
- 5.3.1 North America
- 5.3.1.1 United States
- 5.3.1.2 Canada
- 5.3.1.3 Mexico
- 5.3.2 Europe
- 5.3.2.1 Germany
- 5.3.2.2 United Kingdom
- 5.3.2.3 France
- 5.3.2.4 Italy
- 5.3.2.5 Spain
- 5.3.2.6 Rest of Europe
- 5.3.3 Asia-Pacific
- 5.3.3.1 China
- 5.3.3.2 Japan
- 5.3.3.3 India
- 5.3.3.4 Australia
- 5.3.3.5 South Korea
- 5.3.3.6 Rest of Asia-Pacific
- 5.3.4 Middle-East and Africa
- 5.3.4.1 GCC
- 5.3.4.2 South Africa
- 5.3.4.3 Rest of Middle-East and Africa
- 5.3.5 South America
- 5.3.5.1 Brazil
- 5.3.5.2 Argentina
- 5.3.5.3 Rest of South America
- 5.3.1 North America
6 COMPETITIVE LANDSCAPE
- 6.1 Company Profiles
- 6.1.1 Sentec AG
- 6.1.2 Radiometer
- 6.1.3 HUMARES GMBH
- 6.1.4 Primed AB
- 6.1.5 Koninklijke Philips N.V
7 MARKET OPPORTUNITIES AND FUTURE TRENDS




