PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1406013

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1406013
Pelvic Inflammatory Disease Therapeutics - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts 2024 - 2029
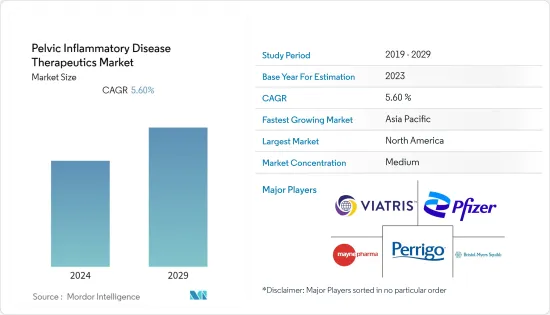
The pelvic inflammatory disease therapeutics market studied was anticipated to witness a CAGR of 5.6% during the forecast period.
The studied market was adversely impacted by the pandemic. Various research studies provide insight into the impact of the pandemic on the market. According to the article titled, 'Sexually transmitted infections during the COVID-19 outbreak: comparison of patients referring to the service of sexually transmitted diseases during the sanitary emergency with those referring during the common practice' was published by researchers based in Italy in June 2020. It stated that before the national lockdown in Italy, a total of 1,081 medical provisions were delivered by the STDs Service of Dermatology, Bologna. The percentage of visits for prophylaxis declined during and after the lockdown, while visits for syphilis, gonococcal pharyngitis, and inflammatory genital diseases increased significantly. As per the article titled, 'Measuring the Impact of the COVID-19 Pandemic on Sexually Transmitted Diseases Public Health Surveillance and Program Operations in the State of California', published in August 2020, a comparative study for January-June 2020 to January-June of 2019 data found that there were decreases in reported cases of chlamydia (31%), late syphilis (19%), primary/secondary syphilis (15%), early nonprimary non-secondary syphilis (14%), and gonorrhea (13%) during the COVID-19 pandemic. Thus, the market was adversely impacted by the pandemic.
The major factors attributing to the growth of the market are a rise in urinary tract infections, sexually transmitted diseases (STDs), and severe complications associated with pelvic inflammatory diseases (PID), such as increased rate of Polycystic ovary syndrome (PCOS) cases, and infertility. According to the article by Ayan Sabih and Stephen W. Leslie, published by the National Institute of Health in February 2021, complicated UTI incidence was highly associated with specific risk factors. For instance, there is a 10% estimated daily risk of developing bacteriuria with indwelling bladder catheters, and there is up to a 25% risk that bacteriuria will progress to a urinary tract infection. In June 2022, the Centers for Disease Control and Prevention recommended and issued guidelines for screening sexually transmitted diseases to create awareness toward screening programs for STDs and the increasing burden of STDs across the world. It has issued separate STD screening guidelines for every group of the population, such as women, pregnant women, men who have sex with women, men who have sex with men, transgender and gender-diverse persons, and persons with HIV. However, in March 2022, the Department of Health of the State of New Jersey organized the Sexually Transmitted Disease (STD) Program to reduce and prevent the effects of sexually transmitted diseases in New Jersey through partner services, education, technical assistance, surveillance, and partnerships with local health and community organizations. Such programs would increase awareness regarding the disease and its treatment, which is expected to increase the demand for PID therapeutics. Therefore, such programs are anticipated to positively impact the market over the forecast period.
However, the stringent regulatory framework that delays the approval of products, expensive patented products, and severe complications associated with the medication are expected to hinder market growth.
Pelvic Inflammatory Disease Therapeutics Market Trends
The Tetracyclines Segment is Expected to Hold the Major Share in the Pelvic Inflammatory Disease Therapeutics Market
Tetracycline drugs are expected to dominate the industry over the forecast period. The lucrative growth of the segment can be attributed to the rise in the adoption of these products due to proven efficiency in the treatment and recommendations of cephalosporin along with doxycycline, a tetracycline drug by the Centers for Disease Control and Prevention (CDC) as the first-line treatment of pelvic inflammatory disease. Rapid advancements in new product development with reduced side effects, coupled with the availability of a wide range of marketed products, are expected to fuel the market. The report published by the World Health Organization in November 2021 reported that more than 1.0 million sexually transmitted infections are acquired globally, and most of them are asymptomatic. It also reported that every year, there are an estimated 374.0 million new infections, with 1 out of 4 sexually transmitted infections: gonorrhea, chlamydia, trichomoniasis, and syphilis. Tetracyclines (tetracycline, doxycycline, minocycline, and tigecycline) are a class of drugs used to manage and cure numerous bacterial infections, according to a research report released in July 2022 under the title "Tetracycline." Tetracyclines are categorized as broad-spectrum antibiotics that suppress protein synthesis. Tetracyclines are an effective treatment for bacterial infections and their indications, mode of action, and contraindications. Such research studies will emphasize the mechanism of action, adverse event profile, and pertinent interactions important for healthcare team members involved in treating these illnesses and infected patients. Thus, such advantages provided in research studies are expected to increase the adoption of drugs, which is expected to increase the market growth.
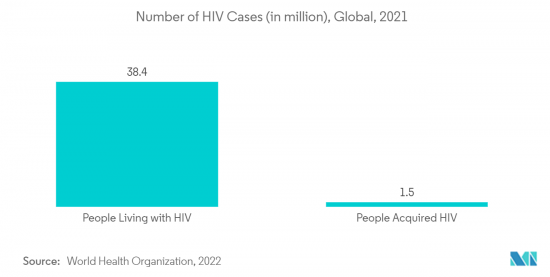
North America Dominates the Market and it is Expected to do the Same in the Forecast Period
North America is expected to dominate the studied market throughout the forecast period. This dominance is mainly owing to rising incidences of menopausal disorders, polycystic ovary syndrome, uterine fibroids, endometriosis, trichomoniasis, HIV/AIDS, etc., due to changes in lifestyles and a rise in stress among women in the United States, which registers the highest revenue in the region. For instance, in April 2023, a report published by the Centers for Disease Control and Prevention (CDC) stated that STDs remain a significant public health concern in the country. According to the same source, Chlamydia trachomatis infection was the most prevalent notifiable sexually transmitted infection in the United States in 2021, with a total of 1,644,416 cases reported to the CDC. The CDC received reports of 710,151 different gonorrhea infections in 2021. A total of 176,713 cases of syphilis were registered in 2021 throughout all phases, including 53,767 cases of the disease's primary and secondary (P&S) stages, which are the most contagious. By raising the demand for such diseases' therapeutic processes, the country's market growth is thus anticipated to expand with the prevalence of STDs. Thus, the increasing prevalence of STIs is expected to increase the demand for therapeutics and will increase market growth.
Pelvic Inflammatory Disease Therapeutics Industry Overview
The pelvic inflammatory disease therapeutics market is moderately competitive and consists of several major players. Some prominent players in the market are implementing various strategies, such as launching new products and making acquisitions to consolidate their market positions across the world. Furthermore, a steep rise in the availability of public funding for the development of these drugs is also expected to fuel the market. Some of the companies that are currently dominating the market are BristolMyers Squibb Company, Pfizer Inc., Mayne Pharma, Perrigo Company PLC, and Viatris, among others.
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope of the Study
2 RESEARCH METHODOLOGY
3 EXECUTIVE SUMMARY
4 MARKET DYNAMICS
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Increasing Prevalence of STD and Infectious Diseases Across the World
- 4.2.2 Rise in Healthcare Awareness along with the Increasing Aging Female Population
- 4.3 Market Restraints
- 4.3.1 Stringent Regulatory Framework that Delays the Approval of Products
- 4.3.2 Expensive Patented Products and Severe Complications Associated with the Medication
- 4.4 Porter's Five Forces Analysis
- 4.4.1 Threat of New Entrants
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Bargaining Power of Suppliers
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5 MARKET SEGMENTATION (Market Size by Value - USD million)
- 5.1 By Drug Class
- 5.1.1 Macrolides
- 5.1.2 Tetracycline
- 5.1.3 Beta-lactam
- 5.1.4 Nitroimidazoles
- 5.1.5 Other Drug Classes
- 5.2 By Route of Administration
- 5.2.1 Oral
- 5.2.2 Parenteral
- 5.2.3 Other Routes of Administration
- 5.3 By Distribution Channel
- 5.3.1 Hospital Pharmacies
- 5.3.2 Retail Pharmacies
- 5.3.3 Online Pharmacies
- 5.4 Geography
- 5.4.1 North America
- 5.4.1.1 United States
- 5.4.1.2 Canada
- 5.4.1.3 Mexico
- 5.4.2 Europe
- 5.4.2.1 Germany
- 5.4.2.2 United Kingdom
- 5.4.2.3 France
- 5.4.2.4 Italy
- 5.4.2.5 Spain
- 5.4.2.6 Rest of Europe
- 5.4.3 Asia-Pacific
- 5.4.3.1 China
- 5.4.3.2 Japan
- 5.4.3.3 India
- 5.4.3.4 Australia
- 5.4.3.5 South Korea
- 5.4.3.6 Rest of Asia-Pacific
- 5.4.4 Middle-East and Africa
- 5.4.4.1 GCC
- 5.4.4.2 South Africa
- 5.4.4.3 Rest of Middle-East and Africa
- 5.4.5 South America
- 5.4.5.1 Brazil
- 5.4.5.2 Argentina
- 5.4.5.3 Rest of South America
- 5.4.1 North America
6 COMPETITIVE LANDSCAPE
- 6.1 Company Profiles
- 6.1.1 AstraZeneca
- 6.1.2 Bristol-Myers Squibb Company
- 6.1.3 F. Hoffmann-La Roche Ltd
- 6.1.4 Galderma Laboratories LP
- 6.1.5 Janssen Pharmaceuticals, Inc. (Johnson and Johnson)
- 6.1.6 Mayne Pharma
- 6.1.7 Perrigo Company PLC
- 6.1.8 Pfizer Inc.
- 6.1.9 Sanofi SA
- 6.1.10 Teligent Inc.
- 6.1.11 Viatris
7 MARKET OPPORTUNITIES AND FUTURE TRENDS




