PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1406567

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1406567
Asia Pacific In Vitro Diagnostics - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts 2024 - 2029
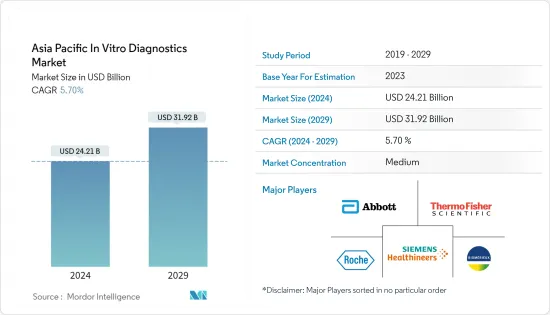
The Asia Pacific In Vitro Diagnostics Market size is estimated at USD 24.21 billion in 2024, and is expected to reach USD 31.92 billion by 2029, growing at a CAGR of 5.70% during the forecast period (2024-2029).
The COVID-19 outbreak affected the Asia-Pacific in-vitro diagnostic market's growth adversely in its preliminary phase. However, this market gained traction due to the significant urge and rapid demand for newly launched emergency-use authorized in-vitro diagnostic tests for COVID-19 detection. For instance, in May 2022, Cipla Limited started the commercialization of the 'RT-Direct' multiplex COVID-19 RT-PCR Test kit in India in partnership with Genes2Me Pvt. Ltd. This kit was validated at an Indian Council of Medical Research (ICMR)-approved center. However, the rapid demand for newly launched emergency-use authorized in-vitro diagnostic tests for COVID-19 detection compensated for this detrimental impact. However, the market is growing at a stable pace due to a resumption of diagnosis procedures, manufacturing activities, and availability of products. It is expected to witness a similar trend over the coming years.
Further, the high cases of chronic diseases, the increasing use of point-of-care (POC) diagnostics, advanced technologies in vitro diagnostic products, and increasing awareness and use of personalized medicine and companion diagnostics are among the major factors driving the growth of the studied market. For instance, the 2021 International Diabetes Federation (IDF) projects that the number of people with diabetes in Southeast Asia will increase by 68%, reaching 152 million by 2045. The same report also reported that the Western Pacific region holds the third-highest prevalence of diabetes (11.9%) globally. The number of diabetic cases is estimated to increase in the Western Pacific region by 27%, reaching 260 million by 2045, and the prevalence of diabetes will increase by 21% in 2045. Such a high incidence of chronic diseases like diabetes in the given region is increasing the demand for diagnostics associated with the diseases.
Additionally, as per the article published in the Chinese Medical Journal in March 2022, China is experiencing a greater cancer incidence. In China, there were about 4.8 million new cancer cases in 2022, with lung cancer being the most prevalent type. Thus, the increasing incidence of cancer cases increased the demand for associated diagnostics, contributing to the market.
The strategies adopted by the market players, such as product launches, partnerships, and collaboration, are expected to propel the market's growth. For instance, in March 2021, OpGen announced that it had received regulatory approval from the Chinese National Medical Products Administration for its Curetis Unyvero System as an in-vitro diagnostics instrument. In April 2021, Otsuka Pharmaceutical Co., Ltd., in collaboration with Denka Company Limited, launched the Helicobacter Pylori test kit, QuickNavi-H. Pylori. It is a diagnostic kit that utilizes immunochromatography to confirm the presence or absence of Helicobacter Pylori antigens in a stool sample. The test kit will be used in medical institutions as it enables rapid evaluation in eight minutes by adding three drops of the sample liquid to the kit. Thus, the market can show significant growth over the forecast period due to these technological advances.
Thus, the reasons mentioned above may drive the market. However, stringent regulations regarding product approvals and cumbersome reimbursement procedures may slow the market's growth.
Asia Pacific In-Vitro Diagnostics Market Trends
Clinical Chemistry Segment is Expected to Record the Highest Growth in the Forecast Period
Clinical chemistry is a combination of analytics, instrumentation, information technology, and the management of workflow. Technological advancements increased the automation of detection techniques for biologically important ions (salts and minerals), small organic molecules, and large molecules, such as enzymes, transport proteins, and lipoproteins.
Furthermore, the increasing prevalence of target diseases, like liver diseases, thyroid functions, cancers, etc., boosts the segment's growth. For instance, in 2022, an article published in Therapeutic Advances in Endocrinology and Metabolism reported that the disease burden of thyroid cancer in the high-income Asia-Pacific region was significantly higher than that in other regions, possibly due to overdiagnosis. The increasing incidence of thyroid cancer indicates that it is still a public health problem in Asia. For instance, according to the International Diabetes Federation Diabetes Atlas 2021, the age-adjusted comparative prevalence of diabetes in China for 2021 is estimated as 10.6%, for 2030 as 11.8%, and for 2045 as 12.5%. Hence, a rise in thyroid cancer and diabetes directly demands portable or benchtop clinical chemistry analyzers. It is expected to reduce healthcare costs and lead to an enhanced diagnosis and treatment rate, propelling the clinical chemistry analyzer segment.
Moreover, product launches by the market players are expected to boost the segment's growth. For instance, in May 2022, Mindray, a China-based company, launched BS-600M. This powerful yet efficient chemistry analyzer empowers medium-volume laboratories with increased productivity, efficiency, and reliability.
Thus, all the factors above are expected to boost segment growth over the forecast period.
India is Expected Hold a Significant Share Over the Forecast Period
The increasing R&D expenditures, the growing burden of chronic diseases, the aging population, and the well-established healthcare infrastructure with advanced diagnostic facilities in the country are expected to drive market growth in Japan.
The increasing number of cases of chronic diseases such as cancer, diabetes, and others in the country is the key factor driving the market's growth. For instance, as per the ICMR 2021 Report, it was observed that the number of cancer patients in India was expected to rise from 26.7 million in 2021 to 29.8 million in 2025. As per 2022 statistics published by the IDF, about 74.1 million people had diabetes in India in 2021, and this number was estimated to reach 92.9 million and 124.8 million by 2030 and 2045, respectively. Thus, the rising burden of cancer raises the need to detect cancer early for more effective and timely treatment, increasing the demand for in-vitro diagnostics. It is anticipated to boost the market's growth over the forecast period.
The product launches by the market players are expected to boost the market in the country. For instance, in August 2021, Mylab Discovery Solutions entered a technology partnership with Hemex Health to develop next-generation diagnostic solutions for point-of-care (POC) testing of coronaviruses and other diseases. Mylab develops test assays in this partnership, and Hemex provides its Gazelle POC testing platform and expertise. Additionally, in August 2022, Molbio Diagnostics and Truenat Technology launched a new test for the differential diagnosis of HIV 1 and HIV 2, the Truenat RT-PCR Test, with viral loads within 60 minutes. The Truenat is a point-of-care portable, battery-operated, IoT-enabled platform that can test for over 35 diseases, including COVID-19, TB, hepatitis, HIV, HPV, dengue, malaria, etc.
Thus, all the factors above are expected to boost the market over the forecast period.
Asia Pacific In-Vitro Diagnostics Industry Overview
The Asia-Pacific in vitro diagnostics market is highly competitive and includes several major players. Regarding market share, a few major players currently dominate the market. However, with technological advancements and product innovations, mid-size to smaller companies are increasing their market presence by introducing new products at lower prices. Some of the major players in the region are BioMerieux SA, Danaher (Beckman Coulter), F Hoffmann-La Roche Ltd., Becton, Dickinson and Company, Bio-Rad Laboratories Inc., Abbott Laboratories, Arkray Inc., Sysmex Corporation, Ortho Clinical Diagnostics, Siemens Healthineers, Thermo Fischer Scientific Inc., and Qiagen NV.
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope of the Study
2 RESEARCH METHODOLOGY
3 EXECUTIVE SUMMARY
4 MARKET DYNAMICS
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 High Prevalence of Chronic Diseases
- 4.2.2 Increasing Use of Point-of-care (POC) Diagnostics
- 4.2.3 Advanced Technologies in In-vitro Diagnostic Products
- 4.2.4 Increasing Awareness and Acceptance of Personalized Medicine and Companion Diagnostics
- 4.3 Market Restraints
- 4.3.1 Stringent Regulations Regarding Product Approvals
- 4.3.2 Cumbersome Reimbursement Procedures
- 4.4 Industry Attractiveness - Porter's Five Forces Analysis
- 4.4.1 Bargaining Power of Suppliers
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Threat of New Entrants
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5 MARKET SEGMENTATION (Market Size by Value - USD)
- 5.1 By Test Type
- 5.1.1 Clinical Chemistry
- 5.1.2 Molecular Diagnostics
- 5.1.3 Immuno Diagnostics
- 5.1.4 Hematology
- 5.1.5 Other Test Types
- 5.2 By Product
- 5.2.1 Instruments
- 5.2.2 Reagents
- 5.2.3 Other Products
- 5.3 By Usability
- 5.3.1 Disposable IVD Devices
- 5.3.2 Reusable IVD Devices
- 5.4 By Application
- 5.4.1 Infectious Diseases
- 5.4.2 Diabetes
- 5.4.3 Cancer/Oncology
- 5.4.4 Cardiology
- 5.4.5 Autoimmune Diseases
- 5.4.6 Nephrology
- 5.4.7 Other Applications
- 5.5 By End User
- 5.5.1 Diagnostic Laboratories
- 5.5.2 Hospitals and Clinics
- 5.5.3 Other End Users
- 5.6 By Geography
- 5.6.1 China
- 5.6.2 Japan
- 5.6.3 India
- 5.6.4 Australia
- 5.6.5 South Korea
- 5.6.6 Rest of Asia-Pacific
6 COMPETITIVE LANDSCAPE
- 6.1 Company Profiles
- 6.1.1 BioMerieux SA
- 6.1.2 Danaher (Beckman Coulter)
- 6.1.3 F Hoffmann-La Roche Ltd.
- 6.1.4 Becton, Dickinson and Company
- 6.1.5 Bio-Rad Laboratories Inc.
- 6.1.6 Abbott Laboratories
- 6.1.7 Arkray Inc.
- 6.1.8 Sysmex Corporation
- 6.1.9 Quidel Corporation (Ortho Clinical Diagnostics)
- 6.1.10 Siemens Healthineers
- 6.1.11 Thermo Fischer Scientific Inc.
- 6.1.12 Qiagen NV
7 MARKET OPPORTUNITIES AND FUTURE TRENDS




