PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1937324

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1937324
Tonometer - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts (2026 - 2031)
Tonometer market size in 2026 is estimated at USD 392.85 million, growing from 2025 value of USD 374.57 million with 2031 projections showing USD 498.67 million, growing at 4.88% CAGR over 2026-2031.
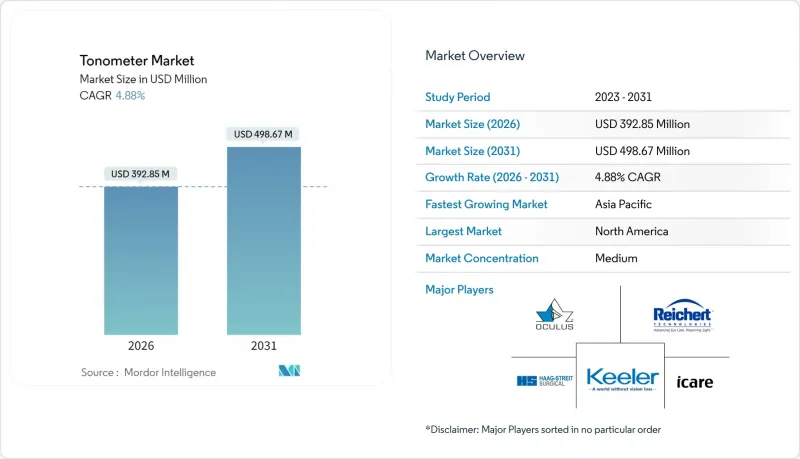
Sustained growth in the tonometer market reflects an escalating glaucoma burden, widening screening programs, and technological shifts toward remote, non-contact devices that lower infection risk and support longitudinal intraocular-pressure (IOP) monitoring. Demographic pressure from aging and diabetic populations galvanizes product development around home-based measurement, while hospitals and specialty centers strengthen procurement to address rising caseloads. Competitive intensity remains moderate as incumbents race to secure regulatory clearances for AI-enabled wearables and smartphone-compatible probes that promise continuous measurement and data integration. In parallel, healthcare payers in North America and parts of Europe expand reimbursement for preventive testing, enhancing adoption prospects across both desktop and handheld platforms.
Global Tonometer Market Trends and Insights
Rising Prevalence of Glaucoma
Rising global glaucoma incidence provides a structural demand floor for tonometry devices. Glaucoma affects 80 million people in 2025 and is projected to reach 111 million by 2040, with Asia accounting for 60% of cases. In the United States, 4.22 million individuals live with the condition, prompting sustained purchasing by hospital networks that support high-volume screening and monitoring. Targeted programs such as CDC SIGHT Studies report 27% detection rates in high-risk communities when portable rebound tonometers are deployed in federally qualified health centers. These statistics underscore why the tonometer market continues to enjoy resilient baseline demand, particularly in emerging regions where undiagnosed cases remain commonplace and screening infrastructure is expanding.
Growing Geriatric & Diabetic Populations
Population aging and rising diabetes prevalence amplify glaucoma risk and fuel demand for continuous IOP assessment. Device loans involving the iCare HOME2 allow optometrists to build patient-specific pressure profiles over 2.5-year horizons, improving therapeutic decisions and adherence. Smart contact lenses capable of measuring both tear glucose and IOP illustrate converging chronic-disease monitoring needs. As value-based care models scale, payers increasingly reimburse longitudinal monitoring, encouraging manufacturers to shift toward subscription-driven consumables and analytics rather than one-time hardware sales. These dynamics reinforce the tonometer market trajectory in both developed and developing economies.
Infection Risk with Contact Devices & Accuracy Gaps in Air-Puff Tonometry
Contact tonometry remains the clinical benchmark but introduces sterilization burdens that became more acute during heightened infection-control protocols. Some facilities pivoted to air-puff systems yet later observed precision deficits in patients with corneal irregularities, prompting parallel use of both modalities and inflating capital requirements. Smartphone tonometers achieve only 63% accuracy within +-2 mmHg versus Goldmann standards, amplifying practitioner hesitancy for high-risk cases. The resulting workflow complexity can slow decision-making and dilute user confidence, tempering purchase velocity for certain device classes.
Other drivers and restraints analyzed in the detailed report include:
- Expansion of Eye-Care Screening Programs
- Rapid Adoption of Non-Contact & Rebound Devices
- High Upfront Cost of Advanced Systems
For complete list of drivers and restraints, kindly check the Table Of Contents.
Segment Analysis
Applanation units captured 47.25% of the tonometer market share in 2025 due to entrenched clinical familiarity and well-documented accuracy thresholds. Revenue stability persists as teaching hospitals retain Goldmann platforms for benchmark validation, yet growth tilts toward non-contact categories. Rebound instruments are charting a 6.28% CAGR, propelled by AMETEK's Tono-Vera launch that satisfied ANSI / ISO +-3 mmHg criteria in 2025 laboratory assessments. The tonometer market size attributable to rebound systems is expected to expand by 2031 in tandem with infection-control imperatives. Dynamic contour and indentation devices remain niche yet offer value in corneal-thickness-adjusted diagnostics and veterinary applications.
Clinical validation underpins adoption curves. Peer-reviewed trials show TRK-3 OMNIA non-contact systems deliver +-5 mmHg equivalence in 89% of readings, reassuring ophthalmologists about interoperability with legacy datasets. FDA 510(k) filings referencing Product Code HKY rose 15% year over year, signaling an active pipeline of modular probes that accept future sensor upgrades. With reimbursement codes already established, the tonometer market welcomes new entrants that iterate on optical alignment, AI-assisted auto-focus, and subscription-based calibration alerts. As evidence accrues, applanation's dominance will continue to erode, though a sizeable installed base assures a long tail of service revenue for incumbent manufacturers.
Desktop configurations maintained 67.70% of the tonometer market size in 2025, anchored by integrated lanes within hospital ophthalmology suites. Their sustained presence hinges on workflow synergy with slit-lamps, autorefractors, and corneal pachymeters found in high-volume centers. Handheld models, however, are set to record a 6.41% CAGR, buoyed by screening caravans, tele-ophthalmology kits, and home-monitoring prescriptions that decentralize care. Optometrists report that handheld loans reduce clinic visits by 23% over two years, producing cost offsets that validate device rentals.
Design evolution favors lightweight casings, long-life batteries, and Bluetooth Low Energy for smartphone pairing. Manufacturers highlight single-button operation and automated multi-reading averaging, addressing training limitations among community-health workers. The tonometer market is therefore witnessing convergence similar to glucometer pathways where primary units anchor data ecosystems feeding cloud dashboards. Desktop systems retain an edge in repeatability and integration, yet coastline clinics, cruise ships, and rural field units increasingly select handheld rebound probes for logistical flexibility. Co-existence will persist, but sub-segments will gravitate toward devices that optimize for either throughput or portability.
The Global Tonometer Market Report is Segmented by Technology (Applanation Tonometry, Indentation Tonometry, Rebound Tonometry, and Other Technologies), Portability Type (Desktop, Handheld), End User (Hospitals, Ophthalmic Centers, Home-Care Settings, Others), Measurement Type (Direct, Indirect), and Geography (North America, Europe, Asia-Pacific, and Others). The Market Forecasts are Provided in Terms of Value (USD).
Geography Analysis
North America held a 39.20% tonometer market share in 2025, leveraging well-established reimbursement schemes and broad glaucoma screening coverage. The CDC SIGHT initiative elevates detection rates in at-risk populations, pushing device utilization in federally qualified centers above historical averages. U.S. hospitals have adopted AI-ready tonometers that integrate with EHRs, while Canadian clinics explore leasing models to accelerate technology refresh without capital spikes. Cross-border regulatory alignment under the United States-Mexico-Canada Agreement eases product circulation and amplifies after-sales service footprints.
Asia-Pacific is set to record a 6.49% CAGR to 2031 as healthcare infrastructure expansion meets the reality that 60% of global glaucoma cases reside in the region. China's procurement budgets favor portable rebound units for provincial outreach, and India's public-health pilots demonstrate favorable cost-per-case averted when handheld devices supplement fixed clinics. Japan and South Korea, already mature device markets, are early adopters of sensor implants now entering post-approval surveillance. Singapore's selective subsidy schema tips demand toward mid-tier devices that blend cost efficiency with acceptable precision.
Europe maintains balanced growth thanks to aging demographics and harmonized CE regulations. Germany spearheads adoption of continuous-monitoring spectacles, while the United Kingdom extends value-based reimbursement to include home-use tonometry kits. Southern European nations deploy mobile ophthalmology buses equipped with non-contact devices to serve rural zones, sustaining replacement cycles for portable units. Brexit regulatory divergence introduces transient logistical hurdles, yet established distributor networks mitigate disruption, preserving regional sales momentum within the tonometer market.
- Haag-Streit Group (Metall Zug)
- Topcon
- Revenio Group Plc (iCare)
- Carl Zeiss
- AMETEK Inc. (Reichert Technologies)
- Keeler Ltd. (Halma)
- Nidek
- Oculus Optikgerate GmbH
- Kowa Co., Ltd.
- Canon
- Rexxam
- Tomey
- Huvitz Co., Ltd.
- Marco Ophthalmic
- Ziemer Group
- 66 Vision Tech Co., Ltd.
- CSO Srl
- Suzhou Kangjie Medical
- MediWorks Optical
- Diaton (BiCOM Inc.)
- Suowei Electronic Technology
- Optomed Plc
- Bausch Health
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 Introduction
- 1.1 Study Assumptions & Market Definition
- 1.2 Scope of the Study
2 Research Methodology
3 Executive Summary
4 Market Landscape
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Rising Prevalence of Glaucoma
- 4.2.2 Growing Geriatric & Diabetic Populations
- 4.2.3 Expansion of Eye-Care Screening Programs
- 4.2.4 Rapid Adoption of Non-Contact & Rebound Devices
- 4.2.5 Tele-Ophthalmology Integration Enabling Remote IOP Monitoring
- 4.2.6 AI-Enabled Continuous IOP Implants & Wearables
- 4.3 Market Restraints
- 4.3.1 Infection Risk with Contact Devices & Accuracy Gaps In Air-Puff Tonometry
- 4.3.2 High Upfront Cost of Advanced Systems
- 4.3.3 Proliferation of Low-Cost Smartphone-Based IOP Apps
- 4.3.4 Sensor-Supply Dependence on Tight Semiconductor Capacity
- 4.4 Supply-Chain Analysis
- 4.5 Regulatory Landscape
- 4.6 Technological Outlook
- 4.7 Porter's Five Forces
- 4.7.1 Bargaining Power of Suppliers
- 4.7.2 Bargaining Power of Buyers / Consumers
- 4.7.3 Threat of New Entrants
- 4.7.4 Threat of Substitute Products
- 4.7.5 Intensity of Competitive Rivalry
5 Market Size & Growth Forecasts
- 5.1 By Technology (Value)
- 5.1.1 Applanation Tonometry
- 5.1.2 Indentation Tonometry
- 5.1.3 Rebound Tonometry
- 5.1.4 Dynamic Contour Tonometry
- 5.1.5 Other Technologies
- 5.2 By Portability Type (Value)
- 5.2.1 Desktop
- 5.2.2 Handheld
- 5.3 By End User (Value)
- 5.3.1 Hospitals
- 5.3.2 Ophthalmic Centers
- 5.3.3 Home-Care Settings
- 5.3.4 Others
- 5.4 By Measurement Type (Value)
- 5.4.1 Direct
- 5.4.2 Indirect
- 5.5 By Geography (Value)
- 5.5.1 North America
- 5.5.1.1 United States
- 5.5.1.2 Canada
- 5.5.1.3 Mexico
- 5.5.2 Europe
- 5.5.2.1 Germany
- 5.5.2.2 United Kingdom
- 5.5.2.3 France
- 5.5.2.4 Italy
- 5.5.2.5 Spain
- 5.5.2.6 Rest of Europe
- 5.5.3 Asia-Pacific
- 5.5.3.1 China
- 5.5.3.2 Japan
- 5.5.3.3 India
- 5.5.3.4 Australia
- 5.5.3.5 South Korea
- 5.5.3.6 Rest of Asia-Pacific
- 5.5.4 Middle East and Africa
- 5.5.4.1 GCC
- 5.5.4.2 South Africa
- 5.5.4.3 Rest of Middle East and Africa
- 5.5.5 South America
- 5.5.5.1 Brazil
- 5.5.5.2 Argentina
- 5.5.5.3 Rest of South America
- 5.5.1 North America
6 Competitive Landscape
- 6.1 Market Concentration
- 6.2 Strategic Moves
- 6.3 Market Share Analysis
- 6.4 Company Profiles {(includes Global level Overview, Market level overview, Core Segments, Financials as available, Strategic Information, Market Rank/Share for key companies, Products, and Recent Developments)}
- 6.4.1 Haag-Streit Group (Metall Zug)
- 6.4.2 Topcon Corporation
- 6.4.3 Revenio Group Plc (iCare)
- 6.4.4 Carl Zeiss Meditec AG
- 6.4.5 AMETEK Inc. (Reichert Technologies)
- 6.4.6 Keeler Ltd. (Halma)
- 6.4.7 Nidek Co., Ltd.
- 6.4.8 Oculus Optikgerate GmbH
- 6.4.9 Kowa Co., Ltd.
- 6.4.10 Canon Medical Systems Corp.
- 6.4.11 Rexxam Co., Ltd.
- 6.4.12 Tomey Corporation
- 6.4.13 Huvitz Co., Ltd.
- 6.4.14 Marco Ophthalmic
- 6.4.15 Ziemer Ophthalmic Systems
- 6.4.16 66 Vision Tech Co., Ltd.
- 6.4.17 CSO Srl
- 6.4.18 Suzhou Kangjie Medical
- 6.4.19 MediWorks Optical
- 6.4.20 Diaton (BiCOM Inc.)
- 6.4.21 Suowei Electronic Technology
- 6.4.22 Optomed Plc
- 6.4.23 Bausch & Lomb Inc.
7 Market Opportunities & Future Outlook
- 7.1 White-space & Unmet-Need Assessment




