PUBLISHER: Mordor Intelligence | PRODUCT CODE: 2035078

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 2035078
Contract Research Organization - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts (2026 - 2031)
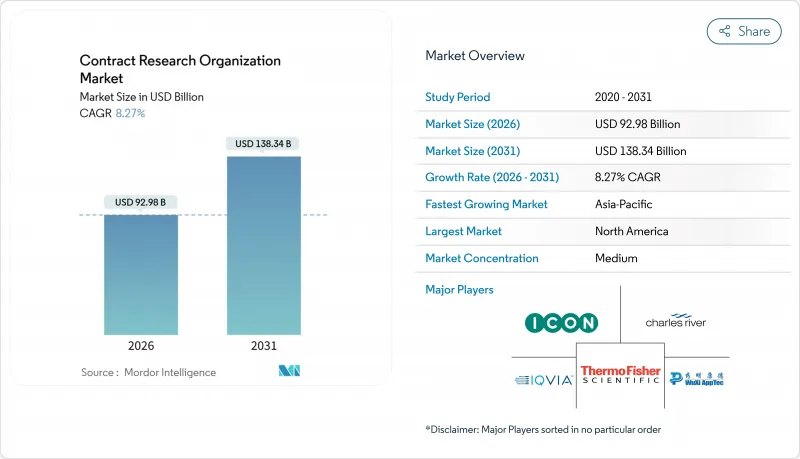
The Contract Research Organization Market size is estimated at USD 92.98 billion in 2026, and is expected to reach USD 138.34 billion by 2031, at a CAGR of 8.27% during the forecast period (2026-2031).
Revenue expansion is driven by sponsors' increasing need to compress development cycles, secure global patient access, and comply with increasingly complex regulatory pathways. Investment in biologics and cell- and gene-therapies is shifting trial portfolios toward high-complexity protocols that few sponsors can execute in-house. Regulatory agencies continue to award expedited designations, which shorten review times and further encourage the outsourcing of critical functions. Venture capital inflows into early-stage biotechs sustain demand for first-in-human expertise, while technology-enabled site selection tools shorten startup timelines and lower screening failure rates.
Global Contract Research Organization Market Trends and Insights
Rising Volume of Biologics and Advanced Therapies Development
Biologics and cell- and gene-therapy pipelines require viral-vector production, ex vivo cell manipulation, and long-term patient monitoring, which most sponsors lack internally. The FDA's Regenerative Medicine Advanced Therapy designation expedites the qualification of products, rewarding CROs that can manage adaptive designs and decentralized sample logistics. New CMC guidelines have reduced IND uncertainties, prompting a reallocation of capital from small-molecule portfolios toward biologics. As biosimilars pressure legacy revenues, sponsors lean on CRO partners to supply the highly specialized infrastructure needed for durable gene-editing trials.
Expansion of Emerging Market Patient Pools and Investigator Sites
China's multi-regional clinical trial framework now accepts foreign data, accelerating site initiation for multinational programs. India's Central Drugs Standard Control Organisation has shortened review cycles, drawing oncology and metabolic sponsors seeking treatment-naive cohorts. Brazil's ANVISA aligned with ICH E6(R2), reducing administrative friction and promoting Latin American enrollment. The demographic breadth in these regions offers rapid accrual and strengthens regulatory submissions through ethnically diverse datasets.
Intensifying Quality Audits and Compliance Penalties
The FDA inspects investigators, IRBs, and sponsors to enforce Good Clinical Practice, pushing CROs to allocate larger budgets to monitoring, electronic audit trails, and third-party verification. The EMA's Clinical Trials Information System increases transparency, thereby raising the reputational stakes. Smaller providers face margin pressure and consolidation risk as compliance costs climb.
Other drivers and restraints analyzed in the detailed report include:
- Accelerated Regulatory Pathways for Orphan and Fast-Track Drugs
- Growing Venture Capital Funding for Early-Stage Biotech Firms
- High Capital Expenditure for Cutting-Edge Lab Automation
For complete list of drivers and restraints, kindly check the Table Of Contents.
Segment Analysis
Early-phase development services, representing the fastest-growing segment of the Contract Research Organization market, are forecast to increase at a 10.72% annual rate through 2031. In value terms, this segment will account for a rising portion of the Contract Research Organization market size as biotech sponsors prioritize rapid proof-of-concept programs. Clinical Research Services remained dominant, with a 61.45% revenue share in 2025; however, heightened procurement scrutiny limits price escalation. Phase I units command premium rates because they have dedicated facilities, experienced medical monitors, and immediate access to academic centers, which mitigate the first-in-human risk. Phase II and Phase III work face commoditization as electronic data capture narrows differentiation. Laboratory Services grow steadily in response to the demand for precision medicine, while Consulting Services retain a niche appeal for complex regulatory strategies.
Sponsors are increasingly deploying biomarker-selected cohorts, which reduces enrollment numbers but increases analytical complexity. Phase IV surveillance expands modestly as agencies request post-approval safety evidence, yet many large sponsors internalize these studies to maintain control over real-world data. Differentiation shifts toward technology platforms, adaptive design expertise, and the seamless integration of decentralized trial components. This bifurcation keeps premium pricing in early-phase activities while mature services compete on operational scale.
Oncology generated the most considerable therapeutic-area revenue, at 21.43% in 2025, benefiting from over 1,000 active clinical assets that encompassed immunotherapies, targeted small molecules, and cell therapies. Infectious Diseases, however, exhibits the fastest expansion at 10.81% CAGR through 2031, reflecting pandemic-preparedness investment and mRNA vaccine platforms. Central Nervous System and Immunology each draw sizable spending, aided by the acceptance of digital biomarkers that reduce the need for subjective evaluations. Cardiovascular and Respiratory categories trail as generic erosion shifts R&D funding toward orphan conditions.
Government incentives for antimicrobial development, along with renewed vaccine pipelines, underpin infectious-disease momentum. Oncology's deceleration represents maturity rather than decline: commercialized checkpoint inhibitors transition activity from late-stage trials to post-marketing commitments. CNS programs still confront high screen-failure rates and lengthy follow-up periods, necessitating collaborations with CROs that include neurology specialists and robust investigator networks. Rare-disease identification remains a bottleneck, granting CROs with proprietary registries a competitive edge.
The Contract Research Organization Market Report is Segmented by Service Type (Early-Phase Development, and More), Therapeutic Area (Oncology, Infectious Diseases, and More), End User (Pharmaceutical & Biopharmaceutical, Medical Device, and Other), Delivery Model (Full-Service, FSP, and Hybrid), and Geography (North America, Europe, Asia-Pacific, MEA, and South America). Market Forecasts are Provided in Value (USD).
Geography Analysis
North America contributed 38.92% revenue in 2025, anchored by the United States' 350,000-site research infrastructure and the FDA's global regulatory influence. Growth lags the Contract Research Organization market average as sponsors diversify to contain costs and access varied patient pools. Canada and Mexico supply cardiovascular and diabetes cohorts through swift ethics approvals, while U.S. academic centers sustain complex oncology and gene-therapy protocols.
The Asia-Pacific region is poised for an 11.26% CAGR to 2031, the Contract Research Organization market's fastest regional trajectory, driven by regulatory modernization and vast treatment-naive populations. China's streamlined IND reviews and acceptance of foreign data under MRCT promote inclusion in global programs. India accelerates oncology enrollment via shortened review cycles, and Japan's ICH alignment eases multinational submissions. Australia leverages R&D tax offsets and rapid ethics reviews to attract first-in-human studies. South Korea invests heavily in cell-therapy infrastructure, leading to an increase in advanced-therapy trials.
Europe maintains mature Phase III capacity with harmonized approvals via the EMA's Clinical Trials Information System. Germany, the United Kingdom, and France remain core hubs, although Brexit now requires parallel UK protocols. Spain and Italy offer relative cost advantages and are capturing incremental respiratory and cardiovascular studies. The Middle East and Africa remain small but are receiving rare-disease and vaccine trials as Gulf states invest in research diversification. Meanwhile, South America, led by Brazil and Argentina, is gaining ground in infectious-disease research amid regulatory harmonization.
- BioAgile Therapeutics Private Limited
- Charles River
- CRITERIUM, INC.
- Evotec
- Eurofins
- Fortrea
- ICON
- IQVIA
- Inotiv
- MedPace
- Parexel International (MA) Corporation
- Pharmaron
- PSI
- SGS
- Syneos Health
- Syngene International
- Thermo Fisher Scientific
- Tigermed
- Worldwide Clinical Trials
- WuXi AppTec (WuXi Clinical)
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 Introduction
- 1.1 Study Assumptions & Market Definition
- 1.2 Scope Of The Study
2 Research Methodology
3 Executive Summary
4 Market Landscape
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Rising Volume of Biologics and Advanced Therapies Development
- 4.2.2 Expansion of Emerging Market Patient Pools and Investigator Sites
- 4.2.3 Accelerated Regulatory Pathways for Orphan and Fast-Track Drugs
- 4.2.4 Growing Venture Capital Funding for Early-Stage Biotech Firms
- 4.2.5 Adoption of Decentralized/Hybrid Clinical Trial Architectures
- 4.2.6 Integration of Real-World Evidence and Digital Biomarkers to Shorten Timelines
- 4.3 Market Restraints
- 4.3.1 Intensifying Quality Audits and Compliance Penalties
- 4.3.2 High Capital Expenditure for Cutting-Edge Lab Automation
- 4.3.3 Rising Geopolitical Risks Affecting Cross-Border Trials
- 4.3.4 Scarcity of GMP-Grade Viral Vector Manufacturing Capacity
- 4.4 Regulatory Outlook
- 4.5 Porter's Five Forces Analysis
- 4.5.1 Threat Of New Entrants
- 4.5.2 Bargaining Power Of Suppliers
- 4.5.3 Bargaining Power Of Buyers
- 4.5.4 Threat Of Substitutes
- 4.5.5 Industry Rivalry
5 Market Size & Growth Forecasts (Value, USD)
- 5.1 By Service Type
- 5.1.1 Early-Phase Development Services
- 5.1.2 Clinical Research Services
- 5.1.2.1 Phase I
- 5.1.2.2 Phase II
- 5.1.2.3 Phase III
- 5.1.2.4 Phase IV
- 5.1.3 Laboratory Services
- 5.1.4 Consulting Services
- 5.2 By Therapeutic Area
- 5.2.1 Oncology
- 5.2.2 Infectious Diseases
- 5.2.3 Central Nervous System (CNS) Disorders
- 5.2.4 Immunological Disorders
- 5.2.5 Cardiovascular Diseases
- 5.2.6 Respiratory Disorders
- 5.2.7 Diabetes
- 5.2.8 Other Therapeutic Areas
- 5.3 By End User
- 5.3.1 Pharmaceutical & Biopharmaceutical Companies
- 5.3.2 Medical Device Companies
- 5.3.3 Other End Users (Academic / Government Institutes)
- 5.4 By Delivery Model
- 5.4.1 Full-Service / Integrated CRO
- 5.4.2 Functional Service Provider (FSP)
- 5.4.3 Hybrid / Modular Model
- 5.5 Geography
- 5.5.1 North America
- 5.5.1.1 United States
- 5.5.1.2 Canada
- 5.5.1.3 Mexico
- 5.5.2 Europe
- 5.5.2.1 Germany
- 5.5.2.2 United Kingdom
- 5.5.2.3 France
- 5.5.2.4 Italy
- 5.5.2.5 Spain
- 5.5.2.6 Rest Of Europe
- 5.5.3 Asia-Pacific
- 5.5.3.1 China
- 5.5.3.2 Japan
- 5.5.3.3 India
- 5.5.3.4 Australia
- 5.5.3.5 South Korea
- 5.5.3.6 Rest Of Asia-Pacific
- 5.5.4 Middle East And Africa
- 5.5.4.1 GCC
- 5.5.4.2 South Africa
- 5.5.4.3 Rest Of Middle East And Africa
- 5.5.5 South America
- 5.5.5.1 Brazil
- 5.5.5.2 Argentina
- 5.5.5.3 Rest Of South America
- 5.5.1 North America
6 Competitive Landscape
- 6.1 Market Concentration
- 6.2 Market Share Analysis
- 6.3 Company Profiles {(Includes Global Level Overview, Market Level Overview, Core Segments, Financials as Available, Strategic Information, Market Rank/Share for Key Companies, Products & Services, and Recent Developments)}
- 6.3.1 BioAgile Therapeutics Private Limited
- 6.3.2 Charles River Laboratories
- 6.3.3 CRITERIUM, INC.
- 6.3.4 Evotec SE
- 6.3.5 Eurofins Scientific
- 6.3.6 Fortrea
- 6.3.7 ICON Plc
- 6.3.8 IQVIA Holdings Inc.
- 6.3.9 Inotiv
- 6.3.10 Medpace, Inc.
- 6.3.11 Parexel International (MA) Corporation
- 6.3.12 Pharmaron
- 6.3.13 PSI
- 6.3.14 SGS S.A.
- 6.3.15 Syneos Health
- 6.3.16 Syngene International Limited
- 6.3.17 Thermo Fisher Scientific Inc. (PPD Inc.)
- 6.3.18 Tigermed
- 6.3.19 Worldwide Clinical Trials
- 6.3.20 WuXi AppTec (WuXi Clinical)
7 Market Opportunities & Future Outlook
- 7.1 White-Space & Unmet-Need Assessment




