PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1851188

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1851188
Pneumococcal Vaccines - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts (2025 - 2030)
The pneumococcal vaccines market is valued at USD 9.23 billion in 2025 and is forecast to reach USD 11.68 billion by 2030, reflecting a 4.82% CAGR.
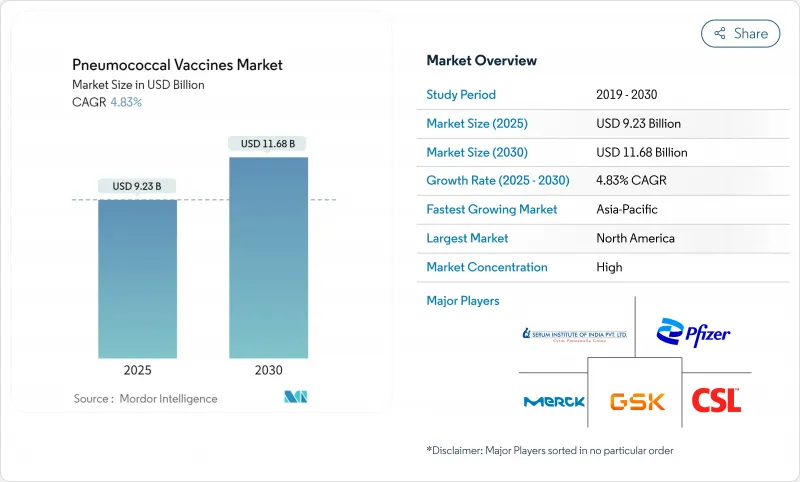
Continued shifts from 7-, 10- and 13-valent formulations toward 15-, 20- and 21-valent conjugate vaccines, broader adult vaccination guidelines, and expanded rollouts in emerging economies anchor this steady growth trajectory. North America remains the primary revenue center, yet Asia-Pacific shows the strongest momentum as domestic manufacturers scale up output and governments integrate pneumococcal conjugate vaccines into routine schedules. Intensifying competition follows Merck's adult-specific Capvaxive launch , the European approval of PCV21 and Vaxcyte's well-funded 31-valent pipeline . Meanwhile, GAVI-driven procurement, declining dose prices and evolving cold-chain technologies widen access in resource-constrained regions. The entrance of serotype-independent, protein-based candidates forms a long-term disruptive undercurrent that could reset product differentiation, manufacturing footprints and pricing strategies.
Global Pneumococcal Vaccines Market Trends and Insights
Increasing Government Awareness Programs on Pneumonia Immunization
Large-scale campaigns transform vaccination coverage by embedding pneumococcal conjugate vaccines into national schedules and securing multiyear procurement budgets. India's program now protects more than 90% of annual births, preventing an estimated 50,000 childhood deaths each year. Indonesia follows a similar path with 1.6 million doses financed through GAVI, underscoring the influence of public-private alliances on demand stability. The WHO Defeat Meningitis 2030 Roadmap aligns donor priorities toward at-risk displaced populations, while climate-driven disease spread compels portfolio reviews that favor thermostable presentations over traditional cold-chain-intensive formats. Persistent government advocacy, coupled with donor subsidies and tiered pricing, therefore sustains predictable order flow for producers across the pneumococcal vaccines market .
Rising Prevalence of Pneumococcal Infections
Despite decades of immunization progress, pneumococcal pneumonia and meningitis remain leading infectious killers of children under five and older adults. Outbreaks such as Togo's 2023 meningitis surge confirm lingering vulnerability in the Sub-Saharan meningitis belt. Adult coverage gaps are pronounced: only 13.4% of US adults aged 19-64 with chronic conditions are fully vaccinated, with state-level rates ranging from 0-34%. Hospitals in Hong Kong report less than one-third of eligible patients receiving a dose even after pneumococcal disease hospitalization. Alongside the emergence of antibiotic-resistant strains, these prevalence indicators reinforce the addressable burden underpinning growth in the pneumococcal vaccines market.
Lengthy Development & Regulatory Timelines
Prevnar 20's path to approval illustrates extended cycles: developers spent 1,434 days in testing phases and 244 days in formal review, stretching total time to 1,678 days. FDA requests for additional pediatric immunogenicity data on Vaxneuvance added further delays. As every additional serotype multiplies clinical workload, higher-valent programs face even longer and costlier pathways, which tempers near-term supply expansion in the pneumococcal vaccines market.
Other drivers and restraints analyzed in the detailed report include:
- Launch of Higher-Valent PCVs (PCV15/20/21)
- Adult Vaccination Expansion to the 50-64 Age Group (ACIP Draft)
- Serotype-Replacement Diminishing Long-Term Efficacy
For complete list of drivers and restraints, kindly check the Table Of Contents.
Segment Analysis
Pneumococcal conjugate vaccines held 65.25% revenue share in 2024, driven by evidence of superior immune memory and the ongoing shift to higher-valent PCV15, PCV20 and PCV21. This dominant position translates into a proportional contribution to the pneumococcal vaccines market. In contrast, polysaccharide offerings attract attention for adult catch-up campaigns and cost-sensitive tenders, producing a 5.25% CAGR that outpaces the total market. Conjugate innovators continue to push valency boundaries, while polysaccharide producers emphasize scale efficiencies to maintain competitiveness. Emerging protein-based concepts that target conserved lipoproteins such as MalX and PrsA could eventually blur current categorizations by offering serotype-agnostic protection. Early animal data showing cross-serotype efficacy drives investment interest.
The pneumococcal vaccines market size for conjugate formulations is projected to climb in absolute terms even as polysaccharides accelerate. Conjugate value growth is underpinned by premium pricing, diverse tender volumes and uptake in high-income adult populations. Meanwhile, polysaccharide expansion is volume-led, with governments procuring cost-effective doses to close coverage gaps in older adults. Both approaches remain complementary, and many national guidelines recommend sequential administration of conjugate followed by polysaccharide strains to maximize serotype breadth and immune response durability.
Prevnar 13 captured 42.22% share of the pneumococcal vaccines market in 2024 thanks to long-standing clinical data, consistent supply and global regulatory recognition. Its successor, Prevnar 20, extends protection but still competes head-to-head with Merck's Capvaxive in adults and with Synflorix in low-income pediatric segments. Pneumovax 23, although an older polysaccharide product, benefits from strong physician familiarity and a lower dose price that attracts tender wins in middle-income economies, which explains its 5.48% CAGR. The "Others" cluster-including Capvaxive and potential 24- and 31-valent entrants-adds diversity and is likely to claim incremental share post-2027.
Multiple antigen presenting system candidates and synthetic glycoconjugate pipelines position newer firms to erode incumbent dominance over time. Yet brand equity, cold-chain networks and post-marketing safety surveillance infrastructure still reinforce incumbents' negotiating leverage during national procurements. Future competitive positioning will hinge on breadth of coverage, targeted adult or pediatric indications and total cost per fully immunized person rather than per-dose sticker price.
The Pneumococcal Vaccines Market is Segmented by Vaccine Type (Pneumococcal Conjugate Vaccine (PCV) and More), Product Type (Prevnar 13, Synflorix, and More), Distribution Channel (Government Authorities, Non-Government NGOs & Multilaterals, and More), Age Group (Adults, Geriatric, and More) and Geography (North America, Europe, Asia-Pacific, and More). The Market and Forecasts are Provided in Terms of Value (USD).
Geography Analysis
North America contributed 40.56% revenue in 2024 and continues to benefit from insurance coverage, pharmacist vaccination authority and corporate wellness uptake. The CDC age-threshold change instantly enlarged the eligible pool, and Capvaxive's adult-specific positioning drives brand-switching campaigns. Provincial programs in Canada align with US guidelines, and Mexico taps Pan American Health Organization revolving funds to co-finance PCV20 for adults at risk. Nevertheless, socio-economic disparities persist, with rural states recording adult coverage rates below 45%.
Asia-Pacific is projected to post a 5.65% CAGR to 2030, the fastest among all regions. India's nationwide roll-out supplies more than 30 million doses annually, powered by domestic low-cost production under the PNEUMOSIL brand. China's expansion of combination vaccine reimbursement, plus Singapore's pioneering PCV20 uptake, confirm rising middle-class demand for premium conjugates. Indonesia's 4.5 million-child target illustrates momentum among populous Association of Southeast Asian Nations (ASEAN) members. Local strain profiling influences product strategies, with region-specific serotypes like 1, 5 and 10A shaping national tenders.
Europe maintains moderate single-digit growth as the European Commission's May 2025 approval of Capvaxive boosts adult coverage campaigns across Germany, Italy and France. Widespread surveillance capacity helps policymakers refine schedules promptly when replacement trends emerge. In the Middle East and Africa, GAVI funding remains the principal catalyst, complemented by innovative solar cold-chain projects such as South Sudan's withstanding 40 °C daytime temperatures. Latin America leverages Brazil's long-standing PCV10 evidence base, showing efficacy across income quartiles and informing tender renewals throughout the region.
- Pfizer
- GlaxoSmithKline
- Merck
- Sanofi
- Serum Institute of India
- CSL Ltd. (Seqirus)
- Walvax Biotechnology Co., Ltd.
- Beijing Minhai Biotechnology Co., Ltd.
- Vaxcyte, Inc.
- Panacea Biotec Ltd.
- SK bioscience Co., Ltd.
- Biological E. Limited
- Bharat Biotech International Ltd.
- KM Biologics Co., Ltd.
- VBI Vaccines Inc.
- Aspen Pharmacare Holdings Ltd.
- Astellas Pharma
- Zhejiang Pukang Biotechnology Co., Ltd.
- Inventprise LLC
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 Introduction
- 1.1 Study Assumptions & Market Definition
- 1.2 Scope of the Study
2 Research Methodology
3 Executive Summary
4 Market Landscape
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Increasing government awareness programs on pneumonia immunization
- 4.2.2 Rising prevalence of pneumococcal infections
- 4.2.3 Launch of higher-valent PCVs (PCV15/20/21)
- 4.2.4 Adult vaccination expansion to 50-64 age group (ACIP draft)
- 4.2.5 Low-cost India-made PCVs (e.g., Pneumosil) boosting GAVI uptake
- 4.2.6 Rapidly ageing global population
- 4.3 Market Restraints
- 4.3.1 Lengthy development & regulatory timelines
- 4.3.2 High manufacturing cost of conjugate vaccines
- 4.3.3 Serotype-replacement diminishing long-term efficacy
- 4.3.4 Cold chain and supply chain issues
- 4.4 Regulatory Landscape
- 4.5 Porters Five Forces Analysis
- 4.5.1 Threat of New Entrants
- 4.5.2 Bargaining Power of Suppliers
- 4.5.3 Bargaining Power of Buyers/Consumers
- 4.5.4 Threat of Substitutes
- 4.5.5 Intensity of Competitive Rivalry
5 Market Size & Growth Forecasts (Value, USD)
- 5.1 By Vaccine Type
- 5.1.1 Pneumococcal Conjugate Vaccine (PCV)
- 5.1.2 Pneumococcal Polysaccharide Vaccine (PPV)
- 5.2 By Product Type
- 5.2.1 Prevnar 13
- 5.2.2 Synflorix
- 5.2.3 Pneumovax 23
- 5.2.4 Others
- 5.3 By Distribution Channel
- 5.3.1 Government Authorities
- 5.3.2 Non-Government NGOs & Multilaterals
- 5.3.3 Distribution-partner Companies
- 5.4 By Age Group
- 5.4.1 Adults
- 5.4.2 Geriatric
- 5.4.3 Pediatric
- 5.5 By Geography
- 5.5.1 North America
- 5.5.1.1 United States
- 5.5.1.2 Canada
- 5.5.1.3 Mexico
- 5.5.2 Europe
- 5.5.2.1 Germany
- 5.5.2.2 United Kingdom
- 5.5.2.3 France
- 5.5.2.4 Italy
- 5.5.2.5 Spain
- 5.5.2.6 Rest of Europe
- 5.5.3 Asia-Pacific
- 5.5.3.1 China
- 5.5.3.2 Japan
- 5.5.3.3 India
- 5.5.3.4 Australia
- 5.5.3.5 South Korea
- 5.5.3.6 Rest of Asia-Pacific
- 5.5.4 Middle East and Africa
- 5.5.4.1 GCC
- 5.5.4.2 South Africa
- 5.5.4.3 Rest of Middle East and Africa
- 5.5.5 South America
- 5.5.5.1 Brazil
- 5.5.5.2 Argentina
- 5.5.5.3 Rest of South America
- 5.5.1 North America
6 Competitive Landscape
- 6.1 Market Concentration
- 6.2 Market Share Analysis
- 6.3 Company Profiles (includes Global level Overview, Market level overview, Core Segments, Financials as available, Strategic Information, Market Rank/Share for key companies, Products & Services, and Recent Developments)
- 6.3.1 Pfizer Inc.
- 6.3.2 GSK plc
- 6.3.3 Merck & Co., Inc.
- 6.3.4 Sanofi S.A.
- 6.3.5 Serum Institute of India Pvt. Ltd.
- 6.3.6 CSL Ltd. (Seqirus)
- 6.3.7 Walvax Biotechnology Co., Ltd.
- 6.3.8 Beijing Minhai Biotechnology Co., Ltd.
- 6.3.9 Vaxcyte, Inc.
- 6.3.10 Panacea Biotec Ltd.
- 6.3.11 SK bioscience Co., Ltd.
- 6.3.12 Biological E. Limited
- 6.3.13 Bharat Biotech International Ltd.
- 6.3.14 KM Biologics Co., Ltd.
- 6.3.15 VBI Vaccines Inc.
- 6.3.16 Aspen Pharmacare Holdings Ltd.
- 6.3.17 Astellas Pharma Inc.
- 6.3.18 Zhejiang Pukang Biotechnology Co., Ltd.
- 6.3.19 Inventprise LLC
7 Market Opportunities & Future Outlook
- 7.1 White-space & Unmet-need Assessment




