PUBLISHER: Roots Analysis | PRODUCT CODE: 2011951

PUBLISHER: Roots Analysis | PRODUCT CODE: 2011951
Japan Point-of-care Diagnostics Market - Distribution by Target Disease Indication and Type of Technology: Industry Trends and Forecasts, till 2035
Japan Point-of-Care Diagnostics Market: Overview
As per Roots Analysis, the Japan point-of-care diagnostics market is estimated to grow from USD 1.39 billion in the current year to USD 2.62 billion by 2035 at a CAGR of 7.0% during the forecast period, till 2035.
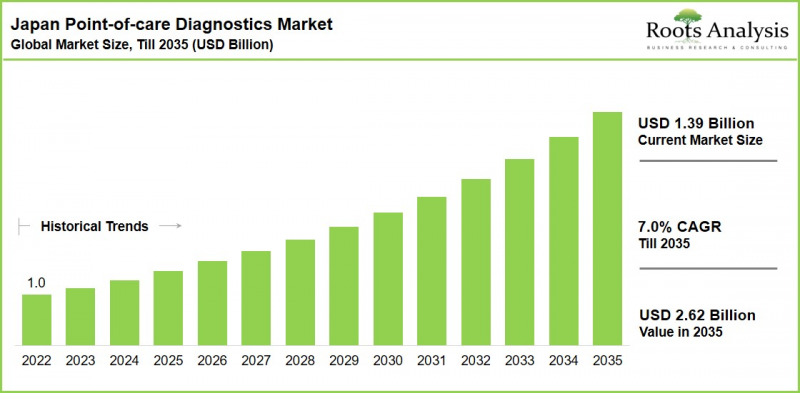
Japan Point-of-Care Diagnostics Market: Growth and Trends
Point-of-care (POC) diagnostics have emerged as a critical component of modern healthcare systems globally, driven by the increasing need for rapid diagnostics, decentralized healthcare delivery, and improved patient outcomes. The Japan point-of-care (POC) diagnostics market represents a key segment within the in vitro diagnostics landscape, enabling rapid, near-patient testing across diverse care settings, including clinics, hospitals, pharmacies, emergency departments, and home environments. By delivering immediate results without reliance on centralized laboratory infrastructure, POC diagnostics support timely clinical decision-making. This capability is critical for the effective management of infectious diseases and chronic conditions, such as diabetes and cardiovascular disorders, while also enhancing operational efficiency and overall patient care within Japan's advanced healthcare system.
Japan's point-of-care (POC) diagnostics market is distinctly positioned, driven by its super-aging demographic profile, with over 29% of the population aged 65 and above. This demographic trend is accelerating demand for convenient, decentralized diagnostic solutions to effectively monitor and manage chronic conditions prevalent among the elderly population. Furthermore, the country's technological leadership in microfluidics, biosensors, artificial intelligence (AI) integration, and portable diagnostic devices, combined with a strong policy focus on preventive and home-based care, is supporting robust market adoption.
The Japan POC diagnostics market also presents significant growth opportunities through the integration of digital health solutions aimed at extending healthy life expectancy. POC devices are increasingly being integrated with digital platforms, wearable technologies, telemedicine infrastructure, and AI-driven analytics to enable real-time monitoring, seamless remote data exchange, and personalized care interventions. This convergence is expected to play a critical role in enhancing healthy longevity and addressing the growing burden of chronic diseases.
Post-COVID relaxations, investments in digital health infrastructure, and promotion of community-based integrated care have encouraged POC adoption, aligning with goals for efficient, accessible healthcare in an aging nation. The market remains competitive, with global and domestic players innovating in multiplex, connected, and user-friendly solutions to meet Japan's demographic and policy-driven needs.
Growth Drivers: Strategic Enablers of Market Expansion
The Japan point-of-care (POC) diagnostics market is powerfully driven by the country's unprecedented aging population, which creates urgent demand for rapid, accessible testing solutions tailored to chronic disease management. Japan possesses the world's largest geriatric demographic, with 36.2 million people aged 65 and older, accounting for a record 29.1% of the total population. This demographic shift, fueled by extended life expectancy and low fertility rates, has resulted in increased prevalence of conditions such as diabetes, cardiovascular disorders, and cancer among older adults. Point-of-care (POC) devices facilitate real-time monitoring and informed clinical decision-making in decentralized settings, including community clinics, pharmacies, and home environments. This capability reduces the need for hospital visits and alleviates pressure on centralized laboratory infrastructure. Additionally, it enables timely interventions, thereby enhancing patient outcomes and overall quality of life.
Market Challenges: Critical Barriers Impeding Progress
Despite the advantages, the Japan point-of-care market faces considerable obstacles that could hinder growth. Stringent regulatory oversight remains a primary obstacle to faster market expansion. It is important to note that all point-of-care (POC) diagnostic devices are subject to stringent evaluation by the Pharmaceuticals and Medical Devices Agency and require formal approval from the Ministry of Health, Labour and Welfare. These regulatory processes are complex, resource-intensive, and often associated with extended timelines. Compliance with high standards for safety, efficacy, and quality assurance, while essential for patient protection, frequently delays product launches and increases development expenses for both domestic and international manufacturers. Further, economic pressures compound these hurdles. In addition, the high cost of advanced POC equipment featuring advanced biosensors, multiplex capabilities, and digital connectivity limits affordability and penetration, particularly outside major urban centers. Together, these factors require manufacturers to focus on cost-optimization, evidence-based value demonstration, and strategic partnerships to achieve broader scalability and sustained growth.
Hepatitis Holds the Largest Market Share
In terms of various target indications, the market is divided into chikungunya, COVID-19, dengue, Ebola, hepatitis, HIV/AIDS, influenza, malaria, pneumonia, tuberculosis, and zika. At present, Notably, hepatitis segment captures the majority of the market share, and this trend is unlikely to change in the future as well. Further, the market for Ebola virus will grow at a higher CAGR owing to the rising incidence of infectious diseases and the need for rapid testing during outbreaks.
Lateral Flow Assay Dominates the Market Growth
In terms of type of technology, the market is segmented into lateral flow, molecular diagnostics, fluorescence immunoassay, solid phase, and others. Our projection indicates that lateral flow assay is likely to constitute the majority of the market share in the current year. The point of care market for molecular diagnostics is expected to increase at a notable CAGR indicating greater demand and acceptance of these devices due to their capacity to provide quick results, reduced costs, and improved outcomes.
Japan Point-of-Care Diagnostics Market: Key Segments
By Target Disease Indication
- Chikungunya
- COVID-19
- Dengue
- Ebola
- Hepatitis
- HIV/AIDS
- Influenza
- Malaria
- Pneumonia
- Tuberculosis
- Zika
By Type of Technology
- Lateral Flow Assay
- Molecular Diagnostics
- Fluorescence Immunoassay
- Solid Phase Assay
- Other Technologies
Example Players in Japan Point-of-care Diagnostics Market
- Abbott Laboratories
- Alere (now part of Abbott)
- Bayer
- Becton, Dickinson and Company (BD)
- Beckman Coulter (part of Danaher Corporation)
- Bio-Rad Laboratories
- BioMerieux
- Cepheid
- Danaher
- F. Hoffmann-La Roche Ltd. (Roche Diagnostics)
- Fujifilm Holdings
- Fujirebio Holdings
- Hologic
- Nihon Kohden
- Nipro
- Nova Biomedical
- Ortho Clinical Diagnostics
- QIAGEN N.V.
- Quidel Corporation (now part of QuidelOrtho)
- Sekisui Medical
- Siemens Healthineers
- Sysmex
- TAUNS Laboratories
- Thermo Fisher Scientific
Key Questions Answered in this Report
- How many Japan point-of-care diagnostics providers are currently engaged in this market?
- Which are the leading companies in this market?
- Which country dominates the Japan point-of-care market?
- What are the key trends observed in the Japan point-of-care market?
- What factors are likely to influence the evolution of this market?
- What are the primary challenges faced by Japan point-of-care solution providers?
- What is the current and future Japan point-of-care market size?
- What is the CAGR of Japan point-of-care market?
- How is the current and future market opportunity likely to be distributed across key market segments?
Reasons to Buy this Report
- The report provides a comprehensive market analysis, offering detailed revenue projections of the overall market and its specific sub-segments. This information is valuable to both established market leaders and emerging entrants.
- The report offers stakeholders a comprehensive overview of the market, including key drivers, barriers, opportunities, and challenges. This information empowers stakeholders to stay abreast of market trends and make data-driven decisions to capitalize on growth prospects.
- The report can aid businesses in identifying future opportunities in any sector. It also helps in understanding if those opportunities are worth pursuing.
- The report helps in identifying customer demand by understanding the needs, preferences, and behavior of the target audience in order to tailor products or services effectively.
- The report equips new entrants with requisite information regarding a particular market to help them build successful business strategies.
- The report allows for more effective communication with the audience and in building strong business relations.
Complementary Benefits
- Complimentary PPT Insights Packs
- Complimentary Excel Data Packs for all Analytical Modules in the Report
- 15% Free Content Customization
- Detailed Report Walkthrough Session with Research Team
- Free Updated report if the report is 6-12 months old or older
TABLE OF CONTENTS
1. PREFACE
- 1.1. Introduction
- 1.2. Market Share Insights
- 1.3. Key Market Insights
- 1.4. Report Coverage
- 1.5. Key Questions Answered
- 1.6. Chapter Outlines
2. RESEARCH METHODOLOGY
- 2.1. Chapter Overview
- 2.2. Research Assumptions
- 2.2.1. Market Landscape and Market Trends
- 2.2.2. Market Forecast and Opportunity Analysis
- 2.2.3. Comparative Analysis
- 2.3. Database Building
- 2.3.1. Data Collection
- 2.3.2. Data Validation
- 2.3.3. Data Analysis
- 2.4. Project Methodology
- 2.4.1. Secondary Research
- 2.4.1.1. Annual Reports
- 2.4.1.2. Academic Research Papers
- 2.4.1.3. Company Websites
- 2.4.1.4. Investor Presentations
- 2.4.1.5. Regulatory Filings
- 2.4.1.6. White Papers
- 2.4.1.7. Industry Publications
- 2.4.1.8. Conferences and Seminars
- 2.4.1.9. Government Portals
- 2.4.1.10. Media and Press Releases
- 2.4.1.11. Newsletters
- 2.4.1.12. Industry Databases
- 2.4.1.13. Roots Proprietary Databases
- 2.4.1.14. Paid Databases and Sources
- 2.4.1.15. Social Media Portals
- 2.4.1.16. Other Secondary Sources
- 2.4.2. Primary Research
- 2.4.2.1. Types of Primary Research
- 2.4.2.1.1. Qualitative Research
- 2.4.2.1.2. Quantitative Research
- 2.4.2.1.3. Hybrid Approach
- 2.4.2.2. Advantages of Primary Research
- 2.4.2.3. Techniques for Primary Research
- 2.4.2.3.1. Interviews
- 2.4.2.3.2. Surveys
- 2.4.2.3.3. Focus Groups
- 2.4.2.3.4. Observational Research
- 2.4.2.3.5. Social Media Interactions
- 2.4.2.4. Key Opinion Leaders Considered in Primary Research
- 2.4.2.4.1. Company Executives (CXOs)
- 2.4.2.4.2. Board of Directors
- 2.4.2.4.3. Company Presidents and Vice Presidents
- 2.4.2.4.4. Research and Development Heads
- 2.4.2.4.5. Technical Experts
- 2.4.2.4.6. Subject Matter Experts
- 2.4.2.4.7. Scientists
- 2.4.2.4.8. Doctors and Other Healthcare Providers
- 2.4.2.5. Ethics and Integrity
- 2.4.2.5.1. Research Ethics
- 2.4.2.5.2. Data Integrity
- 2.4.2.1. Types of Primary Research
- 2.4.3. Analytical Tools and Databases
- 2.4.1. Secondary Research
- 2.5. Robust Quality Control
3. MARKET DYNAMICS
- 3.1. Chapter Overview
- 3.2. Forecast Methodology
- 3.2.1. Top-down Approach
- 3.2.2. Bottom-up Approach
- 3.2.3. Hybrid Approach
- 3.3. Market Assessment Framework
- 3.3.1. Total Addressable Market (TAM)
- 3.3.2. Serviceable Addressable Market (SAM)
- 3.3.3. Serviceable Obtainable Market (SOM)
- 3.3.4. Currently Acquired Market (CAM)
- 3.4. Forecasting Tools and Techniques
- 3.4.1. Qualitative Forecasting
- 3.4.2. Correlation
- 3.4.3. Regression
- 3.4.4. Extrapolation
- 3.4.5. Convergence
- 3.4.6. Sensitivity Analysis
- 3.4.7. Scenario Planning
- 3.4.8. Data Visualization
- 3.4.9. Time Series Analysis
- 3.4.10. Forecast Error Analysis
- 3.5. Key Considerations
- 3.5.1. Demographics
- 3.5.2. Government Regulations
- 3.5.3. Reimbursement Scenarios
- 3.5.4. Market Access
- 3.5.5. Supply Chain
- 3.5.6. Industry Consolidation
- 3.5.7. Pandemic / Unforeseen Disruptions Impact
- 3.6. Limitations
4. MACRO-ECONOMIC INDICATORS
- 4.1. Chapter Overview
- 4.2. Market Dynamics
- 4.2.1. Time Period
- 4.2.1.1. Historical Trends
- 4.2.1.2. Current and Forecasted Estimates
- 4.2.2. Currency Coverage
- 4.2.2.1. Major Currencies Affecting the Market
- 4.2.2.2. Factors Affecting Currency Fluctuations on the Industry
- 4.2.2.3. Impact of Currency Fluctuations on the Industry
- 4.2.3. Foreign Currency Exchange Rate
- 4.2.3.1. Impact of Foreign Exchange Rate Volatility on the Market
- 4.2.3.2. Strategies for Mitigating Foreign Exchange Risk
- 4.2.4. Recession
- 4.2.4.1. Assessment of Current Economic Conditions and Potential Impact on the Market
- 4.2.4.2. Historical Analysis of Past Recessions and Lessons Learnt
- 4.2.5. Inflation
- 4.2.5.1. Measurement and Analysis of Inflationary Pressures in the Economy
- 4.2.5.2. Potential Impact of Inflation on the Market Evolution
- 4.2.6. Interest Rates
- 4.2.6.1. Interest Rates and Their Impact on the Market
- 4.2.6.2. Strategies for Managing Interest Rate Risk
- 4.2.7. Commodity Flow Analysis
- 4.2.7.1. Type of Commodity
- 4.2.7.2. Origins and Destinations
- 4.2.7.3. Value and Weights
- 4.2.7.4. Modes of Transportation
- 4.2.8. Global Trade Dynamics
- 4.2.8.1. Import Scenario
- 4.2.8.2. Export Scenario
- 4.2.8.3. Trade Policies
- 4.2.8.4. Strategies for Mitigating the Risks Associated with Trade Barriers
- 4.2.8.5. Impact of Trade Barriers on the Market
- 4.2.9. War Impact Analysis
- 4.2.9.1. Russian-Ukraine War
- 4.2.9.2. Israel-Hamas War
- 4.2.10. COVID Impact / Related Factors
- 4.2.10.1. Global Economic Impact
- 4.2.10.2. Industry-specific Impact
- 4.2.10.3. Government Response and Stimulus Measures
- 4.2.10.4. Future Outlook and Adaptation Strategies
- 4.2.11. Other Indicators
- 4.2.11.1. Fiscal Policy
- 4.2.11.2. Consumer Spending
- 4.2.11.3. Gross Domestic Product (GDP)
- 4.2.11.4. Employment
- 4.2.11.5. Taxes
- 4.2.11.6. Stock Market Performance
- 4.2.11.7. Cross-Border Dynamics
- 4.2.1. Time Period
- 4.3. Conclusion
5. EXECUTIVE SUMMARY
6. INTRODUCTION
- 6.1. Chapter Overview
- 6.2. Overview of Point-of-Care Diagnostics (POCDs)
- 6.2.1. Characteristics of an Ideal Point-of-Care Diagnostic
- 6.2.2. Technologies Used in Point-of-Care Diagnostics of Infectious Diseases
- 6.3. Challenges Associated with Point-of-Care Diagnostics
- 6.4. Future Perspectives
7. MARKET LANDSCAPE
- 7.1. Chapter Overview
- 7.2. Point-of-care Diagnostics: Overall Developers Landscape
- 7.2.1. Analysis by Year of Establishment
- 7.2.2. Analysis by Company Size
- 7.2.3. Analysis by Location of Headquarters
- 7.2.4. Point-of-care Diagnostics: Overall Market Landscape
- 7.2.4.1. Analysis by Type of Product
- 7.2.4.2. Analysis by Type of Technology
- 7.2.4.3. Analysis by Type of Sample
- 7.2.4.4. Analysis by Turnaround Time
- 7.2.4.5. Analysis by Sensitivity Range
8. PARTNERSHIPS AND COLLABORATIONS
- 8.1. Japan Point-of-care Diagnostics: Market Landscape of Startups
- 8.1.1. Analysis by Year of Partnership
- 8.1.2. Analysis by Type of Partnership
- 8.1.3. Analysis by Type of Product
- 8.1.4. Analysis by Target Disease Indication
- 8.1.5. Most Active Players: Analysis by Number of Partnerships
- 8.1.6. Regional Analysis
9. COMPANY PROFILES: JAPAN POINT-OF-CARE DIAGNOSTICS MARKET
- 9.1. Chapter Overview
- 9.2. Abbott Laboratories
- 9.2.1. Company Overview
- 9.2.2. Product Portfolio
- 9.2.3. Financial Information
- 9.2.4. Recent Developments and Future Outlook
- 9.3. Alere
- 9.4. Bayer
- 9.5. Becton, Dickinson and Company (BD)
- 9.6. Beckman Coulter
- 9.7. Bio-Rad Laboratories
- 9.8. BioMerieux
- 9.9. Cepheid
- 9.10. Danaher
- 9.11. F. Hoffmann-La Roche
10. MARKET IMPACT ANALYSIS
- 10.1. Chapter Overview
- 10.2. Market Drivers
- 10.3. Market Restraints
- 10.4. Market Opportunities
- 10.5. Market Challenges
- 10.6. Conclusion
11. JAPAN POINT-OF-CARE DIAGNOSTICS MARKET
- 11.1. Chapter Overview
- 11.2. Key Assumptions and Methodology
- 11.3. Japan Point-of-care Diagnostics Market, Historical Trends (Since 2022) and Forecasted Estimates (Till 2035)
- 11.4. Roots Analysis Perspective on Market Growth
- 11.5 Scenario Analysis
- 11.5.1. Conservative Scenario
- 11.5.2. Optimistic Scenario
- 11.6. Key Market Segmentations
12. JAPAN POINT-OF-CARE DIAGNOSTICS MARKET, TYPE OF TARGET DISEASE INDICATION
- 12.1. Chapter Overview
- 12.2. Key Assumptions and Methodology
- 12.3. Japan Point-of-care Diagnostics Market: Distribution by Target Disease Indication
- 12.3.1. Japan Point-of-care Diagnostics Market for Chikungunya: Historical Trends (Since 2020) and Forecasted Estimates (Till 2035)
- 12.3.2. Japan Point-of-care Diagnostics Market for Dengue: Historical Trends (Since 2020) and Forecasted Estimates (Till 2035)
- 12.3.3. Japan Point-of-care Diagnostics Market for Ebola: Historical Trends (Since 2020) and Forecasted Estimates (Till 2035)
- 12.3.4. Japan Point-of-care Diagnostics Market for Hepatitis: Historical Trends (Since 2020) and Forecasted Estimates (Till 2035)
- 12.3.5. Japan Point-of-care Diagnostics Market for HIV/AIDSs: Historical Trends (Since 2020) and Forecasted Estimates (Till 2035)
- 12.3.6. Japan Point-of-care Diagnostics Market for Influenza: Historical Trends (Since 2020) and Forecasted Estimates (Till 2035)
- 12.3.7. Japan Point-of-care Diagnostics Market for Malaria: Historical Trends (Since 2020) and Forecasted Estimates (Till 2035)
- 12.3.8. Japan Point-of-care Diagnostics Market for Pneumonia: Historical Trends (Since 2020) and Forecasted Estimates (Till 2035)
- 12.3.9. Japan Point-of-care Diagnostics Market for Malaria: Historical Trends (Since 2020) and Forecasted Estimates (Till 2035)
- 12.3.10. Japan Point-of-care Diagnostics Market for Tuberculosis: Historical Trends (Since 2020) and Forecasted Estimates (Till 2035)
- 12.3.11. Japan Point-of-care Diagnostics Market for Zika: Historical Trends (Since 2020) and Forecasted Estimates (Till 2035)
- 12.4. Data Triangulation and Validation
- 12.4.1. Secondary Sources
- 12.4.2. Primary Sources
- 12.4.3. Statistical Modeling
13. JAPAN POINT-OF-CARE DIAGNOSTICS MARKET, TYPE OF TECHNOLOGY
- 13.1. Chapter Overview
- 13.2. Key Assumptions and Methodology
- 13.3. Japan Point-of-care Diagnostics Market: Distribution by Type of Technology
- 13.3.1. Japan Point-of-care Diagnostics Market for Lateral Flow Assay: Historical Trends (Since 2020) and Forecasted Estimates (Till 2035)
- 13.3.2. Japan Point-of-care Diagnostics Market for Molecular Diagnostics: Historical Trends (Since 2020) and Forecasted Estimates (Till 2035)
- 13.3.3. Japan Point-of-care Diagnostics Market for Fluorescence Immunoassay: Historical Trends (Since 2020) and Forecasted Estimates (Till 2035)
- 13.3.4. Japan Point-of-care Diagnostics Market for Solid Phase Assay: Historical Trends (Since 2020) and Forecasted Estimates (Till 2035)
- 13.3.5. Japan Point-of-care Diagnostics Market for Other Technologies: Historical Trends (Since 2020) and Forecasted Estimates (Till 2035)
- 13.4. Data Triangulation and Validation
- 13.4.1. Secondary Sources
- 13.4.2. Primary Sources
- 13.4.3. Statistical Modeling
14. CONCLUDING REMARKS
15. APPENDIX I: TABULATED DATA
16. APPENDIX II: LIST OF COMPANIES AND ORGANIZATIONS




