PUBLISHER: SNS Research | PRODUCT CODE: 1994827

PUBLISHER: SNS Research | PRODUCT CODE: 1994827
Psoriasis Drugs Market, 2025 - 2035: Opportunities, Challenges, Strategies & Forecasts
Synopsis:
Psoriasis is a chronic inflammatory skin disorder affecting more than 125 million people globally, including over 8 million in the United States. Characterized by thick, reddened, scaly plaques that cause itching and pain, the disease imposes substantial physical, psychosocial, and economic burdens across all severity levels. Growing public awareness and rising expectations around skin, hair, and nail appearance have increased the number of patients seeking more effective and durable treatment options.
Disease management is lifelong and focuses on achieving remission by slowing excessive skin-cell turnover and reducing inflammation. Treatment selection depends on disease type, body-surface involvement, and overall severity. Mild disease is typically managed with topical therapies, whereas moderate-to-severe psoriasis often requires oral systemics, biologics, or combination approaches. Over the past decade, psoriasis treatment has shifted from broad immunosuppression to highly selective therapies. Established biologics (TNF, IL-12/23, IL-17, and IL-23 inhibitors) have been complemented by oral small molecules (notably TYK2 inhibitors) and next-generation biologics. These innovations are achieving unprecedented complete skin clearance rates (PASI 100), extended duration of response, and improved convenience and safety profiles. As a result, they are driving stronger market growth, expanding the treatable patient population, and significantly increasing payer and physician expectations for long-term disease modification in psoriasis.
SNS Research estimates that the psoriasis drugs market will account for nearly $20 Billion in revenue by the end of 2025. Despite challenges relating to drug stability and patent disputes amongst competition, the market is poised for significant growth over the forecast period, fueled by several novel agents advancing through late-stage development.
The "Psoriasis Drugs Market: 2025 - 2035 - Opportunities, Challenges, Strategies & Forecasts" report presents an in-depth assessment of the psoriasis drugs ecosystem including psoriasis disorders, types of treatment options, delivery technologies, key trends, market drivers, challenges, investment potential, leading therapies, drug development pipeline, opportunities, future roadmap, value chain, ecosystem player profiles and strategies. The report also presents market size forecasts for psoriasis drugs from 2025 through to 2035. The forecasts are further segmented by 4 therapeutic classes, 3 routes of administration, 4 distribution channels, 5 regions and 26 leading countries.
Topics Covered:
The report covers the following topics:
- Psoriasis drugs ecosystem
- Market drivers and barriers
- Psoriasis disorders and key trends
- Analysis of key drug classes and leading psoriasis drugs
- Future drug development pipeline
- Psoriasis drug delivery technologies
- Industry roadmap and value chain
- Profiles and strategies of 66 leading ecosystem players, including psoriasis drug developers
- Strategic recommendations for ecosystem players
- Market analysis and forecasts from 2025 till 2035
Sample Figure: Psoriasis Drugs Development Pipeline Breakdown by Therapeutic Area (%)
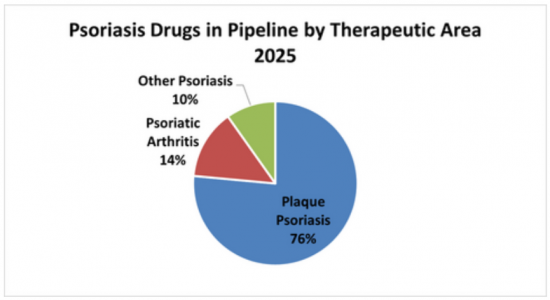
The report comes with an associated Excel datasheet suite covering quantitative data from all numeric forecasts presented in the report, as well as an Excel sheet covering clinical drug development pipeline for the psoriasis drugs market, profiling 51 future candidates.
Key Findings:
The report has the following key findings:
- Global psoriasis drug spending is projected to reach $19.6 billion by the end of 2025, driven by rising disease prevalence, increased diagnosis rates, and expanding access to advanced therapies. Given the chronic, relapsing nature of psoriasis, long-term treatment demand remains stable, supporting steady market growth through 2030 and beyond.
- Biologics remain the backbone of the psoriasis therapeutics market, with IL-17 and IL-23 inhibitors, particularly Skyrizi (risankizumab) and Tremfya (guselkumab), continuing to gain adoption due to superior efficacy and durable response profiles.
- In the U.S., biosimilar penetration remains limited. Interchangeability barriers, litigation timelines, and contracting strategies have slowed the real-world uptake of several FDA-approved biosimilars, sustaining premium biologic pricing and delaying broader cost competition.
- Pipeline activity is increasingly concentrated on next-generation immunomodulators, with strategic emphasis shifting away from legacy TNF and IL-12/23 programs and toward newer, higher-yield mechanisms. Development efforts center on next-generation IL-23 agents, selective IL-17 pathway modulators, and especially oral TYK2 inhibitors, which remain a major commercial focus due to strong patient preference for non-injectable therapies. TNF inhibitors retain clinical utility in psoriatic arthritis and long-term stable disease, but they are no longer a primary R&D growth engine.
- Beyond biologics, innovation is accelerating in oral and topical modalities. Active programs include TYK2 inhibitors, selective JAK pathway modulators, second-generation PDE4 inhibitors, vitamin-D-adjacent or combination-topical platforms, and emerging multi-mechanism regimens aimed at improving adherence and sustained clearance.
- Industry consolidation continues to intensify, reflecting the high strategic value of immunology portfolios. Mergers, acquisitions, and platform partnerships remain central to competitive positioning, reshaping pipelines and accelerating diversification among leading immunology players.
Key Questions Answered:
The report provides answers to the following key questions:
- How big is the psoriasis drugs opportunity?
- What trends, challenges and barriers are influencing its growth?
- How is the ecosystem evolving by segment and region?
- What will the market size be in 2030 and at what rate will it grow?
- Which countries and submarkets will see the highest percentage of growth?
- What are the prospects of biosimilar drugs in psoriasis?
- How big is the market for psoriasis drugs?
- Who are the key market players and what are their strategies?
- How will patent expirations of innovator drugs impact the market?
- What strategies should psoriasis drug manufacturers adopt to remain competitive?
Sample Figure: Distribution of Psoriasis Pipeline Candidates by Developmental Phase (%)
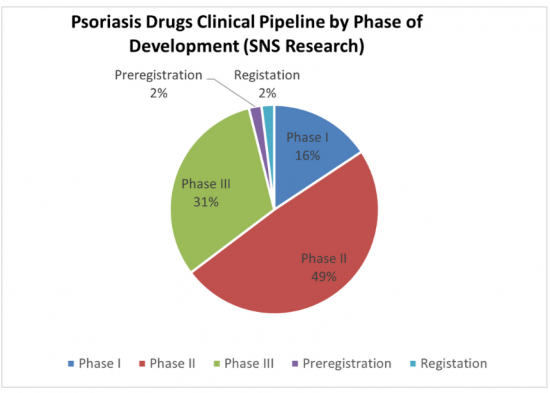
Forecast Segmentation:
Market forecasts are provided for each of the following submarkets and their subcategories:
Therapeutic Class
- TNF Inhibitors
- Interleukins
- Vitamin D Analogues & Topical Steroids
- Others
Route of Administration
- Topical
- Oral
- Parenteral
Distribution Channel
- Point-of-Care / Clinic
- Retail Pharmacies
- Digital Pharmacies
- Direct to Consumer (D2C)
Regional Markets
- Asia Pacific
- Europe
- Middle East & Africa
- North America
- Latin & Central America
Country Markets
- Australia
- Brazil
- Canada
- China
- Egypt
- France
- Germany
- Greece
- India
- Israel
- Italy
- Japan
- Mexico
- Netherlands
- Poland
- Portugal
- Russia
- Saudi Arabia
- South Africa
- South Korea
- Spain
- Switzerland
- Taiwan
- Turkey
- UK
- USA
List of Companies & Organizations:
All the companies and organizations mentioned in the report are listed below:
- Abbott Laboratories
- AbbVie
- AbGenomics International
- Acelyrin
- Aclaris Therapeutics
- Actelion Pharmaceuticals
- Akeso Biopharma
- Akros Pharma
- Akzo Nobel Group
- Almirall
- AltruBio
- Alumis (Previously Esker Therapeutics)
- Alvotech
- Amgen
- Anacor Pharmaceuticals
- AnaptysBio
- Arcutis Biotherapeutics
- Artax Biopharma
- Artelo Biosciences
- AstraZeneca
- Azora Therapeutics
- Bausch & Lomb
- Bausch Health
- Bausch Health Ireland Limited
- Bayer
- Biocad
- Biocon Biologics
- Biofrontera AG
- Biogen
- BioMimetix
- Biosintez
- Bio-Thera Solutions
- BMS (Bristol-Myers Squibb)
- Boehringer Ingelheim
- Boston Pharmaceuticals
- Botanix Pharmaceuticals
- Cadila Healthcare
- Cadila Pharmaceuticals
- Can-Fite BioPharma
- Cantargia
- Celgene Corporation
- Celltrion Healthcare
- Cerbios-Pharma
- Cipher Pharmaceuticals
- Coherus BioSciences
- Dermata Therapeutics
- DICE Therapeutics (Acquired by Eli Lilly)
- Dr. Reddy's Laboratories
- Dusa Pharmaceuticals
- Eli Lilly and Company
- Enavate Sciences
- Encore Dermatology
- EPI Health
- European Medicines Agency (EMA)
- FDA
- Galderma
- Galectin Therapeutics
- Glenmark Pharmaceuticals
- GSK (GlaxoSmithKline)
- Hanwha Pharmaceutical
- Immutep
- Innovent Biologics
- InSite Vision
- Issar Pharma
- Janssen Biotech
- Janssen Pharmaceutical
- Janssen Research & Development
- Japan Tobacco (JT)
- Jiangsu Hengrui Pharmaceuticals Co.
- Johnson & Johnson
- Kyongbo Pharm
- Kyowa Kirin
- LEO Pharma
- Lipidor AB
- Mabpharm
- Maruho
- Mayne Pharma Group
- MC2 Therapeutics
- Meiji Seika Pharma
- Merck
- Merck & Co.
- MIT
- MoonLake Immunotherapeutics
- Mylan
- Nestle Skin Health
- Novartis
- Ono Pharmaceutical
- Ortho Dermatologics
- Pelthos Therapeutics (Part of Ligand Pharmaceuticals)
- Pfizer
- Pharmalucence
- Photogen Technologies Inc.
- Pierre Fabre Laboratories
- Pola Pharma
- Portal Instruments
- Promius Pharma
- Protagonist Therapeutics
- Provectus Biopharmaceuticals
- Rani Therapeutics
- Samsung
- Samsung Bioepis
- Sandoz
- Sanofi
- SFA Therapeutics
- Shire
- Sudo Biosciences
- Sun Pharma
- Suzhou Zelgen Biopharmaceuticals
- Takeda
- Taro Pharmaceutical Industries
- Teva
- Theravance Biopharma
- U.S. Department of Justice
- U.S. Federal Trade Commission
- UCB
- UNION Therapeutics
- URL Pharma
- Vanda Pharmaceuticals
- Viatris
- Vyne Therapeutics
- XBiotech
- Xbrane Biopharma
- Yangji Pharmaceutical
- Zydus Group




