PUBLISHER: Coherent Market Insights | PRODUCT CODE: 1673957

PUBLISHER: Coherent Market Insights | PRODUCT CODE: 1673957
Insulin Glargine Market, By Type, By Diabetes Type, By Distribution Channel, By Geography
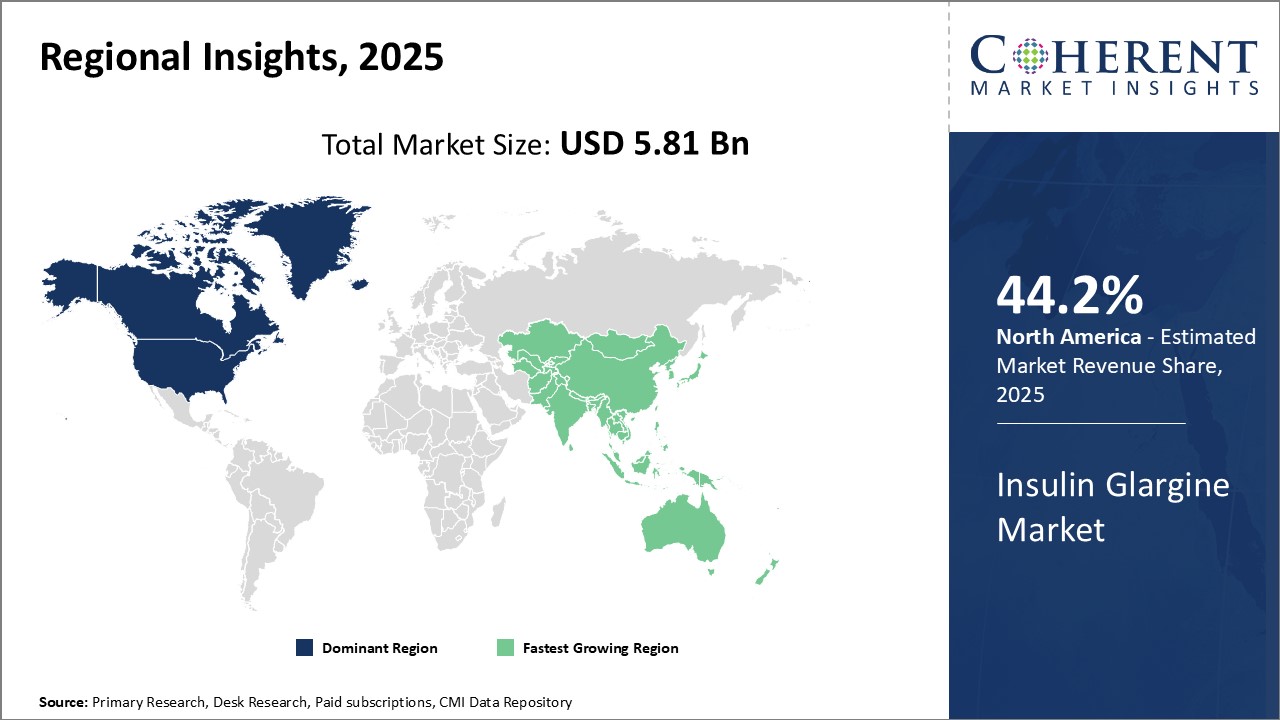
Global Insulin Glargine Market is estimated to be valued at USD 5.81 Bn in 2025 and is expected to reach USD 9.09 Bn by 2032, growing at a compound annual growth rate (CAGR) of 6.6% from 2025 to 2032.
| Report Coverage | Report Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 5.81 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 6.60% | 2032 Value Projection: | USD 9.09 Bn |
Insulin glargine, also known as Lantus, is a long-acting basal insulin that is used for the treatment of diabetes mellitus. It is a synthetic form of human insulin created through recombinant DNA technology, engineered to be soluble in acidic environments. Upon injection, it precipitates and forms microcrystals in the subcutaneous tissue, creating a depot from which insulin is gradually absorbed into the bloodstream. This extended release and duration of action profile allows once-daily dosing to help control blood glucose levels in patients with type 1 and type 2 diabetes over 24 hours. Insulin glargine has a slower absorption profile as compared to other basal insulins such as insulin detemir and insulin NPH, and thus, provides a smooth and consistent level of insulin in the blood.
Market Dynamics:
Global insulin glargine market growth is driven by growing prevalence of diabetes worldwide. According to estimates by the International Diabetes Federation 2021, over 537 million adults lived with diabetes in 2021, and the number is projected to rise to 643 million by 2030 and 783 million by 2045. Increasing geriatric population also contributes to market growth since diabetes is more common in older age groups. However, cost constraints, especially in developing countries, acts as a restrain. On the positive side, ongoing development of more effective and convenient drug delivery technologies is expected to present new opportunities.
Key Features of the Study:
- This report provides in-depth analysis of the global insulin glargine market, and provides market size (USD Bn) and compound annual growth rate (CAGR%) for the forecast period (2025-2032), considering 2024 as the base year.
- It elucidates potential revenue opportunities across different segments and explains attractive investment proposition matrices for this market.
- This study also provides key insights about market drivers, restraints, opportunities, new product launches or approval, market trends, regional outlook, and competitive strategies adopted by key players.
- It profiles key players in the global insulin glargine market based on the following parameters - company highlights, products portfolio, key highlights, financial performance, and strategies.
- Key companies covered as a part of this study includes Sanofi, Biocon, Eli Lilly and Company, Boehringer Ingelheim, Gan & Lee, Kalbe Pharma, Polus Biopharm, Samsung Bioepis, Wockhardt, Novo Nordisk AS, Julphar, and Viatris Inc.
- Insights from this report would allow marketers and the management authorities of the companies to make informed decisions regarding their future product launches, type up-gradation, market expansion, and marketing tactics.
- Global insulin glargine market report caters to various stakeholders in this industry including investors, suppliers, product manufacturers, distributors, new entrants, and financial analysts.
- Stakeholders would have ease in decision-making through various strategy matrices used in analyzing the global insulin glargine market.
Detailed Segmentation
- Type:
- Lantus
- Basaglar
- Toujeo
- Soliqua/Suliqua
- Others
- Diabetes Type:
- Type 2 Diabetes
- Type 1 Diabetes
- Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Regional:
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
- Company Profiles:
- Sanofi
- Biocon
- Eli Lilly and Company
- Boehringer Ingelheim
- Gan & Lee
- Kalbe Pharma
- Polus Biopharm
- Samsung Bioepis
- Wockhardt
- Novo Nordisk AS
- Julphar
- Viatris Inc.
Table of Contents
1. Research Objectives and Assumptions
- Research Objectives
- Assumptions
- Abbreviations
2. Market Purview
- Report Description
- Market Definition and Scope
- Executive Summary
- Market Snapshot, By Type
- Market Snapshot, By Diabetes Type
- Market Snapshot, By Distribution Channel
- Market Snapshot, By Region
- Coherent Opportunity Map (COM)
3. Market Dynamics, Regulations, and Trends Analysis
- Market Dynamics
- Increasing prevalence of diabetes worldwide
- High cost of insulin glargine
- Increase in awareness about diabetes management
- Impact Analysis
- Key Highlights
- Regulatory Scenario
- Product Launch/Approvals
- PEST Analysis
- PORTER's Analysis
- Merger and Acquisition Scenario
4. Global Insulin Glargine Market- Impact of Coronavirus (COVID-19) Pandemic
- COVID-19 Epidemiology
- Supply Side and Demand Side Analysis
- Economic Impact
5. Global Insulin Glargine Market, By Type, 2020-2032, (USD BN)
- Introduction
- Market Share Analysis, 2025 and 2032 (%)
- Y-o-Y Growth Analysis, 2020 - 2032
- Segment Trends
- Lantus
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (USD BN)
- Basaglar
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (USD BN)
- Toujeo
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (USD BN)
- Soliqua/Suliqua
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (USD BN)
- Others
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (USD BN)
6. Global Insulin Glargine Market, By Diabetes Type, 2020-2032, (USD BN)
- Introduction
- Market Share Analysis, 2025 and 2032 (%)
- Y-o-Y Growth Analysis, 2020 - 2032
- Segment Trends
- Type 2 Diabetes
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (USD BN)
- Type 1 Diabetes
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (USD BN)
7. Global Insulin Glargine Market, By Distribution Channel, 2020-2032, (USD BN)
- Introduction
- Market Share Analysis, 2025 and 2032 (%)
- Y-o-Y Growth Analysis, 2020 - 2032
- Segment Trends
- Hospital Pharmacies
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (USD BN)
- Retail Pharmacies
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (USD BN)
- Online Pharmacies
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, 2020-2032, (USD BN)
8. Global Insulin Glargine Market, By Region, 2020-2032, (USD BN)
- Introduction
- Market Share Analysis, 2025 and 2032 (%)
- Y-o-Y Growth Analysis, 2020 - 2032
- Segment Trends
- North America
- Market Size and Forecast, and Y-o-Y Growth, By Type, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Diabetes Type, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Distribution Channel, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2020 - 2032, (USD BN)
- U.S.
- Canada
- Europe
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, By Type, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Diabetes Type, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Diabetes Type, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Distribution Channel, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2020 - 2032, (USD BN)
- U.K.
- Germany
- Italy
- France
- Spain
- Russia
- Asia Pacific
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, By Type, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Diabetes Type, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Distribution Channel, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2020 - 2032, (USD BN)
- China
- India
- Japan
- ASEAN
- Australia
- South Korea
- Rest of Asia Pacific
- Latin America
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, By Type, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Diabetes Type, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Distribution Channel, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2020 - 2032, (USD BN)
- Brazil
- Mexico
- Argentina
- Rest of Latin America
- Middle East
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, By Type, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Diabetes Type, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Distribution Channel, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Country, 2020 - 2032, (USD BN)
- GCC
- Israel
- Rest of Middle East
- Africa
- Introduction
- Market Size and Forecast, and Y-o-Y Growth, By Type, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Diabetes Type, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Distribution Channel, 2020-2032, (USD BN)
- Market Size and Forecast, and Y-o-Y Growth, By Country/Region, 2020 - 2032, (USD BN)
- North Africa
- Central Africa
- South Africa
9. Competitive Landscape
- Sanofi
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- Biocon
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- Eli Lilly and Company
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- Boehringer Ingelheim
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- Gan & Lee
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- Kalbe Pharma
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- Polus Biopharm
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- Samsung Bioepis
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- Wockhardt
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- Novo Nordisk AS
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- Julphar
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
- Viatris Inc.
- Company Highlights
- Product Portfolio
- Key Developments
- Financial Performance
- Strategies
10. Analyst View
- Wheel of Fortune
- Analyst View
- Coherent Opportunity Map
11. References and Research Methodology
- References
- Research Methodology
- About us and Sales Contact




