PUBLISHER: Global Market Insights Inc. | PRODUCT CODE: 1871158

PUBLISHER: Global Market Insights Inc. | PRODUCT CODE: 1871158
Decentralized Clinical Trials Market Opportunity, Growth Drivers, Industry Trend Analysis, and Forecast 2025 - 2034
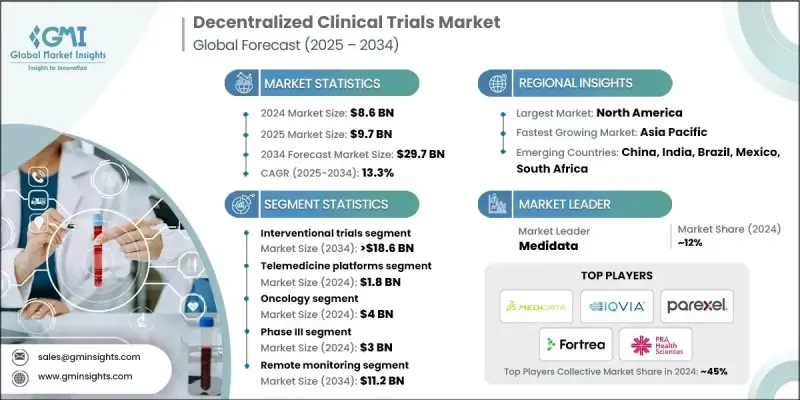
The Global Decentralized Clinical Trials Market was valued at USD 8.6 Billion in 2024 and is estimated to grow at a CAGR of 13.3% to reach USD 29.7 Billion by 2034.
The market's rapid growth is fueled by the widespread use of digital health solutions such as telemedicine platforms, electronic patient-reported outcomes (ePRO), wearable medical devices, and e-consent systems that are reshaping the way clinical research is conducted. Decentralized models remove traditional site constraints, enabling participation from patients across different geographic regions without the need to travel to centralized locations. Regulatory authorities across major markets are increasingly introducing structured frameworks to support the implementation of decentralized trial approaches. This global transition reflects the growing emphasis on patient convenience, real-time data collection, and inclusivity in research participation. With aging populations, higher incidences of chronic diseases, and the shift toward personalized medicine, healthcare systems are moving toward patient-focused models that make clinical trials more accessible and efficient. Decentralized trials align with these evolving trends by integrating digital tools to streamline processes, reduce operational costs, and improve patient engagement throughout the study lifecycle.
| Market Scope | |
|---|---|
| Start Year | 2024 |
| Forecast Year | 2025-2034 |
| Start Value | $8.6 Billion |
| Forecast Value | $29.7 Billion |
| CAGR | 13.3% |
In decentralized studies, some or all trial-related procedures occur outside traditional research sites, allowing participants to engage from home or through local medical facilities. These trials use digital platforms, connected devices, and remote monitoring systems to collect clinical data and deliver treatments directly to participants. This design helps minimize travel demands and enhances inclusivity, creating more diverse and representative study populations.
The interventional trials segment captured a 63.7% share in 2024 and is forecast to reach USD 18.6 Billion by 2034. This growth is supported by remote participation capabilities that minimize the need for physical visits and improve trial accessibility for elderly and geographically distributed populations. Enhanced patient experience leads to stronger retention rates, while connected digital tools such as mobile applications, telehealth, and ePRO platforms enable continuous real-time monitoring and faster data acquisition, making interventional trials more scalable and efficient.
The telemedicine platforms segment generated USD 1.8 Billion in 2024. These platforms help eliminate travel and location barriers, supporting wider participation among patients from remote and underserved areas. Their integration with connected devices and digital reporting systems enhances the quality and speed of data collection, improving overall trial efficiency. The digital nature of telemedicine also lowers site management costs, shortens study durations, and simplifies coordination between sponsors, investigators, and patients. By creating an interconnected digital framework, telemedicine platforms allow more streamlined and adaptive decentralized trial operations.
North America Decentralized Clinical Trials Market held 44.1% share in 2024. The region benefits from highly advanced digital infrastructure, including widespread access to mobile networks, cloud-based systems, and connected health technologies. Growing patient demand for flexible, convenient participation options is driving adoption across various therapeutic areas. Decentralized models in this region are improving recruitment efficiency and retention rates by allowing participation directly from patients' homes or nearby healthcare centers. Sponsors in North America are increasingly relying on these models to optimize development timelines, accelerate product launches, and improve overall return on investment.
Leading companies shaping the Global Decentralized Clinical Trials Market include OpenClinica, Medidata, Covance, Sano Genetics, PPD (Thermo Fisher Scientific), Florence Healthcare, Reify Health, Mahalo Health, Bio-Optronics, Trialize, ClinOne, Veeva Systems, ClinTex, Parexel, PRA Health Sciences (ICON), Sanguine Bio, IQVIA, ProPharma, Fortrea, and Clinical Research IO. Companies in the decentralized clinical trials sector are strengthening their market position by focusing on digital innovation, platform integration, and global partnerships. Many are developing unified cloud-based systems that connect telemedicine, ePRO, and data analytics tools for seamless trial management. Strategic collaborations with biopharma sponsors and technology providers are helping enhance interoperability and expand service portfolios.
Table of Contents
Chapter 1 Methodology and Scope
- 1.1 Market scope and definition
- 1.2 Research design
- 1.2.1 Research approach
- 1.2.2 Data collection methods
- 1.3 Data mining sources
- 1.3.1 Global
- 1.3.2 Regional/Country
- 1.4 Base estimates and calculations
- 1.4.1 Base year calculation
- 1.4.2 Key trends for market estimation
- 1.5 Primary research and validation
- 1.5.1 Primary sources
- 1.6 Forecast model
- 1.7 Research assumptions and limitations
Chapter 2 Executive Summary
- 2.1 Industry 360° synopsis
- 2.2 Key market trends
- 2.2.1 Regional trends
- 2.2.2 Component trends
- 2.2.3 Application trends
- 2.2.4 End Use trends
- 2.3 CXO perspectives: Strategic imperatives
- 2.3.1 Key decision points for industry executives
- 2.3.2 Critical success factors for market players
- 2.4 Future outlook and strategic recommendations
Chapter 3 Industry Insights
- 3.1 Industry ecosystem analysis
- 3.2 Industry impact forces
- 3.2.1 Growth drivers
- 3.2.1.1 Technological advancement in digital health
- 3.2.1.2 Cost efficiency and accelerated timelines
- 3.2.1.3 Regulatory support and framework development
- 3.2.1.4 Improved patient accessibility and diversity
- 3.2.2 Industry pitfalls and challenges
- 3.2.2.1 Data integrity and standardization
- 3.2.2.2 Regulatory complexity
- 3.2.3 Market opportunities
- 3.2.3.1 Integration of AI and wearables
- 3.2.3.2 Expansion into underserved regions
- 3.2.1 Growth drivers
- 3.3 Growth potential analysis
- 3.4 Regulatory landscape
- 3.4.1 North America
- 3.4.1.1 U.S.
- 3.4.1.2 Canada
- 3.4.2 Europe
- 3.4.3 Asia Pacific
- 3.4.4 Latin America
- 3.4.5 Middle East and Africa
- 3.4.1 North America
- 3.5 Technology landscape
- 3.5.1 Current technological trends
- 3.5.2 Emerging technologies
- 3.6 Future market trends
- 3.7 Porter's analysis
- 3.8 PESTEL analysis
Chapter 4 Competitive Landscape, 2024
- 4.1 Introduction
- 4.2 Company market share analysis
- 4.2.1 Global
- 4.2.2 North America
- 4.2.3 Europe
- 4.3 Company matrix analysis
- 4.4 Competitive analysis of major market players
- 4.5 Competitive positioning matrix
- 4.6 Key developments
- 4.6.1 Merger and acquisition
- 4.6.2 Partnership and collaboration
- 4.6.3 New product/services launches
- 4.6.4 Expansion plans
Chapter 5 Market Estimates and Forecast, By Study Design, 2021 - 2034 ($ Mn)
- 5.1 Key trends
- 5.2 Interventional trials
- 5.2.1 Randomized controlled trials (RCTs)
- 5.2.2 Adaptive clinical trials
- 5.2.3 Pragmatic clinical trials
- 5.3 Observational trials
- 5.3.1 Cohort studies
- 5.3.2 Retrospective studies
- 5.3.3 Longitudinal studies
- 5.4 Expanded access trials
Chapter 6 Market Estimates and Forecast, By Technology, 2021 - 2034 ($ Mn)
- 6.1 Key trends
- 6.2 Telemedicine platforms
- 6.3 Wearable devices
- 6.4 Mobile health applications
- 6.5 Electronic data capture (EDC) systems
- 6.6 Other technologies
Chapter 7 Market Estimates and Forecast, By Therapeutic Area, 2021 - 2034 ($ Mn)
- 7.1 Key trends
- 7.2 Oncology
- 7.3 Cardiology
- 7.4 Neurology
- 7.5 Infectious diseases
- 7.6 Respiratory disorders
- 7.7 Other chronic conditions
Chapter 8 Market Estimates and Forecast, By Study Phase, 2021 - 2034 ($ Mn)
- 8.1 Key trends
- 8.2 Phase I
- 8.3 Phase II
- 8.4 Phase III
- 8.5 Phase IV
Chapter 9 Market Estimates and Forecast, By Participant Engagement, 2021 - 2034 ($ Mn)
- 9.1 Key trends
- 9.2 Patient-centric approaches
- 9.3 Remote monitoring
- 9.4 Digital surveys
- 9.5 Virtual site visits
Chapter 10 Market Estimates and Forecast, By Region, 2021 - 2034 ($ Mn)
- 10.1 Key trends
- 10.2 North America
- 10.2.1 U.S.
- 10.2.2 Canada
- 10.3 Europe
- 10.3.1 Germany
- 10.3.2 UK
- 10.3.3 France
- 10.3.4 Spain
- 10.3.5 Italy
- 10.3.6 Netherlands
- 10.4 Asia Pacific
- 10.4.1 China
- 10.4.2 India
- 10.4.3 Japan
- 10.4.4 Australia
- 10.4.5 South Korea
- 10.5 Latin America
- 10.5.1 Brazil
- 10.5.2 Mexico
- 10.5.3 Argentina
- 10.6 Middle East and Africa
- 10.6.1 South Africa
- 10.6.2 Saudi Arabia
- 10.6.3 UAE
Chapter 11 Company Profiles
- 11.1 Propharma
- 11.2 IQVIA
- 11.3 Medidata
- 11.4 Parexel
- 11.5 Fortrea
- 11.6 Covance
- 11.7 PRA Health Sciences (ICON)
- 11.8 PPD (Thermo Fisher Scientific)
- 11.9 Mahalo Health
- 11.10 Clinical Research IO
- 11.11 Veeva Systems
- 11.12 OpenClinica
- 11.13 Bio-Optronics
- 11.14 Sano Genetics
- 11.15 ClinOne
- 11.16 Sanguine Bio
- 11.17 Florence Healthcare
- 11.18 Reify Health
- 11.19 Trialize
- 11.20 ClinTex




