PUBLISHER: Global Market Insights Inc. | PRODUCT CODE: 1998811

PUBLISHER: Global Market Insights Inc. | PRODUCT CODE: 1998811
Glucagon Market Opportunity, Growth Drivers, Industry Trend Analysis, and Forecast 2026 - 2035
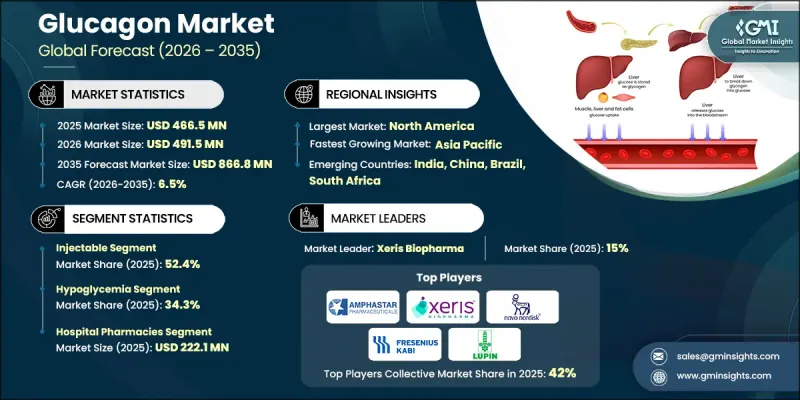
The Global Glucagon Market was valued at USD 466.5 million in 2025 and is estimated to grow at a CAGR of 6.5% to reach USD 866.8 million by 2035.
Market expansion is fueled by the rising prevalence of diabetes and the increasing frequency of hypoglycemic episodes, which continue to impose a significant healthcare burden worldwide. Beyond the growing demand driven by diabetes-related emergencies, innovations in glucagon delivery systems are reshaping the market landscape. The diabetes care sector is increasingly adopting integrated, automated, and patient-friendly solutions supported by digital health ecosystems. Advances in glucagon formulations have transformed severe hypoglycemia management, moving away from multi-step reconstitution kits to ready-to-use, highly stable, and convenient options. Stable liquid glucagon formulations now remain usable at room temperature and can be delivered via autoinjectors or prefilled syringes, eliminating complicated preparation. These innovations reduce administration time, enhance ease of use for caregivers and emergency responders, and improve the likelihood of timely treatment during critical hypoglycemia events.
| Market Scope | |
|---|---|
| Start Year | 2025 |
| Forecast Year | 2026-2035 |
| Start Value | $466.5 Million |
| Forecast Value | $866.8 Million |
| CAGR | 6.5% |
The injectable segment held 52.4% share in 2025. Injectable glucagon provides a rapid onset of action, often within minutes, making it crucial in severe hypoglycemia scenarios to prevent seizures, unconsciousness, or long-term neurological damage. Injectable formulations offer flexibility in administration routes, including subcutaneous, intramuscular, and intravenous options, allowing tailored treatment across home, clinical, and hospital settings. This adaptability ensures the segment retains leadership by meeting diverse patient needs and urgent care requirements.
The hospital pharmacies segment contributed USD 222.1 million in 2025, ensuring continuous availability and safe handling of glucagon across emergency departments, inpatient wards, intensive care units, and diagnostic suites. Hospital pharmacists oversee the correct storage, preparation, and administration of glucagon, maintain its stability, and train nursing and support staff on proper emergency use. These environments also serve as critical educational hubs, emphasizing accurate dosage, administration steps, and post-treatment monitoring to ensure patient safety.
North America Glucagon Market held the largest share in 2025, driven by advanced healthcare infrastructure, high adoption of emergency diabetes therapies, and widespread use of ready-to-use glucagon formulations. Well-established hospital networks, pharmacy accessibility, and robust regulatory frameworks support rapid distribution and approval of innovative glucagon products. Hospital pharmacies maintain glucagon as a vital emergency medication, ensuring immediate access for severe hypoglycemia, preparation of pediatric doses, and use in diagnostic procedures. Retail and online pharmacies complement hospital distribution, providing convenient access to prescription glucagon for home use.
Prominent companies operating in the Global Glucagon Market include Amphastar Pharmaceuticals, Avalon Pharma, Eli Lilly and Company, Fresenius Kabi, Hanmi Pharmaceutical, Lupin Limited, Novo Nordisk A/S, Taj Pharmaceuticals, Xeris Biopharma Holdings, and Zealand Pharma. Companies in the Global Glucagon Market are strengthening their presence through multiple strategic approaches. Firms are investing in research and development to innovate stable, ready-to-use formulations and advanced autoinjector devices that improve ease of administration and patient compliance. Expanding distribution channels, including hospitals, retail, and online pharmacies, ensures broad product availability. Strategic collaborations with healthcare providers, emergency response networks, and patient advocacy organizations enhance education and awareness about timely hypoglycemia management. Regulatory engagement and fast-track approvals for innovative products help accelerate market entry.
Table of Contents
Chapter 1 Methodology and Scope
- 1.1 Research design
- 1.1.1 Research approach
- 1.1.2 Data collection methods
- 1.2 Data mining sources
- 1.2.1 Global
- 1.2.2 Regional/Country
- 1.3 Base estimates and calculations
- 1.3.1 Base year calculation
- 1.3.2 Key trends for market estimation
- 1.4 Primary research and validation
- 1.4.1 Primary sources
- 1.5 Forecast model
- 1.6 Research assumptions and limitations
Chapter 2 Executive Summary
- 2.1 Industry 360° synopsis
- 2.2 Key market trends
- 2.2.1 Product type trends
- 2.2.2 Application trends
- 2.2.3 Distribution channel trends
- 2.3 CXO perspectives: Strategic imperatives
Chapter 3 Industry Insights
- 3.1 Industry ecosystem analysis
- 3.2 Industry impact forces
- 3.2.1 Growth drivers
- 3.2.1.1 Rising prevalence of diabetes worldwide
- 3.2.1.2 Technological advancements in glucagon delivery methods
- 3.2.1.3 Increasing research and development activities
- 3.2.2 Industry pitfalls and challenges
- 3.2.2.1 High cost of medications
- 3.2.2.2 Side effects associated with glucagon
- 3.2.3 Market opportunity
- 3.2.3.1 Growing adoption in emergency response systems
- 3.2.1 Growth drivers
- 3.3 Growth potential analysis
- 3.4 Regulatory landscape
- 3.5 Technological landscape
- 3.5.1 Current technologies
- 3.5.2 Emerging technologies
- 3.6 Future market trends
- 3.7 Impact of AI and generative AI on the market
- 3.8 Porter's analysis
- 3.9 PESTEL analysis
Chapter 4 Competitive Landscape, 2025
- 4.1 Introduction
- 4.2 Company market share analysis
- 4.2.1 Global
- 4.2.2 North America
- 4.2.3 Europe
- 4.3 Company matrix analysis
- 4.4 Competitive analysis of major market players
- 4.5 Competitive positioning matrix
- 4.6 Key developments
- 4.6.1 Merger and acquisition
- 4.6.2 Partnership and collaboration
- 4.6.3 New product launches
- 4.6.4 Expansion plans
Chapter 5 Market Estimates and Forecast, By Product Type, 2022 - 2035 ($ Mn)
- 5.1 Key trends
- 5.2 Injectable
- 5.3 Inhalation
Chapter 6 Market Estimates and Forecast, By Application, 2022 - 2035 ($ Mn)
- 6.1 Key trends
- 6.2 Hypoglycemia
- 6.3 Diagnostic aid
- 6.4 Gastrointestinal disorders
- 6.5 Cardiogenic shock
- 6.6 Other applications
Chapter 7 Market Estimates and Forecast, By Distribution Channel, 2022 - 2035 ($ Mn)
- 7.1 Key trends
- 7.2 Hospital pharmacies
- 7.3 Retail pharmacies
- 7.4 Online pharmacies
Chapter 8 Market Estimates and Forecast, By Region, 2022 - 2035 ($ Mn)
- 8.1 Key trends
- 8.2 North America
- 8.2.1 U.S.
- 8.2.2 Canada
- 8.3 Europe
- 8.3.1 Germany
- 8.3.2 UK
- 8.3.3 France
- 8.3.4 Spain
- 8.3.5 Italy
- 8.3.6 Russia
- 8.3.7 Netherlands
- 8.4 Asia Pacific
- 8.4.1 China
- 8.4.2 Japan
- 8.4.3 India
- 8.4.4 Australia
- 8.4.5 South Korea
- 8.5 Latin America
- 8.5.1 Brazil
- 8.5.2 Mexico
- 8.5.3 Argentina
- 8.6 Middle East and Africa
- 8.6.1 South Africa
- 8.6.2 Saudi Arabia
- 8.6.3 UAE
Chapter 9 Company Profiles
- 9.1 Amphastar Pharmaceuticals
- 9.2 Avalon Pharma
- 9.3 Eli Lilly and Company
- 9.4 Fresenius Kabi
- 9.5 Hanmi Pharmaceutical
- 9.6 Lupin Limited
- 9.7 Novo Nordisk A/S
- 9.8 Taj Pharmaceuticals
- 9.9 Xeris Biopharma Holdings
- 9.10 Zealand Pharma




