PUBLISHER: MarketsandMarkets | PRODUCT CODE: 2027002

PUBLISHER: MarketsandMarkets | PRODUCT CODE: 2027002
Medical Adhesives Market by Technology (Water-Based, Solvent-Based, Solid & Hot Melt Based), Resin Type (Natural Resin, Synthetic & Semi-Synthetic Resin), Application (Dental, Surgery, Medical Device & Equipment), And Region - Global Forecast To 2031
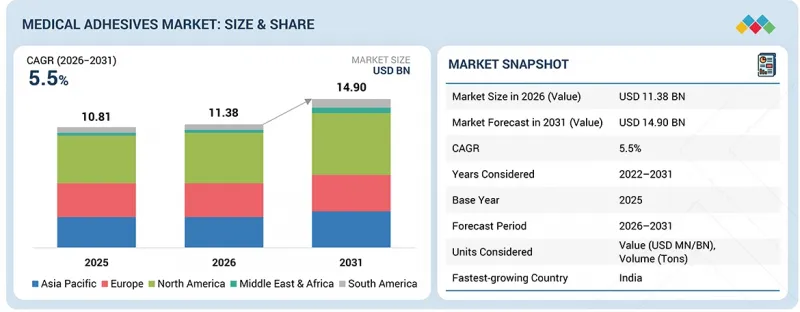
The medical adhesives market is projected to grow from USD 11.38 billion in 2026 to USD 14.90 billion by 2031, at a CAGR of 5.5% during the forecast period.
| Scope of the Report | |
|---|---|
| Years Considered for the Study | 2022-2031 |
| Base Year | 2025 |
| Forecast Period | 2026-2031 |
| Units Considered | Value (USD Million), Volume (Tons) |
| Segments | By Technology, Resin Type, Application, and Region |
| Regions covered | Europe, North America, Asia Pacific, Middle East & Africa, and South America |
The medical adhesives market is growing as healthcare facilities need stronger, biocompatible, and durable bonding materials. The rising number of surgical procedures, together with the growing acceptance of minimally invasive procedures, is driving hospitals to use medical adhesives for wound closure, tissue sealing, and hemostasis.
The increasing use of wearable medical devices, together with transdermal drug delivery systems, creates a higher demand for skin-compatible flexible adhesives that can be worn for extended periods. The combination of growing elderly populations and rising chronic wound cases drives higher demand for advanced wound care products, including adhesive dressings and sealants. The medical device manufacturing industry requires high-performance adhesives, which are essential for assembling and fixing devices. The rapid development of healthcare facilities in emerging markets opens new business opportunities. Product performance and market growth receive boosts from ongoing supply-side product advances, the development of silicone and acrylic adhesive systems, and increased dedication to meeting regulatory requirements and using sustainable materials.
Based on technology, the medical adhesives market is divided into three segments: water-based adhesives, solvent-based adhesives, and solid & hot melt adhesives. Water-based adhesives lead the market because they provide excellent biocompatibility and low toxicity and produce minimal volatile organic compound emissions, making them suitable for skin-contact and wound care applications. Medical tapes, dressings, and wearable devices use these adhesives to ensure patient safety and comfort. The bonding strength and durability of solvent-based adhesives remain strong, yet their market growth continues to decline because of environmental regulations that restrict solvent emissions. Solid and hot-melt adhesives are now seeing increased use in medical device assembly and hygiene applications because they provide fast curing times, simple processing, and effective adhesion to multiple materials. The water-based adhesive market is the dominant force because rising regulations mandate environmentally friendly products without toxic components and because healthcare demands require both safe and effective solutions.
Based on resin type, the medical adhesives market is divided into three categories: fibrin, collagen, and other medical adhesive products. Fibrin-based adhesives maintain the largest market share because they provide better biocompatibility and function as natural components of the body healing process. Fibrin adhesives, which derive from human or animal plasma components, create a complete blood coagulation system that surgeons use for hemostasis, tissue sealing, and wound closure. The medical community uses these products because they help patients heal faster while controlling blood loss and reducing the risk of adverse health effects during cardiovascular, orthopedic, and general surgical operations. Collagen-based adhesives are becoming more popular because they provide excellent tissue compatibility, which helps cells grow, especially in wound care and regenerative medicine. Specialized applications that need strong bonding and controlled degradation use synthetic adhesives, which include cyanoacrylates and polyethylene glycol (PEG)-based formulations as their preferred resin types. Fibrin adhesives represent the preferred choice because they provide safe usage, effective results in emergency medical situations, and growing demand for biologically based medical solutions in contemporary healthcare.
Based on region, Asia Pacific represents the fastest expanding market for medical adhesives because healthcare facilities in China, India, and Southeast Asia experience rapid development while their population numbers and healthcare spending patterns continue to rise. The increasing number of chronic disease cases, together with the expanding elderly population, creates a greater need for advanced wound care solutions and surgical treatments, which results in higher consumption of medical adhesives. The region experiences strong growth in medical device manufacturing due to lower costs, supportive government regulations, and rising foreign investment. Product usage increases as hospitals, clinics, and ambulatory care centers continue to expand their operations. Rising knowledge of minimally invasive surgical methods, together with modern treatment solutions, creates a market need for superior adhesive products. The medical adhesives market in Asia Pacific experiences rapid growth because local manufacturing resources and foreign companies establishing production centers in the area enhance supply distribution and operational efficiency.
Major players operating in the market include Solventum (US), Henkel AG & Co. KGaA (Germany), H.B. Fuller Company (US), Scapa Healthcare (US), Johnson & Johnson (US), Permabond (UK), Chemence Medical, Inc (US), Artivion, Inc (US), Dymax (Ireland), and Bostik (France). These companies have dependable manufacturing facilities across the Asia Pacific region, as well as robust distribution networks. They have a well-established portfolio that includes reliable goods and services, a strong market presence, and effective business plans. These businesses also hold a sizable portion of the market, offer a broader range of products with more applications, and use cases spanning more geographic regions.
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 STUDY OBJECTIVES
- 1.2 MARKET DEFINITION
- 1.3 STUDY SCOPE
- 1.3.1 MARKET COVERED AND REGIONAL SCOPE
- 1.3.2 INCLUSIONS AND EXCLUSIONS
- 1.3.3 YEARS CONSIDERED
- 1.3.4 CURRENCY CONSIDERED
- 1.3.5 UNITS CONSIDERED
- 1.4 STAKEHOLDERS
2 EXECUTIVE SUMMARY
- 2.1 KEY INSIGHTS AND MARKET HIGHLIGHTS
- 2.2 KEY MARKET PARTICIPANTS: MAPPING OF STRATEGIC DEVELOPMENTS
- 2.3 DISRUPTIVE TRENDS IN MEDICAL ADHESIVES MARKET
- 2.4 HIGH GROWTH SEGMENTS
- 2.5 REGIONAL SNAPSHOT: MARKET SIZE, GROWTH RATE, AND FORECAST
3 PREMIUM INSIGHTS
- 3.1 ATTRACTIVE OPPORTUNITIES FOR PLAYERS IN MEDICAL ADHESIVES MARKET
- 3.2 MEDICAL ADHESIVES MARKET, BY TECHNOLOGY AND REGION
- 3.3 MEDICAL ADHESIVES MARKET, BY RESIN TYPE
- 3.4 MEDICAL ADHESIVES MARKET, BY APPLICATION
- 3.5 MEDICAL ADHESIVES MARKET, BY COUNTRY
4 MARKET OVERVIEW
- 4.1 INTRODUCTION
- 4.2 MARKET DYNAMICS
- 4.2.1 DRIVERS
- 4.2.1.1 Rising demand for minimally invasive surgeries
- 4.2.1.2 Growth in wearable and portable medical devices
- 4.2.1.3 Advancements in biocompatible and light-curable technologies
- 4.2.2 RESTRAINTS
- 4.2.2.1 Stringent regulatory requirement
- 4.2.2.2 Risk of skin irritation and biocompatibility issues
- 4.2.2.3 Availability of alternative wound closure methods
- 4.2.3 OPPORTUNITIES
- 4.2.3.1 Growth in home healthcare and remote diagnostics
- 4.2.3.2 Innovations in advanced adhesive technologies
- 4.2.3.3 Integration with smart and minimally invasive surgical solutions
- 4.2.4 CHALLENGES
- 4.2.4.1 Compatibility with new medical devices
- 4.2.4.2 Technological complexity in product development
- 4.2.1 DRIVERS
- 4.3 UNMET NEEDS AND WHITE SPACES
- 4.3.1 UNMET NEEDS IN MEDICAL ADHESIVES MARKET
- 4.3.2 WHITE SPACE OPPORTUNITIES
- 4.4 INTERCONNECTED MARKETS AND CROSS-SECTOR OPPORTUNITIES
- 4.4.1 INTERCONNECTED MARKETS
- 4.4.2 CROSS-SECTOR OPPORTUNITIES
- 4.5 STRATEGIC MOVES BY TIER-1/2/3 PLAYERS
- 4.5.1 KEY MOVES AND STRATEGIC FOCUS
- 4.6 PORTER'S FIVE FORCES ANALYSIS
- 4.6.1 THREAT OF NEW ENTRANTS
- 4.6.2 THREAT OF SUBSTITUTES
- 4.6.3 BARGAINING POWER OF SUPPLIERS
- 4.6.4 BARGAINING POWER OF BUYERS
- 4.6.5 INTENSITY OF COMPETITIVE RIVALRY
- 4.7 VALUE CHAIN ANALYSIS
- 4.8 ECOSYSTEM
- 4.9 PRICING ANALYSIS
- 4.9.1 AVERAGE SELLING PRICE, BY REGION
- 4.9.2 AVERAGE SELLING PRICE, BY TECHNOLOGY
- 4.9.3 AVERAGE SELLING PRICE, BY APPLICATION
- 4.10 MACROECONOMIC INDICATORS
- 4.10.1 GLOBAL GDP TRENDS
- 4.11 IMPACT OF 2025 US TARIFFS ON MEDICAL ADHESIVES MARKET
- 4.11.1 INTRODUCTION
- 4.11.2 KEY TARIFF RATES
- 4.11.3 PRICE IMPACT ANALYSIS
- 4.11.4 IMPACT ON COUNTRIES/REGIONS
- 4.11.4.1 US
- 4.11.4.2 Europe
- 4.11.4.3 Asia Pacific
- 4.11.5 IMPACT ON END-USE INDUSTRIES
- 4.12 TRADE ANALYSIS
- 4.12.1 IMPORT SCENARIO (HS CODE 300590)
- 4.12.2 EXPORT SCENARIO (HS CODE 300590)
- 4.13 TRENDS/DISRUPTIONS IMPACTING CUSTOMER BUSINESS
- 4.14 INVESTMENT AND FUNDING SCENARIO
- 4.15 CASE STUDIES
- 4.15.1 BIOCOMPATIBLE EPOXY ENCAPSULANTS IN MEDICAL DEVICE MANUFACTURING
- 4.15.2 SILICONE ADHESIVES FOR LONG-WEAR WEARABLE DEVICES
- 4.15.3 CYANOACRYLATE ADHESIVES FOR SURGICAL WOUND CLOSURE
- 4.16 KEY CONFERENCES & EVENTS
5 TECHNOLOGICAL ADVANCEMENTS, AI-DRIVEN IMPACT, PATENTS, INNOVATIONS, AND FUTURE APPLICATIONS
- 5.1 TECHNOLOGY ANALYSIS
- 5.1.1 KEY TECHNOLOGIES
- 5.1.1.1 Solids hot melt adhesives
- 5.1.1.2 UV curable adhesives
- 5.1.2 COMPLEMENTARY TECHNOLOGIES
- 5.1.2.1 Water-based (emulsion) adhesives
- 5.1.2.2 Silicone-based adhesives
- 5.1.3 ADJACENT TECHNOLOGIES
- 5.1.3.1 Medical sealants & hemostats
- 5.1.1 KEY TECHNOLOGIES
- 5.2 TECHNOLOGY/PRODUCT ROADMAP
- 5.2.1 SHORT TERM (2025-2027) | FOUNDATION & EARLY COMMERCIALIZATION
- 5.2.2 MID-TERM (2027-2030) | EXPANSION & STANDARDIZATION
- 5.2.3 LONG TERM (2030-2030+) | MASS COMMERCIALIZATION & DISRUPTION
- 5.3 PATENT ANALYSIS
- 5.3.1 LEGAL STATUS OF PATENTS
- 5.3.2 JURISDICTION ANALYSIS
- 5.4 FUTURE APPLICATIONS
- 5.4.1 WEARABLE & REMOTE MONITORING DEVICES
- 5.4.2 MINIMALLY INVASIVE & ROBOTIC SURGERIES
- 5.4.3 IMPLANTABLE MEDICAL DEVICES
- 5.4.4 ADVANCED WOUND CARE & REGENERATIVE MEDICINE
- 5.4.5 TRANSDERMAL DRUG DELIVERY SYSTEMS
- 5.5 IMPACT OF AI/GEN AI ON MEDICAL ADHESIVES MARKET
- 5.5.1 TOP USE CASES AND MARKET POTENTIAL
- 5.5.2 BEST PRACTICES IN MEDICAL ADHESIVES PROCESSING
- 5.5.3 CASE STUDIES OF AI IMPLEMENTATION IN MEDICAL ADHESIVES MARKET
- 5.5.4 INTERCONNECTED ADJACENT ECOSYSTEM AND IMPACT ON MARKET PLAYERS
- 5.5.5 CLIENTS' READINESS TO ADOPT GENERATIVE AI IN MEDICAL ADHESIVES MARKET
6 REGULATORY LANDSCAPE AND SUSTAINABILITY INITIATIVES
- 6.1 REGIONAL REGULATIONS AND COMPLIANCE
- 6.1.1 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- 6.1.2 INDUSTRY STANDARDS
- 6.2 IMPACT OF REGULATORY POLICIES AND SUSTAINABILITY INITIATIVES
- 6.3 CERTIFICATIONS, LABELING, AND ECO-STANDARDS
7 CUSTOMER LANDSCAPE & BUYER BEHAVIOR
- 7.1 INTRODUCTION
- 7.2 DECISION-MAKING PROCESS
- 7.3 KEY STAKEHOLDERS AND BUYING CRITERIA
- 7.3.1 KEY STAKEHOLDERS IN BUYING PROCESS
- 7.3.2 BUYING CRITERIA
- 7.4 ADOPTION BARRIERS & INTERNAL CHALLENGES
- 7.5 UNMET NEEDS IN VARIOUS END-USE INDUSTRIES
- 7.6 MARKET PROFITABILITY
- 7.6.1 REVENUE POTENTIAL
- 7.6.2 COST DYNAMICS
- 7.6.3 MARGIN OPPORTUNITIES IN KEY END-USE INDUSTRIES
8 MEDICAL ADHESIVES MARKET, BY RESIN TYPE
- 8.1 INTRODUCTION
- 8.2 NATURAL RESIN
- 8.2.1 GROWING PREFERENCE FOR BIOCOMPATIBLE AND BIO-BASED SOLUTIONS DRIVING ADOPTION
- 8.2.2 FIBRIN
- 8.2.2.1 Rising surgical procedures boosting adoption of fibrin-based medical adhesives
- 8.2.3 COLLAGEN
- 8.2.3.1 Expanding advanced wound care applications driving demand
- 8.2.4 OTHER NATURAL RESINS
- 8.3 SYNTHETIC & SEMI-SYNTHETIC RESIN
- 8.3.1 RISING DEMAND FOR HIGH-PERFORMANCE AND DURABLE BONDING SOLUTIONS DRIVING MARKET GROWTH
- 8.3.2 ACRYLIC
- 8.3.2.1 Growing adoption of wearable medical devices driving demand
- 8.3.3 SILICONE
- 8.3.3.1 Increasing demand for gentle, skin-friendly adhesion driving adoption
- 8.3.4 CYANOACRYLATE
- 8.3.4.1 Rising preference for sutureless wound closure to support market growth
- 8.3.5 EPOXY
- 8.3.5.1 Demand for high-strength bonding in medical device assembly to drive growth
- 8.3.6 POLYURETHANE
- 8.3.6.1 Need for flexible and durable medical materials fueling demand
- 8.3.7 OTHER SYNTHETIC & SEMI-SYNTHETIC RESINS
9 MEDICAL ADHESIVES MARKET, BY TECHNOLOGY
- 9.1 INTRODUCTION
- 9.2 WATER-BASED
- 9.2.1 RISING DEMAND FOR BIOCOMPATIBLE AND SKIN-FRIENDLY SOLUTIONS DRIVING ADOPTION
- 9.3 SOLVENT-BASED
- 9.3.1 HIGH BOND STRENGTH AND DURABILITY DRIVING DEMAND FOR SOLVENT-BASED MEDICAL ADHESIVES
- 9.4 SOLID & HOT MELT-BASED
- 9.4.1 FAST PROCESSING AND SOLVENT-FREE FORMULATION DRIVING ADOPTION OF HOT MELT MEDICAL ADHESIVES
10 MEDICAL ADHESIVES MARKET, BY APPLICATION
- 10.1 INTRODUCTION
- 10.2 DENTAL
- 10.2.1 RISING DEMAND FOR COSMETIC AND RESTORATIVE DENTISTRY DRIVING MARKET GROWTH
- 10.3 SURGERY
- 10.3.1 GROWING SHIFT TOWARD MINIMALLY INVASIVE PROCEDURES DRIVING ADOPTION
- 10.3.2 INTERNAL
- 10.3.2.1 Increasing complex surgical procedures driving demand for internal medical adhesives
- 10.3.3 EXTERNAL
- 10.3.3.1 Rising preference for sutureless skin closure driving external adhesives demand
- 10.4 MEDICAL DEVICE & EQUIPMENT
- 10.4.1 RAPID GROWTH OF WEARABLE AND MINIATURIZED DEVICES DRIVING DEMAND
- 10.5 OTHER APPLICATIONS
11 MEDICAL ADHESIVES MARKET, BY REGION
- 11.1 INTRODUCTION
- 11.2 NORTH AMERICA
- 11.2.1 US
- 11.2.1.1 Rising surgical volumes and advanced healthcare infrastructure driving demand
- 11.2.2 CANADA
- 11.2.2.1 Growing surgical procedures and aging population fueling demand
- 11.2.3 MEXICO
- 11.2.3.1 Expanding medical device manufacturing and healthcare infrastructure driving market growth
- 11.2.1 US
- 11.3 ASIA PACIFIC
- 11.3.1 CHINA
- 11.3.1.1 Massive healthcare expansion and government support driving demand
- 11.3.2 INDIA
- 11.3.2.1 Expanding healthcare coverage and rising surgical volumes fueling market growth
- 11.3.3 JAPAN
- 11.3.3.1 Rapidly aging population driving demand for advanced medical adhesives
- 11.3.4 SOUTH KOREA
- 11.3.4.1 Presence of advanced medical device industry driving medical adhesives demand
- 11.3.5 REST OF ASIA PACIFIC
- 11.3.1 CHINA
- 11.4 EUROPE
- 11.4.1 GERMANY
- 11.4.1.1 Strong healthcare infrastructure and high surgical volumes driving demand
- 11.4.2 FRANCE
- 11.4.2.1 Advanced healthcare systems and rising surgical procedures fueling market growth
- 11.4.3 UK
- 11.4.3.1 E-commerce sales to drive market expansion
- 11.4.4 ITALY
- 11.4.4.1 Rapidly aging population fueling demand for advanced wound care and surgical adhesives
- 11.4.5 SPAIN
- 11.4.5.1 Growing aging population and chronic disease burden driving demand
- 11.4.6 REST OF EUROPE
- 11.4.1 GERMANY
- 11.5 MIDDLE EAST & AFRICA
- 11.5.1 GCC COUNTRIES
- 11.5.1.1 Saudi Arabia
- 11.5.1.1.1 Vision 2030 healthcare investments accelerating market growth
- 11.5.1.2 Rest of GCC
- 11.5.1.1 Saudi Arabia
- 11.5.2 SOUTH AFRICA
- 11.5.2.1 Rising chronic disease burden and healthcare expansion supporting market growth
- 11.5.3 REST OF MIDDLE EAST & AFRICA
- 11.5.1 GCC COUNTRIES
- 11.6 SOUTH AMERICA
- 11.6.1 BRAZIL
- 11.6.1.1 Expanding public healthcare system driving medical adhesives demand
- 11.6.2 ARGENTINA
- 11.6.2.1 Rising chronic disease burden accelerating demand
- 11.6.3 REST OF SOUTH AMERICA
- 11.6.1 BRAZIL
12 COMPETITIVE LANDSCAPE
- 12.1 OVERVIEW
- 12.2 KEY PLAYERS' STRATEGIES/RIGHT TO WIN
- 12.3 REVENUE ANALYSIS
- 12.4 MARKET SHARE ANALYSIS, 2025
- 12.5 COMPANY VALUATION AND FINANCIAL METRICS
- 12.6 BRAND/PRODUCT COMPARISON ANALYSIS
- 12.7 COMPANY EVALUATION MATRIX: KEY PLAYERS, 2025
- 12.7.1 STARS
- 12.7.2 EMERGING LEADERS
- 12.7.3 PERVASIVE PLAYERS
- 12.7.4 PARTICIPANTS
- 12.7.5 COMPANY FOOTPRINT: KEY PLAYERS, 2024
- 12.7.5.1 Overall company footprint
- 12.7.5.2 Region footprint
- 12.7.5.3 Technology footprint
- 12.7.5.4 Resin type footprint
- 12.7.5.5 Application footprint
- 12.8 COMPANY EVALUATION MATRIX: STARTUPS/SMES, 2025
- 12.8.1 PROGRESSIVE COMPANIES
- 12.8.2 RESPONSIVE COMPANIES
- 12.8.3 DYNAMIC COMPANIES
- 12.8.4 STARTING BLOCKS
- 12.8.5 COMPETITIVE BENCHMARKING: KEY STARTUPS/SMES, 2025
- 12.8.5.1 Detailed list of key startups/SMEs
- 12.8.5.2 Competitive benchmarking of startups/SMEs
- 12.9 COMPETITIVE SCENARIO
- 12.9.1 PRODUCT LAUNCHES
- 12.9.2 DEALS
- 12.9.3 EXPANSIONS
- 12.9.4 OTHERS
13 COMPANY PROFILES
- 13.1 KEY PLAYERS
- 13.1.1 SOLVENTUM (3M)
- 13.1.1.1 Business overview
- 13.1.1.2 Products/Solutions/Services offered
- 13.1.1.3 Recent developments
- 13.1.1.3.1 Product launches
- 13.1.1.3.2 Deals
- 13.1.1.3.3 Others
- 13.1.1.4 MnM view
- 13.1.1.4.1 Key strengths
- 13.1.1.4.2 Strategic choices
- 13.1.1.4.3 Weaknesses and competitive threats
- 13.1.2 HENKEL AG & CO, KGAA
- 13.1.2.1 Business overview
- 13.1.2.2 Products/Solutions/Services offered
- 13.1.2.3 Recent developments
- 13.1.2.3.1 Product launches
- 13.1.2.4 MnM view
- 13.1.2.4.1 Key strengths
- 13.1.2.4.2 Strategic choices
- 13.1.2.4.3 Weaknesses and competitive threats
- 13.1.3 H.B. FULLER COMPANY
- 13.1.3.1 Business overview
- 13.1.3.2 Products/Solutions/Services offered
- 13.1.3.3 Recent developments
- 13.1.3.3.1 Product launches
- 13.1.3.3.2 Deals
- 13.1.3.4 MnM view
- 13.1.3.4.1 Key strengths
- 13.1.3.4.2 Strategic choices
- 13.1.3.4.3 Weaknesses and competitive threats
- 13.1.4 SCAPA HEALTHCARE
- 13.1.4.1 Business overview
- 13.1.4.2 Products/Solutions/Services offered
- 13.1.4.3 Recent developments
- 13.1.4.3.1 Expansions
- 13.1.4.4 MnM view
- 13.1.4.4.1 Key strengths
- 13.1.4.4.2 Strategic choices
- 13.1.4.4.3 Weaknesses and competitive threats
- 13.1.5 JOHNSON & JOHNSON (MEDTECH COMPANY)
- 13.1.5.1 Business overview
- 13.1.5.2 Products/Solutions/Services offered
- 13.1.5.3 MnM view
- 13.1.5.3.1 Key strengths
- 13.1.5.3.2 Strategic choices
- 13.1.5.3.3 Weaknesses and competitive threats
- 13.1.6 PERMABOND
- 13.1.6.1 Business overview
- 13.1.6.2 Products/Solutions/Services offered
- 13.1.6.3 MnM view
- 13.1.7 B. BRAUN SE
- 13.1.7.1 Business overview
- 13.1.7.2 Products/Solutions/Services offered
- 13.1.7.3 MnM view
- 13.1.8 CHEMENCE MEDICAL, INC.
- 13.1.8.1 Business overview
- 13.1.8.2 Products/Solutions/Services offered
- 13.1.8.3 Recent developments
- 13.1.8.3.1 Others
- 13.1.8.4 MnM view
- 13.1.9 ARTIVION, INC.
- 13.1.9.1 Business overview
- 13.1.9.2 Products/Solutions/Services offered
- 13.1.9.3 MnM view
- 13.1.10 DYMAX
- 13.1.10.1 Business overview
- 13.1.10.2 Products/Solutions/Services offered
- 13.1.10.3 MnM view
- 13.1.11 BOSTIK
- 13.1.11.1 Business overview
- 13.1.11.2 Products/Solutions/Services offered
- 13.1.11.3 Recent developments
- 13.1.11.3.1 Deals
- 13.1.11.4 MnM view
- 13.1.1 SOLVENTUM (3M)
- 13.2 OTHER PLAYERS
- 13.2.1 MEDTRONIC
- 13.2.2 DENTSPLY SIRONA
- 13.2.3 MASTERBOND INC.
- 13.2.4 ASHLAND
- 13.2.5 ADVANCED MEDICAL SOLUTIONS GROUP PLC
- 13.2.6 HOENLE AG
- 13.2.7 BECTON, DICKINSON AND COMPANY (BD)
- 13.2.8 VIVOSTAT A/S
- 13.2.9 OCULAR THERAPEUTIX, INC.
- 13.2.10 GLAXOSMITHKLINE PLC
- 13.2.11 NITTO DENKO CORPORATION
- 13.2.12 BAXTER INTERNATIONAL
- 13.2.13 CARTELL CHEMICAL CO., LTD.
- 13.2.14 BIOSEAL INC.
14 RESEARCH METHODOLOGY
- 14.1 RESEARCH DATA
- 14.1.1 SECONDARY DATA
- 14.1.1.1 List of key secondary sources
- 14.1.1.2 Key data from secondary sources
- 14.1.2 PRIMARY DATA
- 14.1.2.1 Key data from primary sources
- 14.1.2.2 List of primary interview participants-demand and supply side
- 14.1.2.3 Key industry insights
- 14.1.2.4 Breakdown of interviews with experts
- 14.1.1 SECONDARY DATA
- 14.2 MARKET SIZE ESTIMATION
- 14.2.1 BOTTOM-UP APPROACH
- 14.2.2 TOP-DOWN APPROACH
- 14.3 FORECAST NUMBER CALCULATION
- 14.4 DATA TRIANGULATION
- 14.5 FACTOR ANALYSIS
- 14.6 ASSUMPTIONS
- 14.7 LIMITATIONS & RISKS
15 APPENDIX
- 15.1 DISCUSSION GUIDE
- 15.2 KNOWLEDGESTORE: MARKETSANDMARKETS' SUBSCRIPTION PORTAL
- 15.3 CUSTOMIZATION OPTIONS
- 15.4 RELATED REPORTS
- 15.5 AUTHOR DETAILS
List of Tables
- TABLE 1 MEDICAL ADHESIVES MARKET: IMPACT OF PORTER'S FIVE FORCES
- TABLE 2 MEDICAL ADHESIVES MARKET: ROLE OF COMPANIES IN ECOSYSTEM
- TABLE 3 PROJECTED REAL GDP GROWTH (ANNUAL PERCENT CHANGE) OF KEY COUNTRIES, 2021-2030
- TABLE 4 HEALTH EXPENDITURE AS PERCENTAGE OF GDP, 2019-2020
- TABLE 5 US-ADJUSTED RECIPROCAL TARIFF RATES
- TABLE 6 IMPORT DATA FOR HS CODE 300590-COMPLIANT PRODUCTS, 2021-2024 (USD THOUSAND)
- TABLE 7 EXPORT DATA FOR HS CODE 300590-COMPLIANT PRODUCTS, 2021-2024 (USD THOUSAND)
- TABLE 8 MEDICAL ADHESIVES MARKET: CONFERENCES & EVENTS, 2026-2027
- TABLE 9 TOP USE CASES AND MARKET POTENTIAL
- TABLE 10 BEST PRACTICES: NOTABLE INDUSTRY PRACTICES BY LEADING COMPANIES
- TABLE 11 MEDICAL ADHESIVES MARKET: CASE STUDIES RELATED TO AI IMPLEMENTATION
- TABLE 12 INTERCONNECTED ADJACENT ECOSYSTEM AND IMPACT ON MARKET PLAYERS
- TABLE 13 MEDICAL ADHESIVES MARKET: LIST OF REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 14 GLOBAL STANDARDS IN MEDICAL ADHESIVES MARKET
- TABLE 15 CERTIFICATIONS, LABELING, AND ECO-STANDARDS IN MEDICAL ADHESIVES MARKET
- TABLE 16 INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS FOR KEY APPLICATIONS (%)
- TABLE 17 KEY BUYING CRITERIA FOR KEY APPLICATIONS
- TABLE 18 MEDICAL ADHESIVES MARKET: UNMET NEEDS IN KEY END-USE INDUSTRIES
- TABLE 19 MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2022-2025 (USD MILLION)
- TABLE 20 MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2026-2031 (USD MILLION)
- TABLE 21 MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2022-2025 (TON)
- TABLE 22 MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2026-2031 (TON)
- TABLE 23 MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2022-2025 (USD MILLION)
- TABLE 24 MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2026-2031 (USD MILLION)
- TABLE 25 MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2022-2025 (TON)
- TABLE 26 MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2026-2031 (TON)
- TABLE 27 MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 28 MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 29 MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 30 MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 31 MEDICAL ADHESIVES MARKET, BY REGION, 2022-2025 (USD MILLION)
- TABLE 32 MEDICAL ADHESIVES MARKET, BY REGION, 2026-2031 (USD MILLION)
- TABLE 33 MEDICAL ADHESIVES MARKET, BY REGION, 2022-2025 (TON)
- TABLE 34 MEDICAL ADHESIVES MARKET, BY REGION, 2026-2031 (TON)
- TABLE 35 NORTH AMERICA: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2022-2025 (USD MILLION)
- TABLE 36 NORTH AMERICA: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2026-2031 (USD MILLION)
- TABLE 37 NORTH AMERICA: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2022-2025 (TON)
- TABLE 38 NORTH AMERICA: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2026-2031 (TON)
- TABLE 39 NORTH AMERICA: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2022-2025 (USD MILLION)
- TABLE 40 NORTH AMERICA: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2026-2031 (USD MILLION)
- TABLE 41 NORTH AMERICA: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2022-2025 (TON)
- TABLE 42 NORTH AMERICA: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2026-2031 (TON)
- TABLE 43 NORTH AMERICA: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2022-2025 (USD MILLION)
- TABLE 44 NORTH AMERICA: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2026-2031 (USD MILLION)
- TABLE 45 NORTH AMERICA: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2022-2025 (TON)
- TABLE 46 NORTH AMERICA: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2026-2031 (TON)
- TABLE 47 NORTH AMERICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 48 NORTH AMERICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 49 NORTH AMERICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 50 NORTH AMERICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 51 US: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 52 US: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 53 US: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 54 US: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 55 CANADA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 56 CANADA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 57 CANADA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 58 CANADA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 59 MEXICO: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 60 MEXICO: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 61 MEXICO: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 62 MEXICO: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 63 ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2022-2025 (USD MILLION)
- TABLE 64 ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2026-2031 (USD MILLION)
- TABLE 65 ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2022-2025 (TON)
- TABLE 66 ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2026-2031 (TON)
- TABLE 67 ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2022-2025 (USD MILLION)
- TABLE 68 ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2026-2031 (USD MILLION)
- TABLE 69 ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2022-2025 (TON)
- TABLE 70 ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2026-2031 (TON)
- TABLE 71 ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2022-2025 (USD MILLION)
- TABLE 72 ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2026-2031 (USD MILLION)
- TABLE 73 ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2022-2025 (TON)
- TABLE 74 ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2026-2031 (TON)
- TABLE 75 ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 76 ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 77 ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 78 ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 79 CHINA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 80 CHINA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 81 CHINA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 82 CHINA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 83 INDIA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 84 INDIA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 85 INDIA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 86 INDIA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 87 JAPAN: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 88 JAPAN: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 89 JAPAN: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 90 JAPAN: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 91 SOUTH KOREA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 92 SOUTH KOREA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 93 SOUTH KOREA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 94 SOUTH KOREA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 95 REST OF ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 96 REST OF ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 97 REST OF ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 98 REST OF ASIA PACIFIC: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 99 EUROPE: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2022-2025 (USD MILLION)
- TABLE 100 EUROPE: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2026-2031 (USD MILLION)
- TABLE 101 EUROPE: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2022-2025 (TON)
- TABLE 102 EUROPE: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2026-2031 (TON)
- TABLE 103 EUROPE: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2022-2025 (USD MILLION)
- TABLE 104 EUROPE: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2026-2031 (USD MILLION)
- TABLE 105 EUROPE: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2022-2025 (TON)
- TABLE 106 EUROPE: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2026-2031 (TON)
- TABLE 107 EUROPE: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2022-2025 (USD MILLION)
- TABLE 108 EUROPE: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2026-2031 (USD MILLION)
- TABLE 109 EUROPE: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2022-2025 (TON)
- TABLE 110 EUROPE: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2026-2031 (TON)
- TABLE 111 EUROPE: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 112 EUROPE: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 113 EUROPE: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 114 EUROPE: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 115 GERMANY: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 116 GERMANY: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 117 GERMANY: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 118 GERMANY: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 119 FRANCE: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 120 FRANCE: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 121 FRANCE: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 122 FRANCE: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 123 UK: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 124 UK: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 125 UK: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 126 UK: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 127 ITALY: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 128 ITALY: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 129 ITALY: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 130 ITALY: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 131 SPAIN: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 132 SPAIN: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 133 SPAIN: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 134 SPAIN: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 135 REST OF EUROPE: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 136 REST OF EUROPE: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 137 REST OF EUROPE: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 138 REST OF EUROPE: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 139 MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2022-2025 (USD MILLION)
- TABLE 140 MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2026-2031 (USD MILLION)
- TABLE 141 MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2022-2025 (TON)
- TABLE 142 MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2026-2031 (TON)
- TABLE 143 MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2022-2025 (USD MILLION)
- TABLE 144 MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2026-2031 (USD MILLION)
- TABLE 145 MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2022-2025 (TON)
- TABLE 146 MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2026-2031 (TON)
- TABLE 147 MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2022-2025 (USD MILLION)
- TABLE 148 MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2026-2031 (USD MILLION)
- TABLE 149 MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2022-2025 (TON)
- TABLE 150 MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2026-2031 (TON)
- TABLE 151 MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 152 MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 153 MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 154 MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 155 SAUDI ARABIA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 156 SAUDI ARABIA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 157 SAUDI ARABIA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 158 SAUDI ARABIA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 159 REST OF GCC: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 160 REST OF GCC: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 161 REST OF GCC: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 162 REST OF GCC: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 163 SOUTH AFRICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 164 SOUTH AFRICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 165 SOUTH AFRICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 166 SOUTH AFRICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 167 REST OF MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 168 REST OF MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 169 REST OF MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 170 REST OF MIDDLE EAST & AFRICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 171 SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2022-2025 (USD MILLION)
- TABLE 172 SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2026-2031 (USD MILLION)
- TABLE 173 SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2022-2025 (TON)
- TABLE 174 SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY COUNTRY, 2026-2031 (TON)
- TABLE 175 SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2022-2025 (USD MILLION)
- TABLE 176 SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2026-2031 (USD MILLION)
- TABLE 177 SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2022-2025 (TON)
- TABLE 178 SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY TECHNOLOGY, 2026-2031 (TON)
- TABLE 179 SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2022-2025 (USD MILLION)
- TABLE 180 SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2026-2031 (USD MILLION)
- TABLE 181 SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2022-2025 (TON)
- TABLE 182 SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY RESIN TYPE, 2026-2031 (TON)
- TABLE 183 SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 184 SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 185 SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 186 SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 187 BRAZIL: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 188 BRAZIL: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 189 BRAZIL: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 190 BRAZIL: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 191 ARGENTINA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 192 ARGENTINA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 193 ARGENTINA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 194 ARGENTINA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 195 REST OF SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (USD MILLION)
- TABLE 196 REST OF SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (USD MILLION)
- TABLE 197 REST OF SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2022-2025 (TON)
- TABLE 198 REST OF SOUTH AMERICA: MEDICAL ADHESIVES MARKET, BY APPLICATION, 2026-2031 (TON)
- TABLE 199 OVERVIEW OF STRATEGIES ADOPTED BY KEY PLAYERS IN MEDICAL ADHESIVES MARKET BETWEEN JANUARY 2020 AND MARCH 2026
- TABLE 200 MEDICAL ADHESIVES MARKET: DEGREE OF COMPETITION
- TABLE 201 MEDICAL ADHESIVES MARKET: REGION FOOTPRINT
- TABLE 202 MEDICAL ADHESIVES MARKET: TECHNOLOGY FOOTPRINT
- TABLE 203 MEDICAL ADHESIVES MARKET: RESIN TYPE FOOTPRINT
- TABLE 204 MEDICAL ADHESIVES MARKET: APPLICATION FOOTPRINT
- TABLE 205 MEDICAL ADHESIVES MARKET: DETAILED LIST OF KEY STARTUPS/SMES
- TABLE 206 MEDICAL ADHESIVES MARKET: COMPETITIVE BENCHMARKING OF STARTUPS/SMES
- TABLE 207 MEDICAL ADHESIVES MARKET: PRODUCT LAUNCHES, JANUARY 2020-MARCH 2026
- TABLE 208 MEDICAL ADHESIVES MARKET: DEALS, JANUARY 2020-MARCH 2026
- TABLE 209 MEDICAL ADHESIVES MARKET: EXPANSIONS, JANUARY 2020-MARCH 2026
- TABLE 210 MEDICAL ADHESIVES MARKET: OTHERS, JANUARY 2020-MARCH 2026
- TABLE 211 SOLVENTUM (3M): COMPANY OVERVIEW
- TABLE 212 SOLVENTUM (3M): PRODUCTS/SOLUTIONS/SERVICES OFFERED
- TABLE 213 SOLVENTUM (3M): PRODUCT LAUNCHES
- TABLE 214 SOLVENTUM (3M): DEALS
- TABLE 215 SOLVENTUM (3M): OTHERS
- TABLE 216 HENKEL AG & CO. KGAA: COMPANY OVERVIEW
- TABLE 217 HENKEL AG & CO. KGAA: PRODUCTS/SOLUTIONS/SERVICES OFFERED
- TABLE 218 HENKEL AG & CO. KGAA: PRODUCT LAUNCHES
- TABLE 219 H.B. FULLER COMPANY: COMPANY OVERVIEW
- TABLE 220 H. B. FULLER COMPANY: PRODUCTS/SOLUTIONS/SERVICES OFFERED
- TABLE 221 H.B. FULLER COMPANY: PRODUCT LAUNCHES
- TABLE 222 H.B. FULLER COMPANY: DEALS
- TABLE 223 SCAPA HEALTHCARE: COMPANY OVERVIEW
- TABLE 224 SCAPA HEALTHCARE: PRODUCTS/SOLUTIONS/SERVICES OFFERED
- TABLE 225 SCAPA HEALTHCARE: EXPANSIONS
- TABLE 226 JOHNSON & JOHNSON (MEDTECH COMPANY): COMPANY OVERVIEW
- TABLE 227 JOHNSON & JOHNSON (MEDTECH COMPANY): PRODUCTS/SOLUTIONS/SERVICES OFFERED
- TABLE 228 PERMABOND: COMPANY OVERVIEW
- TABLE 229 PERMABOND: PRODUCTS/SOLUTIONS/SERVICES OFFERED
- TABLE 230 B. BRAUN SE: COMPANY OVERVIEW
- TABLE 231 B. BRAUN SE: PRODUCTS/SOLUTIONS/SERVICES OFFERED
- TABLE 232 CHEMENCE MEDICAL, INC.: COMPANY OVERVIEW
- TABLE 233 CHEMENCE MEDICAL, INC.: PRODUCTS/SOLUTIONS/SERVICES OFFERED
- TABLE 234 CHEMENCE MEDICAL, INC.: OTHERS
- TABLE 235 ARTIVION, INC.: COMPANY OVERVIEW
- TABLE 236 ARTIVION, INC.: PRODUCTS/SOLUTIONS/SERVICES OFFERED
- TABLE 237 DYMAX: COMPANY OVERVIEW
- TABLE 238 DYMAX: PRODUCTS/SOLUTIONS/SERVICES OFFERED
- TABLE 239 BOSTIK: COMPANY OVERVIEW
- TABLE 240 BOSTIK: PRODUCTS/SOLUTIONS/SERVICES OFFERED
- TABLE 241 BOSTIK: DEALS
- TABLE 242 MEDTRONIC: COMPANY OVERVIEW
- TABLE 243 DENTSPLY SIRONA: COMPANY OVERVIEW
- TABLE 244 MASTERBOND INC.: COMPANY OVERVIEW
- TABLE 245 ASHLAND: COMPANY OVERVIEW
- TABLE 246 ADVANCED MEDICAL SOLUTIONS GROUP PLC: COMPANY OVERVIEW
- TABLE 247 HOENLE AG: COMPANY OVERVIEW
- TABLE 248 BECTON, DICKINSON AND COMPANY (BD): COMPANY OVERVIEW
- TABLE 249 VIVOSTAT A/S: COMPANY OVERVIEW
- TABLE 250 OCULAR THERAPEUTIX, INC.: COMPANY OVERVIEW
- TABLE 251 GLAXOSMITHKLINE PLC: COMPANY OVERVIEW
- TABLE 252 NITTO DENKO CORPORATION: COMPANY OVERVIEW
- TABLE 253 BAXTER INTERNATIONAL: COMPANY OVERVIEW
- TABLE 254 CARTELL CHEMICAL CO., LTD.: COMPANY OVERVIEW
- TABLE 255 BIOSEAL INC.: COMPANY OVERVIEW
List of Figures
- FIGURE 1 MEDICAL ADHESIVES MARKET SEGMENTATION AND REGIONAL SCOPE
- FIGURE 2 KEY INSIGHTS AND MARKET HIGHLIGHTS
- FIGURE 3 MEDICAL ADHESIVES MARKET, 2026-2031
- FIGURE 4 MAJOR STRATEGIES ADOPTED BY KEY PLAYERS IN MEDICAL ADHESIVES MARKET, 2020-2025
- FIGURE 5 DISRUPTIONS INFLUENCING GROWTH OF MEDICAL ADHESIVES MARKET
- FIGURE 6 HIGH-GROWTH SEGMENTS IN MEDICAL ADHESIVES MARKET, 2026-2031
- FIGURE 7 ASIA PACIFIC TO REGISTER HIGHEST CAGR IN MEDICAL ADHESIVES MARKET DURING FORECAST PERIOD
- FIGURE 8 ASIA PACIFIC TO OFFER LUCRATIVE OPPORTUNITIES IN MEDICAL ADHESIVES MARKET DURING FORECAST PERIOD
- FIGURE 9 WATER-BASED ACCOUNTED FOR LARGEST MARKET SHARE IN 2025
- FIGURE 10 SYNTHETIC & SEMI-SYNTHETIC RESIN SEGMENT DOMINATED MEDICAL ADHESIVES MARKET IN 2025
- FIGURE 11 ELECTRICAL & ELECTRONICS APPLICATION ACCOUNTED FOR LARGEST MARKET SHARE IN 2025
- FIGURE 12 INDIA TO REGISTER HIGHEST CAGR DURING FORECAST PERIOD
- FIGURE 13 DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES IN MEDICAL ADHESIVES MARKET
- FIGURE 14 MEDICAL ADHESIVES MARKET: PORTER'S FIVE FORCES ANALYSIS
- FIGURE 15 MEDICAL ADHESIVES MARKET: VALUE CHAIN ANALYSIS
- FIGURE 16 MEDICAL ADHESIVES MARKET: ECOSYSTEM ANALYSIS
- FIGURE 17 AVERAGE SELLING PRICE TREND, BY REGION, 2022-2025
- FIGURE 18 AVERAGE SELLING PRICE TREND, BY TECHNOLOGY, 2022-2025
- FIGURE 19 AVERAGE SELLING PRICE TREND, BY APPLICATION, 2022-2025
- FIGURE 20 IMPORT SCENARIO FOR HS CODE 300590-COMPLIANT PRODUCTS, BY COUNTRY, 2021-2024
- FIGURE 21 EXPORT SCENARIO FOR HS CODE 300590-COMPLIANT PRODUCTS, BY COUNTRY, 2021-2024
- FIGURE 22 DISRUPTIONS/TRENDS SHAPING MEDICAL ADHESIVES MARKET
- FIGURE 23 MEDICAL ADHESIVES MARKET: INVESTMENT AND FUNDING SCENARIO, 2023-2025
- FIGURE 24 PATENTS APPLIED AND GRANTED, 2015-2025
- FIGURE 25 PATENT ANALYSIS, BY LEGAL STATUS
- FIGURE 26 TOP JURISDICTION, BY DOCUMENT
- FIGURE 27 MEDICAL ADHESIVES MARKET: DECISION-MAKING FACTORS
- FIGURE 28 INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS FOR KEY APPLICATIONS
- FIGURE 29 KEY BUYING CRITERIA FOR TOP THREE APPLICATIONS
- FIGURE 30 ADOPTION BARRIERS & INTERNAL CHALLENGES
- FIGURE 31 SYNTHETIC & SEMI-SYNTHETIC RESIN SEGMENT TO ACCOUNT FOR LARGER MARKET SHARE DURING FORECAST PERIOD
- FIGURE 32 SOLIDS & HOT MELT-BASED SEGMENT TO ACCOUNT FOR LARGEST MARKET SHARE DURING FORECAST PERIOD
- FIGURE 33 SURGERY TO ACCOUNT FOR LARGEST MARKET SHARE DURING FORECAST PERIOD
- FIGURE 34 INDIA TO REGISTER HIGHEST CAGR IN MEDICAL ADHESIVES MARKET DURING FORECAST PERIOD
- FIGURE 35 NORTH AMERICA: MEDICAL ADHESIVES MARKET SNAPSHOT
- FIGURE 36 ASIA PACIFIC: MEDICAL ADHESIVES MARKET SNAPSHOT
- FIGURE 37 EUROPE: MEDICAL ADHESIVES MARKET SNAPSHOT
- FIGURE 38 REVENUE ANALYSIS OF KEY COMPANIES IN MEDICAL ADHESIVES MARKET, 2023-2025
- FIGURE 39 SHARES OF LEADING COMPANIES IN MEDICAL ADHESIVES MARKET, 2025
- FIGURE 40 COMPANY VALUATION OF LEADING COMPANIES IN MEDICAL ADHESIVES MARKET, 2025
- FIGURE 41 EV/EBITDA OF LEADING COMPANIES IN MEDICAL ADHESIVES MARKET, 2024
- FIGURE 42 MEDICAL ADHESIVES MARKET: BRAND/PRODUCT COMPARISON
- FIGURE 43 MEDICAL ADHESIVES MARKET: COMPANY EVALUATION MATRIX (KEY PLAYERS), 2025
- FIGURE 44 MEDICAL ADHESIVES MARKET: OVERALL COMPANY FOOTPRINT
- FIGURE 45 MEDICAL ADHESIVES MARKET: COMPANY EVALUATION MATRIX (STARTUPS/SMES), 2025
- FIGURE 46 SOLVENTUM (3M): COMPANY SNAPSHOT
- FIGURE 47 HENKEL AG & CO. KGAA: COMPANY SNAPSHOT
- FIGURE 48 H. B. FULLER COMPANY: COMPANY SNAPSHOT
- FIGURE 49 JOHNSON & JOHNSON (MEDTECH COMPANY): COMPANY SNAPSHOT
- FIGURE 50 ARTIVION, INC.: COMPANY SNAPSHOT
- FIGURE 51 MEDICAL ADHESIVES MARKET: RESEARCH DESIGN
- FIGURE 52 MEDICAL ADHESIVES MARKET: BOTTOM-UP APPROACH
- FIGURE 53 MEDICAL ADHESIVES MARKET: TOP-DOWN APPROACH - 1
- FIGURE 54 MEDICAL ADHESIVES MARKET: TOP-DOWN APPROACH - 2
- FIGURE 55 MEDICAL ADHESIVES MARKET: DEMAND-SIDE FORECAST
- FIGURE 56 MEDICAL ADHESIVES MARKET: DATA TRIANGULATION




