PUBLISHER: MarketsandMarkets | PRODUCT CODE: 2029868

PUBLISHER: MarketsandMarkets | PRODUCT CODE: 2029868
Digital Biomarkers Market by Type (Physiological, Vocal, Idiosyncratic), Therapy, Application, End User (Pharma & Biotech Companies, CROs) - Global Forecast to 2031
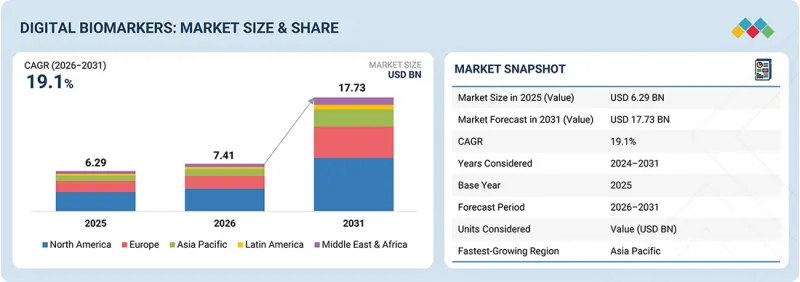
The global digital biomarkers market is projected to reach USD 17.73 billion by 2031, growing from USD 7.41 billion in 2026 at a CAGR of 19.1%. The digital biomarkers market is being driven by the rapid evolution of data-driven clinical trials and the growing trend of integrating digital health technology into clinical trials.
| Scope of the Report | |
|---|---|
| Years Considered for the Study | 2026-2031 |
| Base Year | 2025 |
| Forecast Period | 2026-2031 |
| Units Considered | Value (USD billion) |
| Segments | Type, Therapeutic Area, Application, and End User |
| Regions covered | North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa |
Pharmaceutical and biotech companies are increasingly using digital biomarkers in various stages of clinical trials to enable continuous and objective patient monitoring, thus improving data quality and endpoint accuracy. This is a part of a larger trend towards more efficient and patient-centric clinical trials, in which data from real-world sources plays a crucial role in determining the effectiveness of a drug. At the same time, there is a growing trend toward more decentralized clinical trials, in which data is collected more frequently by using remote data collection technology such as wearable devices and sensor technology. This is a part of a larger trend towards more digital biomarker software technology in clinical trials.
"By type, the physiological biomarkers segment is expected to account for the largest market share of the digital biomarkers market during the forecast period."
By type, the physiological biomarkers segment is expected to hold the largest share of the digital biomarkers market during the forecast period. This is mainly due to the widespread use of continuous physiological measures like heart rate, ECG, respiratory rate, and activity levels in various clinical research and patient care settings. Physiological biomarkers are scalable and are well validated, making them more suitable for remote patient monitoring and chronic disease management. In addition, the growing trend of using software platforms and AI-based tools associated with wearable devices is further propelling their adoption, particularly in cardiovascular and metabolic conditions where continuous monitoring is critical in making clinical decisions. This is also evident from published research that states that physiological data streams like heart rate and activity levels are some of the most commonly used digital endpoints in clinical trials, mainly due to their objectivity and ability to provide high-frequency and real-world data. In addition, the move towards decentralized trials and RWE generation is also further solidifying their position in the field of digital biomarkers, as they allow for patient monitoring outside traditional clinical environments.
In June 2025, AliveCor, Inc. (US) partnered with the British Heart Foundation (UK), donating proceeds from its KardiaMobile 6L device to support cardiovascular research and promote the adoption of digital heart rhythm monitoring technologies. These examples further hint at the growing role of physiological digital biomarkers in advancing cardiac care and research.
"By end user, the hospitals & specialty clinics segment is expected to achieve the fastest growth during the forecast period."
By end user, the hospitals & specialty clinics segment is expected to witness the highest growth in the digital biomarkers market over the forecast period. The growth in this segment can be attributed to the rising adoption of digital biomarkers in hospitals and specialty clinics for real-time patient monitoring. Digital biomarkers are being increasingly integrated into the workflows of hospitals and specialty clinics for data-driven decision-making. Hospitals and specialty clinics are adopting wearable-enabled platforms and artificial intelligence-based analytics solutions for continuously monitoring physiological, cognitive, and behavioral changes in patients. Digital biomarkers help hospitals and specialty clinics in continuously monitoring patient data in real-time, thus helping in the early detection of changes in patient health. Moreover, the rising trend of value-based care is fueling the adoption of remote monitoring solutions in hospitals and specialty clinics. The rising prevalence of chronic diseases such as cardiovascular diseases, neurological disorders, and diabetes is also contributing to the growth of the digital biomarkers market in hospitals and specialty clinics. The rising trend of health information technology, which allows for seamless integration of digital biomarker solutions into electronic health records, is also fueling the growth of the digital biomarkers market in hospitals and specialty clinics.
"Asia Pacific to witness the highest growth rate during the forecast period."
The digital biomarkers market in Asia Pacific is growing rapidly, with the region witnessing high growth in the expansion of clinical research activities. The region is witnessing the incorporation of digital technologies into the healthcare infrastructure. Countries such as India, South Korea, Japan, and Australia are gaining importance as hubs for clinical research activities. Due to the improvement in the regulatory environment and the cost advantage, the pharmaceutical and biotech industries are increasingly using the Asia Pacific as a hub for conducting clinical research activities. As the pharmaceutical and biotech industries increasingly rely on digital technologies to conduct clinical research activities, the digital biomarkers market is growing rapidly.
Asia Pacific is witnessing the incorporation of digital technologies into the healthcare infrastructure. The region is witnessing the expansion of clinical research activities. In November 2025, Lunit Inc., a South Korean-based company, announced its collaboration with Labcorp to conduct research using digital pathology technology powered by artificial intelligence and expand access to imaging-based biomarkers for clinical and translational oncology research globally. Such developments indicate the potential of the region as a lucrative market for digital biomarkers.
The breakdown of primary participants is as follows:
- By Company Type - Tier 1 Companies: 60%, Tier 2 Companies: 30%, and Tier 3 Companies: 10%
- By Designation - C-level Executives: 30%, Director-level Personnel: 50%, and Other Designations: 20%
- By Region - North America: 45%, Europe: 20%, Asia Pacific: 25%, Rest of the world: 10%
IXICO PLC (UK), Ametris, LLC (US), and Empatica, Inc. (US) are some of the key players in the digital biomarkers market. The study includes an in-depth competitive analysis of these key players, their company profiles, recent developments, and key market strategies.
Research Coverage:
This research report categorizes the digital biomarkers market by type (physiological biomarkers, idiosyncratic biomarkers, cognitive biomarkers, vocal biomarkers, and other biomarkers), therapeutic area {cardiovascular [atherosclerotic cardiovascular disease (ASCVD)/secondary prevention, heart failure (HFrEF and HFpEF), hypertension, atrial fibrillation (AF)/stroke prevention, pulmonary hypertension, and structural heart disease/interventional cardiology], oncology (solid tumors, hematologic malignancies), diabetes, mental health & behavioral health, respiratory disorders, lifestyle & wellness improvement, neurology, musculoskeletal disorders/pain management, women's health & reproductive health, and other diseases}, application, [clinical research applications (phase II, phase III, phase IV) and clinical care applications], end user [pharmaceutical and biotechnology companies, contract research organizations (CROs), hospitals and specialty clinics, and others (including research institutes, etc.)] and region (North America, Europe, Asia Pacific, Latin America, and Middle East & Africa).
The scope of the report covers detailed information regarding the major factors, such as drivers, restraints, challenges, and opportunities, influencing the growth of the digital biomarkers market. A detailed analysis of the key industry players has been done to provide insights into their business overview; solutions and services; contracts, partnerships, agreements, product & service launches, mergers & acquisitions; and recent developments associated with the digital biomarkers market. Competitive analysis of upcoming startups in the digital biomarkers market ecosystem is covered in this report.
Reasons to Buy this Report:
The report will help the market leaders/new entrants in this market with information on the closest approximations of the revenue numbers for the digital biomarkers market and the subsegments. This report will help stakeholders understand the competitive landscape and gain more insights to position their businesses better and to plan suitable go-to-market strategies. The report also helps stakeholders understand the pulse of the market and provides them with information on key market drivers, restraints, challenges, and opportunities.
The report provides insights into the following pointers:
- Analysis of key drivers (Expansion of smartphones, wearables, and connected health ecosystems; expansion of decentralized and remote clinical trials; rising prevalence of chronic diseases requiring continuous monitoring; advancements in AI and digital health analytics) restraints (Data privacy and security concerns related to health data, limited digital literacy and technology access among patients) opportunities (Increasing use of digital biomarkers in drug development and clinical trial endpoints, growth of precision medicine and personalized healthcare, emerging applications in neurological and mental health monitoring), challenges (Lack of standardized validation frameworks for digital biomarkers, regulatory uncertainty around digital endpoints and biomarker validation) influencing the growth of the digital biomarkers market
- Product Development/Innovation: Detailed insights on upcoming technologies, research & development activities, and product & service launches in the digital biomarkers market
- Market Development: Comprehensive information about lucrative markets across varied regions
- Market Diversification: Exhaustive information about new products & services, untapped geographies, recent developments, and investments in the digital biomarkers market
- Competitive Assessment: In-depth assessment of market share, growth strategies, and service offerings of leading players, namely IXICO PLC (UK), Ametris, LLC (US), Empatica, Inc. (US), AliveCor, Inc. (US), CONNEQT Health (US), VivoSense (US), BioSensics (US), Lunit, Inc. (South Korea), among others, in the digital biomarkers market
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 STUDY OBJECTIVES
- 1.2 MARKET DEFINITION
- 1.3 MARKET SCOPE
- 1.3.1 MARKET SEGMENTATION & REGIONAL SCOPE
- 1.3.2 INCLUSIONS & EXCLUSIONS
- 1.3.3 YEARS CONSIDERED
- 1.4 CURRENCY CONSIDERED
- 1.5 LIMITATIONS
- 1.6 STAKEHOLDERS
2 EXECUTIVE SUMMARY
- 2.1 KEY INSIGHTS AND MARKET HIGHLIGHTS
- 2.2 KEY MARKET PARTICIPANTS: SHARE INSIGHTS AND STRATEGIC DEVELOPMENTS
- 2.3 DISRUPTIVE TRENDS SHAPING THE MARKET
- 2.4 HIGH-GROWTH SEGMENTS & EMERGING FRONTIERS
- 2.5 SNAPSHOT: GLOBAL MARKET SIZE, GROWTH RATE, AND FORECAST
3 PREMIUM INSIGHTS
- 3.1 DIGITAL BIOMARKERS MARKET OVERVIEW
- 3.2 DIGITAL BIOMARKERS MARKET, BY APPLICATION & REGION
- 3.3 DIGITAL BIOMARKERS MARKET: GEOGRAPHIC SNAPSHOT
4 MARKET OVERVIEW
- 4.1 INTRODUCTION
- 4.2 MARKET DYNAMICS
- 4.2.1 DRIVERS
- 4.2.1.1 Expansion of smartphones, wearables, and connected health ecosystems
- 4.2.1.2 Expansion of decentralized and remote clinical trials
- 4.2.1.3 Rising prevalence of chronic diseases that require continuous monitoring
- 4.2.1.4 Advancements in AI and digital health analytics
- 4.2.2 RESTRAINTS
- 4.2.2.1 Data privacy and cybersecurity concerns
- 4.2.2.2 Limited digital literacy and technology access among patients
- 4.2.3 OPPORTUNITIES
- 4.2.3.1 Increasing use in drug development and clinical trial endpoints
- 4.2.3.2 Growth of precision medicine and personalized healthcare
- 4.2.3.3 Emerging applications in neurological and mental health monitoring
- 4.2.4 CHALLENGES
- 4.2.4.1 Lack of standardized validation frameworks for digital biomarkers
- 4.2.4.2 Regulatory uncertainty around digital endpoints and biomarker validation
- 4.2.1 DRIVERS
- 4.3 UNMET NEEDS AND WHITE SPACES
- 4.4 INTERCONNECTED MARKETS AND CROSS-SECTOR OPPORTUNITIES
- 4.5 STRATEGIC MOVES BY TIER-1/2/3 PLAYERS
5 INDUSTRY TRENDS
- 5.1 PORTER'S FIVE FORCES ANALYSIS
- 5.1.1 BARGAINING POWER OF SUPPLIERS (MODERATE)
- 5.1.2 BARGAINING POWER OF BUYERS (HIGH)
- 5.1.3 THREAT OF SUBSTITUTES (MODERATE TO HIGH)
- 5.1.4 THREAT OF NEW ENTRANTS (MODERATE)
- 5.1.5 INTENSITY OF COMPETITIVE RIVALRY (HIGH)
- 5.2 MACROECONOMIC INDICATORS
- 5.2.1 INTRODUCTION
- 5.2.2 GDP TRENDS AND FORECAST
- 5.2.3 TRENDS IN GLOBAL HEALTHCARE IT INDUSTRY
- 5.3 SUPPLY CHAIN ANALYSIS
- 5.4 ECOSYSTEM ANALYSIS
- 5.5 PRICING ANALYSIS
- 5.5.1 INDICATIVE PRICE FOR DIGITAL BIOMARKERS, BY KEY PLAYER (2025)
- 5.5.2 INDICATIVE PRICE FOR DIGITAL BIOMARKERS, BY REGION (2025)
- 5.6 KEY CONFERENCES AND EVENTS, 2026-2027
- 5.7 TRENDS/DISRUPTIONS IMPACTING CUSTOMER BUSINESS
- 5.8 INVESTMENT AND FUNDING SCENARIO
- 5.9 CASE STUDY ANALYSIS
- 5.10 IMPACT OF 2025 US TARIFF ON DIGITAL BIOMARKERS MARKET
- 5.10.1 INTRODUCTION
- 5.10.2 KEY TARIFF RATES
- 5.10.3 PRICE IMPACT ANALYSIS
- 5.10.4 IMPACT ON COUNTRY/REGION
- 5.10.4.1 US
- 5.10.4.2 Europe
- 5.10.4.3 Asia Pacific
- 5.10.5 IMPACT ON END USERS
- 5.10.5.1 Pharmaceutical and biotechnology companies
- 5.10.5.2 Contract research organizations
- 5.10.5.3 Hospitals and specialty clinics
- 5.10.5.4 Others (research institutes, etc.)
6 CUSTOMER LANDSCAPE & BUYER BEHAVIOR
- 6.1 INTRODUCTION
- 6.2 DECISION-MAKING PROCESS
- 6.3 KEY STAKEHOLDERS AND BUYING EVALUATION CRITERIA
- 6.3.1 KEY STAKEHOLDERS IN BUYING PROCESS
- 6.3.2 BUYING CRITERIA
- 6.4 ADOPTION BARRIERS & INTERNAL CHALLENGES
- 6.5 UNMET NEEDS FROM VARIOUS END-USE INDUSTRIES
- 6.5.1 UNMET NEEDS
- 6.5.2 END USER EXPECTATIONS
- 6.6 MARKET PROFITABILITY
7 REGULATORY LANDSCAPE
- 7.1 REGIONAL REGULATIONS AND COMPLIANCE
- 7.1.1 REGULATORY BODIES, GOVERNMENT AGENCIES, & OTHER ORGANIZATIONS
- 7.1.2 REGULATORY FRAMEWORK
- 7.1.2.1 North America
- 7.1.2.2 Europe
- 7.1.2.3 Asia Pacific
- 7.1.2.4 Latin America
- 7.1.2.5 Middle East & Africa
- 7.1.3 INDUSTRY STANDARDS
8 STRATEGIC DISRUPTION THROUGH TECHNOLOGY, PATENTS, AND AI ADOPTION
- 8.1 KEY EMERGING TECHNOLOGIES
- 8.1.1 WEARABLE SENSOR TECHNOLOGIES
- 8.1.2 ARTIFICIAL INTELLIGENCE AND MACHINE LEARNING ANALYTICS
- 8.1.3 MOBILE HEALTH (MHEALTH) PLATFORMS AND SMARTPHONE SENSORS
- 8.2 COMPLEMENTARY TECHNOLOGIES
- 8.2.1 INTERNET OF MEDICAL THINGS (IOMT)
- 8.2.2 TELEHEALTH AND REMOTE PATIENT MONITORING
- 8.3 ADJACENT TECHNOLOGIES
- 8.3.1 DIGITAL THERAPEUTICS
- 8.3.2 DIGITAL PATHOLOGY AND IMAGING AI
- 8.4 TECHNOLOGY/PRODUCT ROADMAP
- 8.5 PATENT ANALYSIS
- 8.5.1 PATENT PUBLICATION TRENDS IN DIGITAL BIOMARKERS MARKET
- 8.5.2 INSIGHTS: JURISDICTION AND TOP APPLICANT ANALYSIS
- 8.6 FUTURE APPLICATIONS
- 8.6.1 AI-ENABLED INTELLIGENT SCREENING AND INTERPRETATION
- 8.6.2 CONTINUOUS AND PREDICTIVE HEALTH MONITORING APPLICATIONS
- 8.6.3 DECENTRALIZED, REMOTE, AND REAL-WORLD DATA ECOSYSTEMS
- 8.7 IMPACT OF AI/GEN AI ON DIGITAL BIOMARKERS MARKET
- 8.7.1 INTRODUCTION
- 8.7.2 MARKET POTENTIAL OF AI/GEN AI IN DIGITAL BIOMARKERS MARKET
- 8.7.3 CASE STUDIES RELATED TO AI/GEN AI IMPLEMENTATION
- 8.7.3.1 Early Alzheimer's detection using mobile-based cognitive assessment platforms
- 8.7.4 IMPACT OF AI/GEN AI ON INTERCONNECTED AND ADJACENT ECOSYSTEMS
- 8.7.4.1 Digital health market
- 8.7.4.2 Remote patient monitoring market
- 8.7.4.3 Telehealth & telemedicine market
- 8.7.5 USER READINESS AND IMPACT ASSESSMENT
- 8.7.5.1 User readiness
- 8.7.5.1.1 User A: Pharmaceutical & biotechnology companies
- 8.7.5.1.2 User B: Contract research organizations (CROs)
- 8.7.5.2 Impact assessment
- 8.7.5.2.1 User A: Pharmaceutical & biotechnology companies
- 8.7.5.2.1.1 Implementation
- 8.7.5.2.1.2 Impact
- 8.7.5.2.2 User B: Contract research organizations
- 8.7.5.2.2.1 Implementation
- 8.7.5.2.2.2 Impact
- 8.7.5.2.1 User A: Pharmaceutical & biotechnology companies
- 8.7.5.1 User readiness
9 DIGITAL BIOMARKERS MARKET, BY TYPE
- 9.1 INTRODUCTION
- 9.2 PHYSIOLOGICAL BIOMARKERS
- 9.2.1 SEGMENT LEADS MARKET ADOPTION THROUGH CLINICAL VALIDATION AND AI-DRIVEN ANALYTICS
- 9.3 IDIOSYNCRATIC BIOMARKERS
- 9.3.1 PRECISION MEDICINE DRIVEN THROUGH PERSONALIZED, INDIVIDUAL-LEVEL DATA INSIGHTS
- 9.4 COGNITIVE BIOMARKERS
- 9.4.1 ENABLE SCALABLE AND OBJECTIVE MEASUREMENT OF NEUROLOGICAL FUNCTION THROUGH ADVANCED ANALYTICS
- 9.5 VOCAL BIOMARKERS
- 9.5.1 AI-POWERED VOICE ANALYSIS ENABLES SCALABLE MENTAL HEALTH MONITORING
- 9.6 OTHER BIOMARKERS
10 DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA
- 10.1 INTRODUCTION
- 10.2 CARDIOVASCULAR
- 10.2.1 ATHEROSCLEROTIC CARDIOVASCULAR DISEASE/SECONDARY PREVENTION
- 10.2.1.1 Continuous risk monitoring and predictive insights in cardiovascular disease management - key driver
- 10.2.2 HEART FAILURE (HFREF & HFPEF)
- 10.2.2.1 Demand driven by advanced early detection and remote monitoring in heart failure management
- 10.2.3 HYPERTENSION
- 10.2.3.1 Transforming blood pressure assessment from episodic to continuous monitoring
- 10.2.4 ATRIAL FIBRILLATION/STROKE PREVENTION
- 10.2.4.1 Unlocking subclinical AF identification via long-duration rhythm analytics
- 10.2.5 PULMONARY HYPERTENSION
- 10.2.5.1 Advancement of functional endpoint measurement beyond traditional walk tests to boost growth
- 10.2.6 STRUCTURAL HEART DISEASE/INTERVENTIONAL CARDIOLOGY
- 10.2.6.1 Remote post-procedural surveillance in structural heart interventions to drive segment
- 10.2.1 ATHEROSCLEROTIC CARDIOVASCULAR DISEASE/SECONDARY PREVENTION
- 10.3 ONCOLOGY
- 10.3.1 SOLID TUMORS
- 10.3.1.1 BREAST CANCER
- 10.3.1.1.1 Segment driven by enhanced tumor characterization through AI-driven imaging and pathology analytics
- 10.3.1.2 LUNG CANCER
- 10.3.1.2.1 Enhanced immunotherapy response prediction using imaging biomarkers to boost growth
- 10.3.1.3 Prostate cancer
- 10.3.1.3.1 Segment growth propelled by reduced variability in cancer grading with quantitative digital pathology
- 10.3.1.4 COLORECTAL CANCER
- 10.3.1.4.1 Enhanced gastrointestinal oncology trials with decentralized functional endpoints - major driver
- 10.3.1.5 BRAIN TUMOR
- 10.3.1.5.1 Enhanced tumor monitoring with advanced MRI and volumetric analytics
- 10.3.1.6 OTHER SOLID TUMORS
- 10.3.1.6.1 Extension of digital biomarker utility across rare and heterogeneous tumor types to drive segment
- 10.3.1.1 BREAST CANCER
- 10.3.2 HEMATOLOGIC MALIGNANCIES
- 10.3.2.1 LEUKEMIA
- 10.3.2.1.1 Enhanced MRD detection and quantification through AI-driven analytics to boost market
- 10.3.2.2 LYMPHOMA
- 10.3.2.2.1 Capturing immune response dynamics with continuous digital monitoring - key benefit
- 10.3.2.3 MULTIPLE MYELOMA
- 10.3.2.3.1 Segment growth driven by enhanced disease burden assessment with integrated imaging and pathology analytics
- 10.3.2.4 OTHER HEMATOLOGIC MALIGNANCIES
- 10.3.2.4.1 High-sensitivity endpoint generation in small patient populations to boost market
- 10.3.2.1 LEUKEMIA
- 10.3.1 SOLID TUMORS
- 10.4 DIABETES
- 10.4.1 CONTINUOUS MONITORING PLATFORMS EXPANDING INTO HOLISTIC CARDIOMETABOLIC RISK PREDICTION
- 10.5 MENTAL HEALTH & BEHAVIORAL HEALTH
- 10.5.1 PASSIVE BEHAVIORAL AND VOICE DATA ENABLE SCALABLE OBJECTIVE CONDITION MONITORING
- 10.6 RESPIRATORY DISORDERS
- 10.6.1 EARLY EXACERBATION DETECTION THROUGH CONTINUOUS MONITORING REDUCES HOSPITALIZATIONS AND COSTS
- 10.7 LIFESTYLE & WELLNESS IMPROVEMENT
- 10.7.1 EXPANDING PREVENTIVE HEALTH THROUGH CONTINUOUS BEHAVIORAL DATA INSIGHTS
- 10.8 NEUROLOGY
- 10.8.1 REGULATORY VALIDATION OF DIGITAL ENDPOINTS TO ACCELERATE EARLY DETECTION AND TRIAL ADOPTION
- 10.9 MUSCULOSKELETAL DISORDERS/PAIN MANAGEMENT
- 10.9.1 EMPLOYER-BACKED DIGITAL CARE AND MOTION TRACKING TO DRIVE SCALABLE REHABILITATION SOLUTIONS
- 10.10 WOMEN'S HEALTH & REPRODUCTIVE HEALTH
- 10.10.1 ADDRESSING UNMET NEEDS THROUGH DATA-DRIVEN WOMEN'S HEALTH INSIGHTS
- 10.11 OTHER DISEASES
- 10.11.1 EXPANDING USE CASES TO DRIVE ADOPTION ACROSS CHRONIC AND UNDERSERVED CONDITIONS
11 DIGITAL BIOMARKERS MARKET, BY APPLICATION
- 11.1 INTRODUCTION
- 11.2 CLINICAL RESEARCH
- 11.2.1 PHASE II
- 11.2.1.1 Critical role in early-stage validation and risk reduction in clinical development
- 11.2.2 PHASE III
- 11.2.2.1 Regulatory-grade endpoint validation drives high-stakes clinical trial adoption
- 11.2.3 PHASE IV
- 11.2.3.1 Real-world evidence demand drives scalable continuous monitoring and reimbursement adoption
- 11.2.1 PHASE II
- 11.3 CLINICAL CARE
- 11.3.1 RISING CHRONIC DISEASE BURDEN DRIVING SHIFT TO CONTINUOUS PROACTIVE CARE
12 DIGITAL BIOMARKER MARKET, BY END USER
- 12.1 INTRODUCTION
- 12.2 PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES
- 12.2.1 CLINICAL TRIAL DRIVEN EFFICIENCY THROUGH HIGH-IMPACT DIGITAL ENDPOINTS
- 12.3 CONTRACT RESEARCH ORGANIZATIONS
- 12.3.1 END-TO-END DIGITAL TRIAL EXECUTION ENABLED THROUGH ADVANCED ANALYTICS
- 12.4 HOSPITALS & SPECIALTY CLINICS
- 12.4.1 ENHANCED CLINICAL OUTCOMES WITH REAL-TIME, DATA-DRIVEN CARE DELIVERY
- 12.5 OTHER END USERS
- 12.5.1 PUBLIC FUNDING AND REAL-WORLD DATA INITIATIVES ACCELERATE ECOSYSTEM-WIDE ADOPTION
13 DIGITAL BIOMARKERS MARKET, BY REGION
- 13.1 INTRODUCTION
- 13.2 NORTH AMERICA
- 13.2.1 MACROECONOMIC OUTLOOK FOR NORTH AMERICA
- 13.2.2 US
- 13.2.2.1 Regulatory advancements and pharma-tech convergence drive market growth
- 13.2.3 CANADA
- 13.2.3.1 Increasing integration of AI-driven screening and remote monitoring to drive market
- 13.3 EUROPE
- 13.3.1 MACROECONOMIC OUTLOOK FOR EUROPE
- 13.3.2 GERMANY
- 13.3.2.1 Strong clinical trial ecosystem and progressive digital health regulation to drive market
- 13.3.3 FRANCE
- 13.3.3.1 Strong clinical trial base and evidence-driven regulatory pathways to drive market
- 13.3.4 UK
- 13.3.4.1 Strong NHS data infrastructure and push for data-enabled clinical trials to drive market
- 13.3.5 ITALY
- 13.3.5.1 Strong clinical research activity and oncology-driven demand to shape market growth
- 13.3.6 SPAIN
- 13.3.6.1 Strong clinical trial leadership and high domestic research concentration to drive market
- 13.3.7 REST OF EUROPE
- 13.4 LATIN AMERICA
- 13.4.1 MACROECONOMIC OUTLOOK FOR LATIN AMERICA
- 13.4.2 BRAZIL
- 13.4.2.1 Expanding digital health ecosystem and regulatory reforms to drive digital biomarkers adoption
- 13.4.3 MEXICO
- 13.4.3.1 Growing clinical trial participation and digital transformation to drive digital biomarkers adoption
- 13.4.4 REST OF LATIN AMERICA
- 13.5 ASIA PACIFIC
- 13.5.1 ASIA PACIFIC: CLINICAL TRIAL INITIATIONS IN 2014 AND 2024
- 13.5.2 MACROECONOMIC OUTLOOK FOR ASIA PACIFIC
- 13.5.3 CHINA
- 13.5.3.1 Expanding clinical trial leadership and advancing digital health ecosystem to drive market
- 13.5.4 JAPAN
- 13.5.4.1 Declining clinical trial activity despite strong legacy position to impact market
- 13.5.5 INDIA
- 13.5.5.1 Rapid clinical trial growth, cost advantages, and regulatory reforms to drive market
- 13.5.6 AUSTRALIA
- 13.5.6.1 Strong early-phase trial capabilities and advanced research infrastructure to drive market
- 13.5.7 SOUTH KOREA
- 13.5.7.1 Steady clinical trial growth and strong innovation ecosystem to drive market
- 13.5.8 REST OF ASIA PACIFIC
- 13.6 MIDDLE EAST & AFRICA
- 13.6.1 MACROECONOMIC OUTLOOK FOR MIDDLE EAST & AFRICA
- 13.6.2 GCC COUNTRIES
- 13.6.2.1 Strong digital health transformation and growing clinical research activity to drive adoption
- 13.6.3 SAUDI ARABIA
- 13.6.3.1 Expanding clinical research activity and government-led initiatives to drive market
- 13.6.4 UAE
- 13.6.4.1 Advancing digital health innovation and strategic collaborations - key growth drivers
- 13.6.5 OTHER GCC COUNTRIES
- 13.6.5.1 Regulatory initiatives, national health visions, and digital health infrastructure boost market growth
- 13.6.6 SOUTH AFRICA
- 13.6.6.1 Leading clinical research hub in Africa with strong infrastructure
- 13.6.7 REST OF MIDDLE EAST & AFRICA
14 COMPETITIVE LANDSCAPE
- 14.1 OVERVIEW
- 14.2 KEY PLAYER STRATEGIES/RIGHT TO WIN
- 14.2.1 OVERVIEW OF STRATEGIES ADOPTED BY KEY PLAYERS IN DIGITAL BIOMARKERS MARKET
- 14.3 REVENUE SHARE ANALYSIS OF THE TOP MARKET PLAYERS
- 14.4 MARKET SHARE ANALYSIS, 2025
- 14.5 BRAND COMPARISON
- 14.6 COMPANY VALUATION & FINANCIAL METRICS
- 14.6.1 FINANCIAL METRICS
- 14.6.2 COMPANY VALUATION
- 14.7 COMPANY EVALUATION MATRIX
- 14.7.1 STARS
- 14.7.2 EMERGING LEADERS
- 14.7.3 PERVASIVE PLAYERS
- 14.7.4 PARTICIPANTS
- 14.7.5 COMPANY FOOTPRINT: KEY PLAYERS, 2025
- 14.7.5.1 Company footprint
- 14.7.5.2 Region footprint
- 14.7.5.3 Type footprint
- 14.7.5.4 Therapeutic area footprint
- 14.7.5.5 Application footprint
- 14.7.5.6 End user footprint
- 14.8 COMPANY EVALUATION MATRIX: STARTUPS/SMES, 2025
- 14.8.1 PROGRESSIVE COMPANIES
- 14.8.2 RESPONSIVE COMPANIES
- 14.8.3 DYNAMIC COMPANIES
- 14.8.4 STARTING BLOCKS
- 14.8.5 COMPETITIVE BENCHMARKING: STARTUPS/SMES, 2024
- 14.8.5.1 Detailed list of key startups/SMEs
- 14.8.5.2 Competitive benchmarking of startups/SMEs
- 14.9 COMPETITIVE SCENARIO
- 14.9.1 PRODUCT/SERVICE LAUNCHES & APPROVALS
- 14.9.2 DEALS
- 14.9.3 OTHER DEVELOPMENTS
15 COMPANY PROFILES
- 15.1 KEY PLAYERS
- 15.1.1 IXICO PLC
- 15.1.1.1 Business overview
- 15.1.1.2 Products offered
- 15.1.1.3 Recent developments
- 15.1.1.3.1 Product launches/approvals
- 15.1.1.3.2 Deals
- 15.1.1.4 MnM view
- 15.1.1.4.1 Key strengths
- 15.1.1.4.2 Strategic choices
- 15.1.1.4.3 Weaknesses and competitive threats
- 15.1.2 AMETRIS, LLC
- 15.1.2.1 Business overview
- 15.1.2.2 Products Offered
- 15.1.2.3 Recent developments
- 15.1.2.3.1 Product launches/enhancements
- 15.1.2.3.2 Deals
- 15.1.2.4 MnM view
- 15.1.2.4.1 Key strengths
- 15.1.2.4.2 Strategic choices
- 15.1.2.4.3 Weaknesses and competitive threats
- 15.1.3 EMPATICA INC.
- 15.1.3.1 Business overview
- 15.1.3.2 Products/Solutions offered
- 15.1.3.3 Recent developments
- 15.1.3.3.1 Product launches/approvals
- 15.1.3.3.2 Deals
- 15.1.3.4 MnM view
- 15.1.3.4.1 Key strengths
- 15.1.3.4.2 Strategic choices
- 15.1.3.4.3 Weaknesses and competitive threats
- 15.1.4 ALIVECOR, INC.
- 15.1.4.1 Business overview
- 15.1.4.2 Products offered
- 15.1.4.3 Recent developments
- 15.1.4.3.1 Product launches/approvals
- 15.1.4.3.2 Deals
- 15.1.4.4 MnM view
- 15.1.4.4.1 Key strengths
- 15.1.4.4.2 Strategic choices
- 15.1.4.4.3 Weaknesses and competitive threats
- 15.1.5 CONNEQT HEALTH
- 15.1.5.1 Business overview
- 15.1.5.2 Products offered
- 15.1.5.3 Recent developments
- 15.1.5.3.1 Product enhancements/approvals
- 15.1.5.4 MnM view
- 15.1.5.4.1 Key strengths
- 15.1.5.4.2 Strategic choices
- 15.1.5.4.3 Weaknesses and competitive threats
- 15.1.6 VIVOSENSE
- 15.1.6.1 Business overview
- 15.1.6.2 Products offered
- 15.1.6.2.1 Other developments
- 15.1.7 BIOSENSICS
- 15.1.7.1 Business overview
- 15.1.7.2 Products offered
- 15.1.7.3 Recent developments
- 15.1.7.3.1 Product launches/approvals
- 15.1.7.3.2 Deals
- 15.1.8 LUNIT INC.
- 15.1.8.1 Business overview
- 15.1.8.2 Products offered
- 15.1.8.3 Recent developments
- 15.1.8.3.1 Product launches/approvals
- 15.1.8.3.2 Deals
- 15.1.9 AKILI, INC.
- 15.1.9.1 Business overview
- 15.1.9.2 Products offered
- 15.1.9.3 Recent developments
- 15.1.9.3.1 Deals
- 15.1.10 QUIBIM
- 15.1.10.1 Business overview
- 15.1.10.2 Products offered
- 15.1.10.3 Recent developments
- 15.1.10.3.1 Product launches/approvals/enhancements
- 15.1.10.3.2 Deals
- 15.1.11 CLARIO
- 15.1.11.1 Business overview
- 15.1.11.2 Products offered
- 15.1.11.3 Recent developments
- 15.1.11.3.1 Deals
- 15.1.12 PROSCIA INC.
- 15.1.12.1 Business overview
- 15.1.12.2 Products offered
- 15.1.12.3 Recent developments
- 15.1.12.3.1 Product launches/approvals
- 15.1.12.3.2 Deals
- 15.1.13 KONEKSA HEALTH
- 15.1.13.1 Business overview
- 15.1.13.2 Products offered
- 15.1.13.3 Recent developments
- 15.1.13.3.1 Product launches
- 15.1.13.3.2 Deals
- 15.1.14 LINUS HEALTH
- 15.1.14.1 Business overview
- 15.1.14.2 Products offered
- 15.1.14.3 Recent developments
- 15.1.14.3.1 Product launches/approvals
- 15.1.14.3.2 Deals
- 15.1.1 IXICO PLC
- 15.2 OTHER PLAYERS
- 15.2.1 SONDE HEALTH, INC.
- 15.2.2 ALTOIDA
- 15.2.3 IMVARIA INC.
- 15.2.4 CUMULUS NEUROSCIENCE LIMITED
- 15.2.5 DELVE HEALTH
- 15.2.6 FEEL THERAPEUTICS
- 15.2.7 NANOWEAR INC.
- 15.2.8 EXOSYSTEMS
- 15.2.9 CARDIOSIGNAL
- 15.2.10 IMAGENE AI LTD.
- 15.2.11 NEUROTRACK TECHNOLOGIES, INC.
16 RESEARCH METHODOLOGY
- 16.1 RESEARCH APPROACH
- 16.1.1 SECONDARY RESEARCH
- 16.1.1.1 Key data from secondary sources
- 16.1.2 PRIMARY RESEARCH
- 16.1.2.1 Primary sources
- 16.1.2.2 Key data from primary sources
- 16.1.2.3 Breakdown of primaries
- 16.1.2.4 Insights from primary experts
- 16.1.1 SECONDARY RESEARCH
- 16.2 RESEARCH METHODOLOGY DESIGN
- 16.3 MARKET SIZE ESTIMATION
- 16.4 MARKET BREAKDOWN DATA TRIANGULATION
- 16.5 MARKET SHARE ESTIMATION
- 16.6 STUDY ASSUMPTIONS
- 16.7 RESEARCH LIMITATIONS
- 16.7.1 METHODOLOGY-RELATED LIMITATIONS
- 16.8 RISK ASSESSMENT
17 APPENDIX
- 17.1 DISCUSSION GUIDE
- 17.2 KNOWLEDGESTORE: MARKETSANDMARKETS' SUBSCRIPTION PORTAL
- 17.3 CUSTOMIZATION OPTIONS
- 17.4 RELATED REPORTS
- 17.5 AUTHOR DETAILS
List of Tables
- TABLE 1 USD EXCHANGE RATES
- TABLE 2 DIGITAL BIOMARKERS MARKET: PORTER'S FIVE FORCES ANALYSIS
- TABLE 3 DIGITAL BIOMARKERS MARKET: ROLE IN ECOSYSTEM
- TABLE 4 INDICATIVE PRICE FOR DIGITAL BIOMARKERS, BY KEY PLAYER (2025)
- TABLE 5 INDICATIVE PRICE FOR DIGITAL BIOMARKERS, BY REGION (2025)
- TABLE 6 CASE 1: EMPATICA - INTEGRATION OF DIGITAL BIOMARKERS INTO HIGH-ALTITUDE ENDOCRINOLOGY RESEARCH (APEX 7 HICORT STUDY)
- TABLE 7 CASE 2: VIVOSENSE - DIGITAL BIOMARKERS FOR PTSD MONITORING AND ASSESSMENT
- TABLE 8 CASE 3: DELVE HEALTH - IMPROVED DATA INTEGRITY IN CLINICAL TRIALS WITH DIGITAL BIOMARKERS
- TABLE 9 US ADJUSTED RECIPROCAL TARIFF RATES
- TABLE 10 INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS OF TOP THREE END USERS (%)
- TABLE 11 KEY BUYING CRITERIA FOR TOP THREE END USERS
- TABLE 12 UNMET NEEDS IN DIGITAL BIOMARKERS MARKET
- TABLE 13 END USER EXPECTATIONS IN DIGITAL BIOMARKERS MARKET
- TABLE 14 NORTH AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 15 EUROPE: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 16 ASIA PACIFIC: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 17 LATIN AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 18 MIDDLE EAST & AFRICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 19 REGULATORY SCENARIO IN NORTH AMERICA
- TABLE 20 REGULATORY SCENARIO IN EUROPE
- TABLE 21 REGULATORY SCENARIO IN ASIA PACIFIC
- TABLE 22 REGULATORY SCENARIO IN LATIN AMERICA
- TABLE 23 REGULATORY SCENARIO IN MIDDLE EAST & AFRICA
- TABLE 24 DIGITAL BIOMARKERS MARKET: JURISDICTION ANALYSIS OF TOP APPLICANT COUNTRIES
- TABLE 25 DIGITAL BIOMARKERS MARKET: LIST OF PATENTS/PATENT APPLICATIONS
- TABLE 26 DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 27 PHYSIOLOGICAL BIOMARKERS: MAJOR PLAYERS AND OFFERINGS
- TABLE 28 PHYSIOLOGICAL BIOMARKERS: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 29 IDIOSYNCRATIC BIOMARKERS: MAJOR PLAYERS AND OFFERINGS
- TABLE 30 IDIOSYNCRATIC BIOMARKERS: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 31 COGNITIVE BIOMARKERS: MAJOR PLAYERS AND OFFERINGS
- TABLE 32 COGNITIVE BIOMARKERS: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 33 VOCAL BIOMARKERS: MAJOR PLAYERS AND OFFERINGS
- TABLE 34 VOCAL BIOMARKERS: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 35 OTHER BIOMARKERS: MAJOR PLAYERS AND OFFERINGS
- TABLE 36 OTHER BIOMARKERS: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 37 DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 38 CARDIOVASCULAR: MAJOR PLAYERS AND OFFERINGS
- TABLE 39 CARDIOVASCULAR: DIGITAL BIOMARKERS MARKET, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 40 CARDIOVASCULAR: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 41 ASCVD/SECONDARY PREVENTION: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 42 HEART FAILURE (HFREF & HFPEF): DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 43 HYPERTENSION: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 44 ATRIAL FIBRILLATION/STROKE PREVENTION: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 45 PULMONARY HYPERTENSION: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 46 STRUCTURAL HEART DISEASE/INTERVENTIONAL CARDIOLOGY: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 47 ONCOLOGY: MAJOR PLAYERS AND OFFERINGS
- TABLE 48 ONCOLOGY: DIGITAL BIOMARKERS MARKET, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 49 ONCOLOGY: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 50 SOLID TUMORS: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 51 SOLID TUMORS: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 52 BREAST CANCER: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 53 LUNG CANCER: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 54 PROSTATE CANCER: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 55 COLORECTAL CANCER: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 56 BRAIN TUMOR: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 57 OTHER SOLID TUMORS: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 58 HEMATOLOGIC MALIGNANCIES: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 59 HEMATOLOGIC MALIGNANCIES: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 60 LEUKEMIA: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 61 LYMPHOMA: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 62 MULTIPLE MYELOMA: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 63 OTHER HEMATOLOGIC MALIGNANCIES: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 64 DIABETES: MAJOR PLAYERS AND OFFERINGS
- TABLE 65 DIABETES: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 66 MENTAL HEALTH & BEHAVIORAL HEALTH: MAJOR PLAYERS AND OFFERINGS
- TABLE 67 MENTAL HEALTH & BEHAVIORAL HEALTH: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 68 RESPIRATORY DISORDERS: MAJOR PLAYERS AND OFFERINGS
- TABLE 69 RESPIRATORY DISORDERS: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 70 LIFESTYLE & WELLNESS IMPROVEMENT: MAJOR PLAYERS AND OFFERINGS
- TABLE 71 LIFESTYLE & WELLNESS IMPROVEMENT: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 72 NEUROLOGY: MAJOR PLAYERS AND OFFERINGS
- TABLE 73 NEUROLOGY: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 74 MUSCULOSKELETAL DISORDERS/PAIN MANAGEMENT: MAJOR PLAYERS AND OFFERINGS
- TABLE 75 MUSCULOSKELETAL DISORDERS/PAIN MANAGEMENT: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 76 WOMEN'S HEALTH & REPRODUCTIVE HEALTH: MAJOR PLAYERS AND OFFERINGS
- TABLE 77 WOMEN'S HEALTH & REPRODUCTIVE HEALTH: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 78 OTHER DISEASES: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 79 DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 80 CLINICAL RESEARCH: DIGITAL BIOMARKERS MARKET, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 81 CLINICAL RESEARCH: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 82 PHASE II CLINICAL RESEARCH: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 83 PHASE III CLINICAL RESEARCH: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 84 PHASE IV CLINICAL RESEARCH: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 85 CLINICAL CARE: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 86 DIGITAL BIOMARKER MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 87 PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 88 CONTRACT RESEARCH ORGANIZATIONS: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 89 HOSPITALS & SPECIALTY CLINICS: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 90 OTHER END USERS: DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 91 DIGITAL BIOMARKERS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 92 NORTH AMERICA: DIGITAL BIOMARKERS MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 93 NORTH AMERICA: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 94 NORTH AMERICA: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 95 NORTH AMERICA: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 96 NORTH AMERICA: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 97 NORTH AMERICA: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 98 NORTH AMERICA: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 99 NORTH AMERICA: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 100 NORTH AMERICA: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 101 NORTH AMERICA: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 102 US: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 103 US: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 104 US: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 105 US: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 106 US: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 107 US: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 108 US: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 109 US: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 110 US: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 111 CANADA: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 112 CANADA: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 113 CANADA: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 114 CANADA: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 115 CANADA: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 116 CANADA: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 117 CANADA: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 118 CANADA: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 119 CANADA: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 120 EUROPE: DIGITAL BIOMARKERS MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 121 EUROPE: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 122 EUROPE: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 123 EUROPE: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 124 EUROPE: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 125 EUROPE: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 126 EUROPE: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 127 EUROPE: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 128 EUROPE: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 129 EUROPE: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 130 GERMANY: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 131 GERMANY: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 132 GERMANY: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 133 GERMANY: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 134 GERMANY: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 135 GERMANY: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 136 GERMANY: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 137 GERMANY: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 138 GERMANY: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 139 FRANCE: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 140 FRANCE: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 141 FRANCE: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 142 FRANCE: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 143 FRANCE: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 144 FRANCE: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 145 FRANCE: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 146 FRANCE: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 147 FRANCE: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 148 UK: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 149 UK: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 150 UK: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 151 UK: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 152 UK: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 153 UK: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 154 UK: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 155 UK: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 156 UK: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 157 ITALY: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 158 ITALY: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 159 ITALY: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 160 ITALY: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 161 ITALY: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 162 ITALY: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 163 ITALY: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 164 ITALY: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 165 ITALY: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 166 SPAIN: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 167 SPAIN: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 168 SPAIN: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 169 SPAIN: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 170 SPAIN: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 171 SPAIN: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 172 SPAIN: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 173 SPAIN: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 174 SPAIN: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 175 REST OF EUROPE: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 176 REST OF EUROPE: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 177 REST OF EUROPE: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 178 REST OF EUROPE: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 179 REST OF EUROPE: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 180 REST OF EUROPE: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 181 REST OF EUROPE: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 182 REST OF EUROPE: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 183 REST OF EUROPE: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 184 LATIN AMERICA: DIGITAL BIOMARKERS MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 185 LATIN AMERICA: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 186 LATIN AMERICA: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 187 LATIN AMERICA: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 188 LATIN AMERICA: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 189 LATIN AMERICA: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 190 LATIN AMERICA: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 191 LATIN AMERICA: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 192 LATIN AMERICA: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 193 LATIN AMERICA: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 194 BRAZIL: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 195 BRAZIL: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 196 BRAZIL: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 197 BRAZIL: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 198 BRAZIL: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 199 BRAZIL: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 200 BRAZIL: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 201 BRAZIL: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 202 BRAZIL: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 203 MEXICO: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 204 MEXICO: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 205 MEXICO: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 206 MEXICO: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 207 MEXICO: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 208 MEXICO: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 209 MEXICO: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 210 MEXICO: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 211 MEXICO: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 212 REST OF LATIN AMERICA: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 213 REST OF LATIN AMERICA: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 214 REST OF LATIN AMERICA: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 215 REST OF LATIN AMERICA: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 216 REST OF LATIN AMERICA: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 217 REST OF LATIN AMERICA: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 218 REST OF LATIN AMERICA: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 219 REST OF LATIN AMERICA: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 220 REST OF LATIN AMERICA: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 221 ASIA PACIFIC: DIGITAL BIOMARKERS MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 222 ASIA PACIFIC: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 223 ASIA PACIFIC: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 224 ASIA PACIFIC: DIGITAL BIOMARKERS MARKET, FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 225 ASIA PACIFIC: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 226 ASIA PACIFIC: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 227 ASIA PACIFIC: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 228 ASIA PACIFIC: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 229 ASIA PACIFIC: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 230 ASIA PACIFIC: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 231 CHINA: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 232 CHINA: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 233 CHINA: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 234 CHINA: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 235 CHINA: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 236 CHINA: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 237 CHINA: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 238 CHINA: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 239 CHINA: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 240 JAPAN: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 241 JAPAN: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 242 JAPAN: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 243 JAPAN: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 244 JAPAN: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 245 JAPAN: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 246 JAPAN: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 247 JAPAN: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 248 JAPAN: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 249 INDIA: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 250 INDIA: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 251 INDIA: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 252 INDIA: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 253 INDIA: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 254 INDIA: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 255 INDIA: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 256 INDIA: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 257 INDIA: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 258 AUSTRALIA: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 259 AUSTRALIA: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 260 AUSTRALIA: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 261 AUSTRALIA: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 262 AUSTRALIA: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 263 AUSTRALIA: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 264 AUSTRALIA: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 265 AUSTRALIA: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 266 AUSTRALIA: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 267 SOUTH KOREA: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 268 SOUTH KOREA: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 269 SOUTH KOREA: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 270 SOUTH KOREA: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 271 SOUTH KOREA: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 272 SOUTH KOREA: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 273 SOUTH KOREA: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 274 SOUTH KOREA: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 275 SOUTH KOREA: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 276 REST OF ASIA PACIFIC: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 277 REST OF ASIA PACIFIC: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 278 REST OF ASIA PACIFIC: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 279 REST OF ASIA PACIFIC: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 280 REST OF ASIA PACIFIC: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 281 REST OF ASIA PACIFIC: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 282 REST OF ASIA PACIFIC: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 283 REST OF ASIA PACIFIC: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 284 REST OF ASIA PACIFIC: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 285 MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 286 MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 287 MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 288 MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 289 MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 290 MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 291 MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 292 MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 293 MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 294 MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 295 GCC COUNTRIES: DIGITAL BIOMARKERS MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 296 GCC COUNTRIES: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 297 GCC COUNTRIES: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 298 GCC COUNTRIES: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 299 GCC COUNTRIES: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 300 GCC COUNTRIES: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 301 GCC COUNTRIES: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 302 GCC COUNTRIES: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 303 GCC COUNTRIES: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 304 GCC COUNTRIES: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 305 SAUDI ARABIA: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 306 SAUDI ARABIA: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 307 SAUDI ARABIA: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 308 SAUDI ARABIA: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 309 SAUDI ARABIA: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 310 SAUDI ARABIA: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 311 SAUDI ARABIA: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 312 SAUDI ARABIA: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 313 SAUDI ARABIA: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 314 UAE: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 315 UAE: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 316 UAE: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 317 UAE: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 318 UAE: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 319 UAE: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 320 UAE: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 321 UAE: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 322 UAE: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 323 OTHER GCC COUNTRIES: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 324 OTHER GCC COUNTRIES: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 325 OTHER GCC COUNTRIES: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 326 OTHER GCC COUNTRIES: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 327 OTHER GCC COUNTRIES: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 328 OTHER GCC COUNTRIES: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 329 OTHER GCC COUNTRIES: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 330 OTHER GCC COUNTRIES: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 331 OTHER GCC COUNTRIES: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 332 SOUTH AFRICA: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 333 SOUTH AFRICA: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 334 SOUTH AFRICA: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 335 SOUTH AFRICA: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 336 SOUTH AFRICA: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 337 SOUTH AFRICA: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 338 SOUTH AFRICA: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 339 SOUTH AFRICA: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 340 SOUTH AFRICA: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 341 REST OF MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 342 REST OF MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET, BY THERAPEUTIC AREA, 2024-2031 (USD MILLION)
- TABLE 343 REST OF MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET FOR CARDIOVASCULAR, BY CONDITION, 2024-2031 (USD MILLION)
- TABLE 344 REST OF MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET FOR ONCOLOGY, BY TUMOR TYPE, 2024-2031 (USD MILLION)
- TABLE 345 REST OF MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET FOR SOLID TUMORS, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 346 REST OF MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET FOR HEMATOLOGIC MALIGNANCIES, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 347 REST OF MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 348 REST OF MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET FOR CLINICAL RESEARCH, BY PHASE, 2024-2031 (USD MILLION)
- TABLE 349 REST OF MIDDLE EAST & AFRICA: DIGITAL BIOMARKERS MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 350 OVERVIEW OF STRATEGIES ADOPTED BY KEY PLAYERS IN DIGITAL BIOMARKERS MARKET, JANUARY 2022-MARCH 2026
- TABLE 351 DIGITAL BIOMARKERS MARKET: DEGREE OF COMPETITION
- TABLE 352 DIGITAL BIOMARKERS MARKET: REGION FOOTPRINT (14 COMPANIES)
- TABLE 353 DIGITAL BIOMARKERS MARKET: TYPE FOOTPRINT (14 COMPANIES)
- TABLE 354 DIGITAL BIOMARKERS MARKET: THERAPEUTIC AREA FOOTPRINT (14 COMPANIES)
- TABLE 355 DIGITAL BIOMARKERS MARKET: APPLICATION FOOTPRINT (14 COMPANIES)
- TABLE 356 DIGITAL BIOMARKERS MARKET: END USER FOOTPRINT (14 COMPANIES)
- TABLE 357 DIGITAL BIOMARKERS MARKET: DETAILED LIST OF KEY STARTUPS/SMES
- TABLE 358 DIGITAL BIOMARKERS MARKET: COMPETITIVE BENCHMARKING OF STARTUPS/SMES, BY REGION
- TABLE 359 DIGITAL BIOMARKERS MARKET: COMPETITIVE BENCHMARKING OF STARTUPS/SMES, BY TYPE
- TABLE 360 DIGITAL BIOMARKERS MARKET: PRODUCT/SERVICE LAUNCHES & APPROVALS, JANUARY 2022-MARCH 2026
- TABLE 361 DIGITAL BIOMARKERS MARKET: DEALS, JANUARY 2022-MARCH 2026
- TABLE 362 DIGITAL BIOMARKERS MARKET: OTHER DEVELOPMENTS, JANUARY 2022-MARCH 2026
- TABLE 363 IXICO PLC: COMPANY OVERVIEW
- TABLE 364 IXICO PLC: PRODUCTS OFFERED
- TABLE 365 IXICO PLC.: PRODUCT LAUNCHES/APPROVALS, JANUARY 2022-MARCH 2026
- TABLE 366 IXICO PLC.: DEALS, JANUARY 2022-MARCH 2026
- TABLE 367 AMETRIS, LLC: COMPANY OVERVIEW
- TABLE 368 AMETRIS LLC: PRODUCTS OFFERED
- TABLE 369 AMETRIS LLC: PRODUCT LAUNCHES/ENHANCEMENTS, JANUARY 2022-MARCH 2026
- TABLE 370 AMETRIS, LLC: DEALS, JANUARY 2022-MARCH 2026
- TABLE 371 EMPATICA INC.: COMPANY OVERVIEW
- TABLE 372 EMPATICA INC.: PRODUCTS/SOLUTIONS OFFERED
- TABLE 373 EMPATICA INC.: PRODUCT LAUNCHES/APPROVALS, JANUARY 2022-MARCH 2026
- TABLE 374 EMPATICA INC.: DEALS, JANUARY 2022-MARCH 2026
- TABLE 375 ALIVECOR, INC.: COMPANY OVERVIEW
- TABLE 376 ALIVECOR, INC.: PRODUCTS OFFERED
- TABLE 377 ALIVECOR, INC.: PRODUCT LAUNCHES/APPROVALS, JANUARY 2022-MARCH 2026
- TABLE 378 ALIVECOR, INC.: DEALS, JANUARY 2022-MARCH 2026
- TABLE 379 CONNEQT HEALTH: COMPANY OVERVIEW
- TABLE 380 CONNEQT HEALTH: PRODUCTS OFFERED
- TABLE 381 CONNEQT HEALTH: PRODUCT ENHANCEMENTS/APPROVALS, JANUARY 2022-MARCH 2026
- TABLE 382 VIVOSENSE: COMPANY OVERVIEW
- TABLE 383 VIVOSENSE: PRODUCTS OFFERED
- TABLE 384 VIVOSENSE: OTHER DEVELOPMENTS, JANUARY 2022-MARCH 2026
- TABLE 385 BIOSENSICS: COMPANY OVERVIEW
- TABLE 386 BIOSENSICS: PRODUCTS OFFERED
- TABLE 387 BIOSENSICS: PRODUCT LAUNCHES/APPROVALS, JANUARY 2022-MARCH 2026
- TABLE 388 BIOSENSICS: DEALS, JANUARY 2022-MARCH 2026
- TABLE 389 LUNIT INC.: COMPANY OVERVIEW
- TABLE 390 LUNIT INC.: PRODUCTS OFFERED
- TABLE 391 LUNIT INC.: PRODUCT LAUNCHES/APPROVALS, JANUARY 2022-MARCH 2026
- TABLE 392 LUNIT INC.: DEALS, JANUARY 2022-MARCH 2026
- TABLE 393 AKILI, INC.: COMPANY OVERVIEW
- TABLE 394 AKILI, INC.: PRODUCTS OFFERED
- TABLE 395 AKILI, INC.: DEALS, JANUARY 2022-MARCH 2026
- TABLE 396 QUIBIM: COMPANY OVERVIEW
- TABLE 397 QUIBIM: PRODUCTS OFFERED
- TABLE 398 QUIBIM: PRODUCT LAUNCHES/APPROVALS/ENHANCEMENTS, JANUARY 2022-MARCH 2026
- TABLE 399 QUIBIM: DEALS, JANUARY 2022-MARCH 2026
- TABLE 400 CLARIO: COMPANY OVERVIEW
- TABLE 401 CLARIO: PRODUCTS OFFERED
- TABLE 402 CLARIO: DEALS, JANUARY 2022-MARCH 2026
- TABLE 403 PROSCIA INC.: COMPANY OVERVIEW
- TABLE 404 PROSCIA INC.: PRODUCTS OFFERED
- TABLE 405 PROSCIA INC.: PRODUCT LAUNCHES/APPROVALS, JANUARY 2022-MARCH 2026
- TABLE 406 PROSCIA INC.: DEALS, JANUARY 2022-MARCH 2026
- TABLE 407 KONEKSA HEALTH: COMPANY OVERVIEW
- TABLE 408 KONEKSA HEALTH: PRODUCTS OFFERED
- TABLE 409 KONEKSA HEALTH: PRODUCT LAUNCHES, JANUARY 2022-MARCH 2026
- TABLE 410 KONEKSA HEALTH: DEALS, JANUARY 2022- MARCH 2026
- TABLE 411 LINUS HEALTH: COMPANY OVERVIEW
- TABLE 412 LINUS HEALTH: PRODUCTS OFFERED
- TABLE 413 LINUS HEALTH: PRODUCT LAUNCHES/APPROVALS, JANUARY 2022-MARCH 2026
- TABLE 414 LINUS HEALTH: DEALS, JANUARY 2022-MARCH 2026
- TABLE 415 SONDE HEALTH, INC.: COMPANY OVERVIEW
- TABLE 416 ALTOIDA: COMPANY OVERVIEW
- TABLE 417 IMVARIA INC.: COMPANY OVERVIEW
- TABLE 418 CUMULUS NEUROSCIENCE LIMITED: COMPANY OVERVIEW
- TABLE 419 DELVE HEALTH: COMPANY OVERVIEW
- TABLE 420 FEEL THERAPEUTICS: COMPANY OVERVIEW
- TABLE 421 NANOWEAR INC.: COMPANY OVERVIEW
- TABLE 422 EXOSYSTEMS: COMPANY OVERVIEW
- TABLE 423 CARDIOSIGNAL: COMPANY OVERVIEW
- TABLE 424 IMAGENE AI LTD.: COMPANY OVERVIEW
- TABLE 425 NEUROTRACK TECHNOLOGIES, INC.: COMPANY OVERVIEW
- TABLE 426 RISK ASSESSMENT: DIGITAL BIOMARKERS MARKET
List of Figures
- FIGURE 1 DIGITAL BIOMARKERS MARKET SEGMENTATION
- FIGURE 2 MARKET SCENARIO
- FIGURE 3 GLOBAL DIGITAL BIOMARKERS MARKET, 2024-2031
- FIGURE 4 MAJOR STRATEGIES ADOPTED BY KEY PLAYERS IN DIGITAL BIOMARKERS MARKET, 2023-2026
- FIGURE 5 DISRUPTIONS INFLUENCING GROWTH OF DIGITAL BIOMARKERS MARKET
- FIGURE 6 HIGH-GROWTH SEGMENTS IN DIGITAL BIOMARKERS MARKET, 2026-2031
- FIGURE 7 ASIA PACIFIC TO REGISTER HIGHEST CAGR IN DIGITAL BIOMARKERS MARKET DURING FORECAST PERIOD
- FIGURE 8 RISING IN CLINICAL TRIAL VOLUMES TO DRIVE GROWTH IN DIGITAL BIOMARKERS MARKET
- FIGURE 9 CLINICAL RESEARCH APPLICATIONS ACCOUNTED FOR LARGEST MARKET SHARE IN NORTH AMERICA IN 2025
- FIGURE 10 INDIA TO REGISTER HIGHEST GROWTH RATE DURING FORECAST PERIOD
- FIGURE 11 DIGITAL BIOMARKERS MARKET: PORTER'S FIVE FORCES ANALYSIS
- FIGURE 12 DIGITAL BIOMARKERS MARKET: SUPPLY CHAIN ANALYSIS
- FIGURE 13 DIGITAL BIOMARKERS MARKET: ECOSYSTEM ANALYSIS
- FIGURE 14 TRENDS/DISRUPTIONS IMPACTING CUSTOMERS' BUSINESSES
- FIGURE 16 INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS FOR END USERS
- FIGURE 17 KEY BUYING CRITERIA FOR TOP THREE END USERS
- FIGURE 18 DIGITAL BIOMARKERS MARKET: PATENT PUBLICATION TRENDS, 2015-2026
- FIGURE 19 DIGITAL BIOMARKERS MARKET: JURISDICTION AND TOP APPLICANT ANALYSIS
- FIGURE 20 DIGITAL BIOMARKERS MARKET: TOP APPLICANTS & OWNERS (COMPANIES/INSTITUTIONS), JANUARY 2015-APRIL 2026
- FIGURE 21 MARKET POTENTIAL OF AI/GEN AI ON DIGITAL BIOMARKERS ACROSS INDUSTRIES
- FIGURE 22 IMPACT OF AI/GEN AI ON INTERCONNECTED AND ADJACENT ECOSYSTEMS
- FIGURE 23 TRIAL PHASES WITH DIGITAL ENDPOINTS
- FIGURE 24 NORTH AMERICA: DIGITAL BIOMARKERS MARKET SNAPSHOT
- FIGURE 25 ASIA PACIFIC: DIGITAL BIOMARKERS MARKET SNAPSHOT
- FIGURE 26 DIGITAL BIOMARKERS MARKET: REVENUE ANALYSIS OF KEY PLAYERS
- FIGURE 27 DIGITAL BIOMARKERS MARKET SHARE ANALYSIS OF KEY PLAYERS (2025)
- FIGURE 28 DIGITAL BIOMARKERS MARKET: BRAND COMPARATIVE ANALYSIS
- FIGURE 29 EV/EBITDA OF KEY VENDORS
- FIGURE 30 YEAR-TO-DATE (YTD) PRICE TOTAL RETURN AND 5-YEAR STOCK BETA OF KEY VENDORS
- FIGURE 31 DIGITAL BIOMARKERS MARKET: COMPANY EVALUATION MATRIX (2025)
- FIGURE 32 DIGITAL BIOMARKERS MARKET: COMPANY FOOTPRINT
- FIGURE 33 DIGITAL BIOMARKERS MARKET: COMPANY EVALUATION MATRIX (STARTUPS/SMES), 2025
- FIGURE 34 IXICO PLC: COMPANY SNAPSHOT (2025)
- FIGURE 35 CONNEQT HEALTH: COMPANY SNAPSHOT (2025)
- FIGURE 36 LUNIT INC.: COMPANY SNAPSHOT (2025)
- FIGURE 37 RESEARCH DESIGN
- FIGURE 38 BREAKDOWN OF PRIMARY INTERVIEWS: BY COMPANY TYPE, DESIGNATION, AND REGION
- FIGURE 39 RESEARCH METHODOLOGY: HYPOTHESIS BUILDING
- FIGURE 40 BOTTOM-UP APPROACH
- FIGURE 41 TOP-DOWN APPROACH
- FIGURE 42 CAGR PROJECTIONS BASED ON DROC ANALYSIS OF DIGITAL BIOMARKERS INDUSTRY
- FIGURE 43 CAGR PROJECTIONS: SUPPLY-SIDE ANALYSIS
- FIGURE 44 DATA TRIANGULATION METHODOLOGY




