PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1851078

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1851078
Gene Therapy - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts (2025 - 2030)
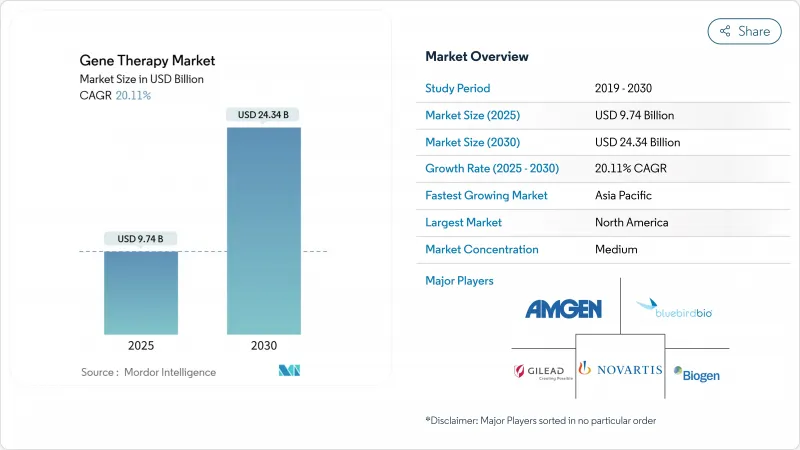
The Gene Therapy Market size is estimated at USD 9.74 billion in 2025, and is expected to reach USD 24.34 billion by 2030, at a CAGR of 20.11% during the forecast period (2025-2030).
Regulatory authorities cleared nine cell and gene therapies in 2024 alone, signalling rising confidence in clinical and commercial viability and unlocking wider reimbursement pathways in key markets. Heightened biopharma and private-equity funding is accelerating late-stage assets, while expanding manufacturing capacity both viral and non-viral reduces historic supply-chain friction. Rapid advances in in-vivo editing and vector engineering are broadening therapeutic scope beyond rare diseases to high-prevalence disorders, especially in neurology and ophthalmology. Asia-Pacific's double-digit trial growth and North America's policy incentives together ensure a diversified demand base, even as high product prices and safety-monitoring requirements pose access headwinds.
Global Gene Therapy Market Trends and Insights
Rising Regulatory Approvals Demonstrating Clinical and Commercial Viability
Frequent clearances from major regulators are reducing development risk and compressing launch timelines. The FDA's cumulative total of 37 approved cell and gene products by 2024 shows that agency reviewers now apply dedicated frameworks such as the Regenerative Medicine Advanced Therapy designation, which trims development time by about 40% for qualifying programs.Europe's PRIME scheme and the United Kingdom's 2025 rule for point-of-care manufacture further ease market entry. Approval of ZEVASKYN for recessive dystrophic epidermolysis bullosa in 2025 exemplifies how indications are extending beyond ultra-rare diseases into broader dermatological segments. Such widening therapeutic reach supports multi-year growth in the gene therapy market and stimulates follow-on investment from payers expecting durable clinical benefit.
Continuous Technological Advances in Vector Engineering and In-Vivo Editing Platforms
Enhanced capsid libraries, machine-learning-guided sequence optimisation, and novel lipid constructs are sharpening tissue tropism and reducing immunogenicity. Researchers at the Perelman School of Medicine reported DNA-loaded lipid nanoparticles that persist in vivo for months, opening durable dosing for chronic diseases.Organoid platforms offer human tissue-specific test beds that improve translational predictability, supported by the FDA Modernization Act's acceptance of non-animal models. AI-assisted algorithms such as OpenCRISPR-1 cut design cycles and elevate editing precision. Collectively these innovations shift the therapeutic ceiling higher, create room for repeat dosing, and fuel a virtuous loop of clinical success and capital deployment that enlarges the gene therapy market.
High Therapy Price Tags Creating Access and Affordability Barriers
Single-dose cures command list prices between USD 373,000 and USD 4.25 million, straining payer budgets and testing societal willingness to fund curative therapies. Providers cite social support gaps, travel distance, and prior authorisation hurdles as the top access blockers. Innovative reimbursement options-outcome-based contracts, amortisation models, and warranties are emerging but still require complex data sharing and long-term follow-up infrastructure. The Centers for Medicare & Medicaid Services launched a Cell and Gene Therapy Access Model in 2025 that encourages state-level pooled purchasing to temper budget impact. These financial headwinds are expected to moderate adoption in cost-sensitive regions, softening the gene therapy market growth curve despite strong clinical value propositions.
Other drivers and restraints analyzed in the detailed report include:
- Expansion of Global Viral-Vector and Non-Viral Manufacturing Infrastructure
- Escalating Biopharma & Private-Equity Funding into Gene-Therapy R&D
- Manufacturing Complexity and Constrained Supply of GMP-Grade Vectors
For complete list of drivers and restraints, kindly check the Table Of Contents.
Segment Analysis
AAV vectors held 38.54% of the gene therapy market share in 2024 owing to favourable safety and sustained transgene expression that underpin multiple approved products for haemophilia and inherited retinal disorders. This leadership is reinforced by broad tissue tropism and an expanding pool of engineered serotypes that improve specificity. However, non-viral lipid nanoparticle systems are the fastest-rising alternative, forecast to post a 24.34% CAGR through 2030 as they carry larger genetic payloads and simplify manufacturing economics. The gene therapy market size for non-viral platforms is therefore set to close part of the current revenue gap. Lentiviral vectors remain the mainstay for ex-vivo CAR-T manufacture, whereas herpes-based systems gain traction in oncology for their payload capacity. Continued optimisation of capsid libraries and synthetic promoters will determine whether viral vectors can defend share against cost-efficient non-viral challengers.
The surge in non-viral approaches also alleviates raw-material constraints because lipid components are amenable to standard pharmaceutical supply chains. DNA-loaded nanoparticles that remain active for months lower repeat-dosing requirements and can be tuned for organ-selective uptake. Such flexibility attracts developers targeting polygenic neurological disorders where payload size exceeds AAV's packaging limit. Consequently, the competitive dynamic inside the gene therapy market is shifting from vector availability toward delivery precision, creating space for platform-agnostic CDMOs to capture emerging demand.
Oncology applications generated 42.92% of revenue in 2024, supported by a steady flow of CAR-T approvals and a deep pipeline aimed at solid tumours. Robust evidence for complete responses in relapsed haematological malignancies sustains payer acceptance despite price pressures. Yet neurology claims the highest growth trajectory at a 25.62% CAGR to 2030. Emerging capsids that cross the blood-brain barrier and precise in-vivo editing tools permit direct intervention in disorders like Rett syndrome and Huntington's disease. The gene therapy market size allocated to CNS disorders is expected to multiply as bilateral regulatory incentives compress development risk.
Rare metabolic and ophthalmology indications continue to yield new approvals because tissue immune privilege and clear biomarkers simplify clinical endpoints. Cardiovascular targets such as hypertrophic cardiomyopathy are gaining venture funding, hinting at broader systemic disease uptake. As therapeutic breadth widens, portfolio optimisation becomes critical; sponsors must balance oncology cash flows with high-potential but scientifically complex neurology assets.
The Gene Therapy Market Report Segments the Industry Into by Vector Type (Adeno Virus Vector, and More), Indication (Oncology, Rare Metabolic Disorders, and More), Delivery Mode (In Vivo Gene Delivery and Ex Vivo Gene Delivery), End User (Hospitals and Clinics, and More), and Geography (North America, Europe, Asia-Pacific, Middle-East and Africa, South America). The Market Forecasts are Provided in Terms of Value (USD)
Geography Analysis
North America held 41.78% of global revenue in 2024, sustained by an accommodating regulatory environment, deep venture capital pools, and extensive clinical infrastructure. The United States approved 34 first-in-class gene therapies in 2024 alone, highlighting its central role in product launch sequencing. Federal initiatives such as ARPA-H grants channel significant funds toward manufacturing innovation and safety surveillance, reinforcing the region's leadership in the gene therapy market.
Europe maintains strong scientific output and benefits from the EMA's adaptive pathways that expedite breakthrough treatments. The forthcoming UK framework for decentralised manufacture, effective July 2025, may reduce logistical burdens and stimulate point-of-care production. However, unequal national reimbursement policies prolong time-to-patient across the bloc; the new EU Health Technology Assessment regulation in 2025 aims to harmonise evidence requirements but could also lengthen negotiations. Outcomes-based payment models, as demonstrated by Hemgenix agreements in England and Denmark, are gradually easing adoption hurdles.
Asia-Pacific registers the fastest growth at a 27.68% CAGR. China hosts more than 400 cell-based studies and invests heavily in domestic vector plants, positioning itself as both demand and supply engine. Japan's accelerated pathways for regenerative medicines and Singapore's digital licensing portal further streamline approvals. While regulatory standards converge with Western agencies, variable reimbursement and export controls on genetic materials remain challenges. Continuous policy support and maturing local CDMOs nevertheless assure that Asia-Pacific will capture an increasing proportion of the gene therapy market.
- Novartis
- Gilead Sciences
- Roche
- bluebird bio Inc.
- UniQure N.V.
- Amgen
- Biogen
- Astellas Pharma
- Sarepta Therapeutics
- Regenxbio Inc.
- Krystal Biotech Inc.
- Orchard Therapeutics plc
- Rocket Pharmaceuticals Inc.
- Editas Medicine
- Intellia Therapeutics Inc.
- Sangamo Therapeutics
- CSL Behring
- Mustang Bio Inc.
- Poseida Therapeutics Inc.
- Generation Bio Co.
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 Introduction
- 1.1 Study Assumptions & Market Definition
- 1.2 Scope of the Study
2 Research Methodology
3 Executive Summary
4 Market Landscape
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Rising number of regulatory approvals demonstrating clinical and commercial viability
- 4.2.2 Continuous technological advances in vector engineering and in-vivo editing platforms
- 4.2.3 Expansion of global viral-vector and non-viral manufacturing infrastructure
- 4.2.4 Rapid scale-up of commercial AAV CDMO capacity
- 4.2.5 Escalating biopharma & private-equity funding into gene-therapy R&D
- 4.2.6 Growing inclusion of rare-disease gene therapies
- 4.3 Market Restraints
- 4.3.1 High therapy price tags creating access and affordability barriers
- 4.3.2 Manufacturing complexity and constrained supply of GMP-grade vectors
- 4.3.3 Safety concerns necessitating long-term monitoring
- 4.3.4 Shortage of GMP-grade plasmid/capsid raw materials extending CMC timelines
- 4.4 Regulatory Landscape
- 4.5 Porter's Five Forces Analysis
- 4.5.1 Threat of New Entrants
- 4.5.2 Bargaining Power of Buyers
- 4.5.3 Bargaining Power of Suppliers
- 4.5.4 Threat of Substitute Products
- 4.5.5 Intensity of Competitive Rivalry
5 Market Size & Growth Forecasts (Value in USD)
- 5.1 By Vector Type
- 5.1.1 Adeno Virus Vector
- 5.1.2 Adeno-associated Virus Vector
- 5.1.3 Lentiviral Vector
- 5.1.4 Retroviral Vector
- 5.1.5 Herpes Virus Vector
- 5.1.6 Other Vector Types
- 5.2 By Indication
- 5.2.1 Oncology
- 5.2.2 Rare Metabolic Disorders
- 5.2.3 Ophthalmology
- 5.2.4 Neurology / CNS
- 5.2.5 Cardiovascular and Musculoskeletal
- 5.2.6 Other Indications
- 5.3 By Delivery Mode
- 5.3.1 In Vivo Gene Delivery
- 5.3.2 Ex Vivo Gene Delivery
- 5.4 By End User
- 5.4.1 Hospitals and Clinics
- 5.4.2 Specialty Treatment / Infusion Centers
- 5.4.3 Academic and Research Institutes
- 5.5 By Geography
- 5.5.1 North America
- 5.5.1.1 United States
- 5.5.1.2 Canada
- 5.5.1.3 Mexico
- 5.5.2 Europe
- 5.5.2.1 Germany
- 5.5.2.2 United Kingdom
- 5.5.2.3 France
- 5.5.2.4 Italy
- 5.5.2.5 Spain
- 5.5.2.6 Rest of Europe
- 5.5.3 Asia-Pacific
- 5.5.3.1 China
- 5.5.3.2 Japan
- 5.5.3.3 India
- 5.5.3.4 Australia
- 5.5.3.5 South Korea
- 5.5.3.6 Rest of Asia-Pacific
- 5.5.4 Middle East & Africa
- 5.5.4.1 GCC
- 5.5.4.2 South Africa
- 5.5.4.3 Rest of Middle East & Africa
- 5.5.5 South America
- 5.5.5.1 Brazil
- 5.5.5.2 Argentina
- 5.5.5.3 Rest of South America
- 5.5.1 North America
6 Competitive Landscape
- 6.1 Market Concentration
- 6.2 Market Share Analysis
- 6.3 Company Profiles (includes Global level Overview, Market level overview, Core Segments, Financials as available, Strategic Information, Market Rank/Share for key companies, Products & Services, and Recent Developments)
- 6.3.1 Novartis AG
- 6.3.2 Gilead Sciences Inc.
- 6.3.3 F. Hoffmann-La Roche Ltd
- 6.3.4 bluebird bio Inc.
- 6.3.5 UniQure N.V.
- 6.3.6 Amgen Inc.
- 6.3.7 Biogen Inc.
- 6.3.8 Astellas Pharma Inc.
- 6.3.9 Sarepta Therapeutics Inc.
- 6.3.10 Regenxbio Inc.
- 6.3.11 Krystal Biotech Inc.
- 6.3.12 Orchard Therapeutics plc
- 6.3.13 Rocket Pharmaceuticals Inc.
- 6.3.14 Editas Medicine Inc.
- 6.3.15 Intellia Therapeutics Inc.
- 6.3.16 Sangamo Therapeutics Inc.
- 6.3.17 CSL Behring
- 6.3.18 Mustang Bio Inc.
- 6.3.19 Poseida Therapeutics Inc.
- 6.3.20 Generation Bio Co.
7 Market Opportunities & Future Outlook
- 7.1 White-space & Unmet-Need Assessment




