PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1273403

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1273403
Monoclonal Antibody Therapeutics Market - Growth, Trends, and Forecasts (2023 - 2028)
The monoclonal antibody therapeutics market studied was anticipated to grow with a CAGR of nearly 13.2% during the forecast period.
COVID-19 had a significant impact on the monoclonal antibody therapeutics market owing to the factors such as increased utilization of monoclonal antibodies to treat the virus during the pandemic. For instance, as per the February 2022 update, the FDA issued an emergency use authorization (EUA) for a new monoclonal antibody for the treatment of COVID-19 that retains activity against the omicron variant. The emergency use authorization (EUA) for bebtelovimab was for the treatment of mild to moderate COVID-19 in adults and pediatric patients. Furthermore, as per the article published in January 2022 in JBS journal, monoclonal antibodies (mAbs), which had great specificity and dependability, had become effective instruments for diagnosing and treating a wide range of disorders. Many scientists have started working immediately on developing antibody-based assays for the detection of SARS-CoV-2 and Ab medications for use as COVID-19 treatment agents. Thus, the monoclonal antibody therapeutics market is anticipated to have a significant impact in the coming years due to the increase in the prevalence of chronic disease and the rise in the development of monoclonal antibody therapeutics.
The major factors attributing to the growth of the market are a rise in the global prevalence of autoimmune disorders such as rheumatoid arthritis, inflammatory bowel disorder, Crohn's disease, and cancers, and an increased geriatric population causing a global burden. For instance, as per the American Cancer Society 2022 update, the estimated number of new cancer cases in 2022 is found to be 1.9 million. Monoclonal antibodies are a promising class of targeted anticancer agents that improve the functions of the body's immune system to inhibit the growth and spread of cancer cells. Thus increase in cancer cases is anticipated to increase the monoclonal antibody therapeutics, thereby boosting the market growth over the forecast period.
A few more factors play a pivotal role in taking the market to the next level, such as the approval of new products in recent years and a strong pipeline expected to fuel the monoclonal antibody therapeutics across the forecast period. Moreover, the increased purchase of monoclonal antibodies in various diseases in bulk is fuelling market growth. For instance, as per the February 2022 update, United States Department of Health and Human Services (HHS) Secretary Xavier Becerra reported a purchase of 600,000 treatment courses of a new monoclonal antibody treatment that data showed works against the Omicron variant. Such initiatives in the field of monoclonal antibodies are major factors fuelling the market growth. Moreover, the research and development and strong pipeline studies in the field of monoclonal antibodies are uplifting the market growth. For instance, as per the June 2022 update, Genentech presented positive data from a broad blood cancer portfolio at European Hematology Association Annual Meeting 2022. After eight years of follow-up, the Phase III GALLIUM study's final analysis revealed that patients with previously untreated follicular lymphoma who received Gazyva (obinutuzumab) with chemotherapy continued to see a considerable improvement in progression-free survival.
Hence, due to the rise in autoimmune disease, and the increase in product launches in monoclonal antibody therapeutics, the monoclonal antibody market is likely to witness growth over the forecast period. However, the high risk of complications such as heart failure, liver injury, neurological disorders, psoriasis, and many allergic reactions, high challenges while in the development stage, and expensive patented products restrain the market growth.
Monoclonal Antibody Therapeutics Market Trends
Autoimmune Diseases are Expected to Hold the Significant Market Share in the Monoclonal Antibody Therapeutics Market
Autoimmune diseases are anticipated to witness growth in the market due to the global prevalence of various diseases such as rheumatoid arthritis, psoriatic arthritis, Crohn's disease, uveitis, and juvenile idiopathic arthritis. For instance, as per the October 2021 CDC update, by 2040, an estimated 78 million (26%) United States adults aged 18 years or older are projected to have doctor-diagnosed arthritis.
Also, the availability of products such as Humira, a fully human monoclonal antibody approved by the US FDA for the treatment of rheumatoid arthritis fueling the market. In August 2021, FDA approved the use of a new version of a HUMIRA biosimilar known as HADLIMA to treat certain autoimmune disorders. The increased research based on the advancement of monoclonal antibodies in autoimmune disease is an uplifting growth factor in the market. For instance, as per the article published in February 2022 in Technology Networks, using an anti-CD6 monoclonal antibody to block the CD6-ALCAM pathway in models of systemic lupus erythematosus (SLE) and lupus nephritis (LN) resulted in prolonged survival. Furthermore, as per the August 2022 update from Penn Medicine, an experimental drug called litifilimab (known as BIIB059) is likely to be a powerful long-term treatment for cutaneous lupus erythematosus (CLE), a form of lupus that occurs in the skin and can severely impact the quality of life. This kind of clinical study is fuelling the growth of the segment and propelling the global monoclonal antibody therapeutics market revenue in the forecast period.
Hence, due to the rise in autoimmune disease, and the increase in product launches in monoclonal antibody therapeutics for autoimmune disease, the autoimmune segment in the monoclonal antibody market is likely to witness growth over the forecast period.
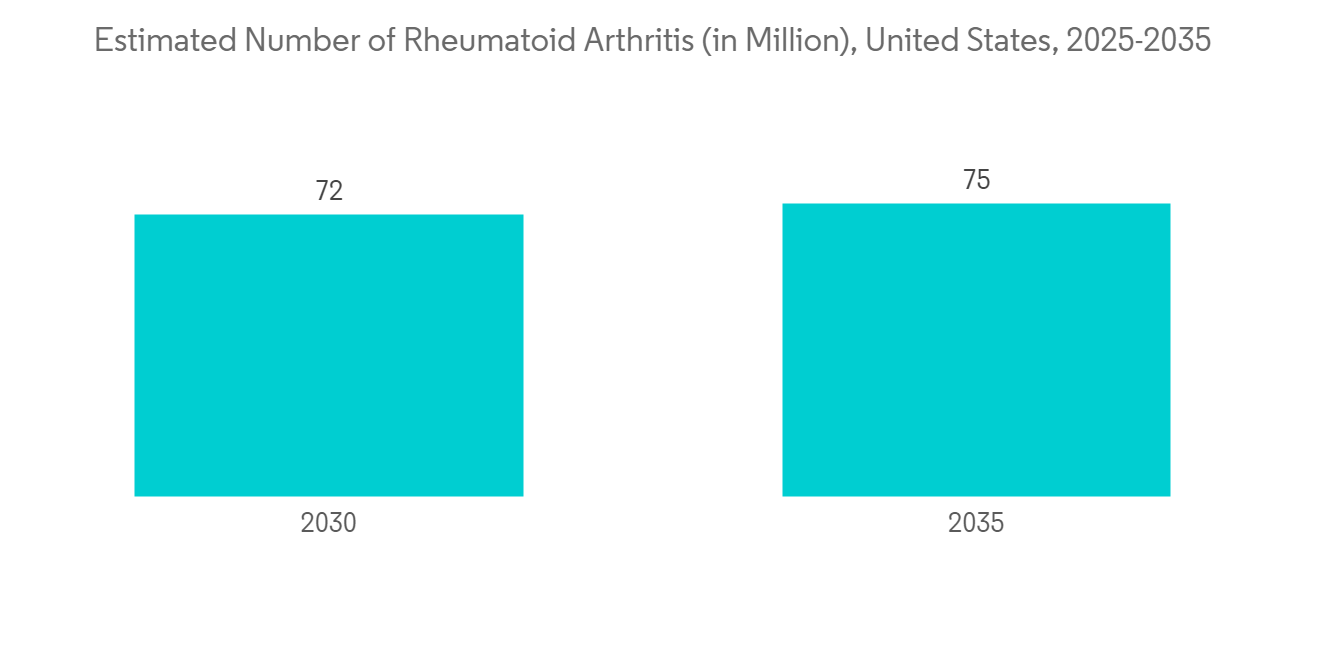
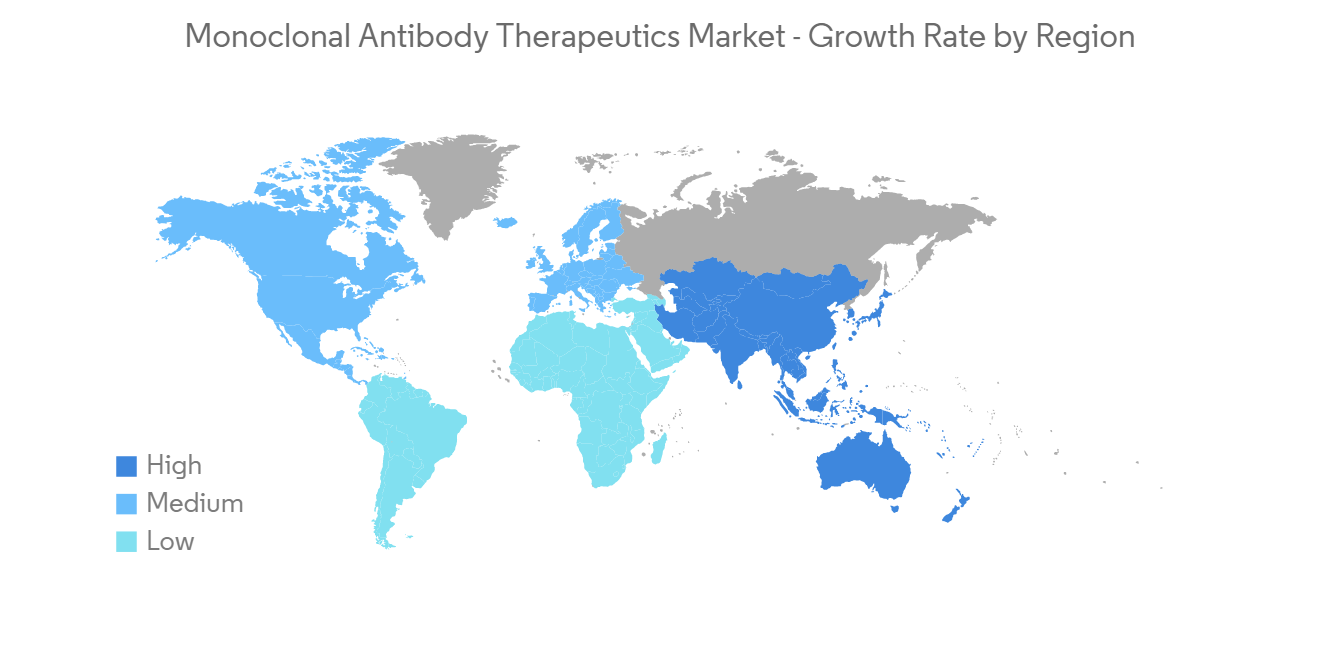
North America is Anticipated to Witness a Growth in the Monoclonal Antibody Therapeutics Market Over the Forecast Period
North America is expected to hold a significant share in the overall monoclonal antibody therapeutics market throughout the forecast period owing to the factors such as a rise in autoimmune diseases, increased healthcare expenditure, huge investments by the key players, and continuous growth in R&D activities with the rise in application of these therapeutics in the treatment of various disorders such as (PCOS) polycystic ovary syndrome, breast cancers in the United States. For instance, as per Breast Cancer.Org, in 2022, an estimated 287,850 new cases of invasive breast cancer are expected to be diagnosed in women in the United States., along with 51,400 new cases of non-invasive (in situ) breast cancer.
Furthermore, the presence of competitors and their product launches relating to the area of monoclonal antibodies are a major fuelling factor for the market growth. For instance, as per the August 2022 update, AstraZeneca and Daiichi Sankyo's Enhertu (trastuzumab deruxtecan) was approved in the United States for the treatment of adult patients with unresectable or metastatic HER2-low (IHC 1+ or IHC 2+/ISH-) breast cancer who have received prior chemotherapy in the metastatic setting or developed disease recurrence during or within six months of completing adjuvant chemotherapy. Moreover, collaborations relating to the research and development of monoclonal antibodies in different cancer treatment is likely to boost the market growth. For instance, as per the March 2022 update, Sanofi and Blackstone agreed to a strategic, risk-sharing collaboration under which funds managed by Blackstone Life Sciences (BXLS) that is likely to contribute up to EUR 300 million to accelerate the pivotal global studies and the clinical development program for the subcutaneous formulation and delivery of the anti-CD38 antibody Sarclisa, to treat patients with multiple myeloma (MM). If successful, BXLS is anticipated to be eligible to receive royalties on future subcutaneous sales. The pivotal study for the subcutaneous formulation is expected to begin in the second half of 2022.
Hence, due to the rise in chronic diseases and autoimmune diseases, and the increase in product launches in monoclonal antibody therapeutics, strategic partnerships involving the expansion of monoclonal antibody development, North America is anticipated to witness growth in the monoclonal antibody market over the forecast period.
Monoclonal Antibody Therapeutics Industry Overview
The monoclonal antibody therapeutics market is moderately competitive and consists of several major players. In terms of market share, a few of the major players are currently dominating the market. Some prominent players are acquiring other companies to consolidate their market positions across the globe and while others are launching new products. Some of the companies which are currently dominating the market are Abbvie Inc, Daiichi Sankyo Company Limited, Johnson & Johnson, Amgen Inc, and UCB S.A., Belgium (UCB Inc).
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope of the Study
2 RESEARCH METHODOLOGY
3 EXECUTIVE SUMMARY
4 MARKET DYNAMICS
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Increasing Global Prevalence of Autoimmune Disorders and Rise in Geriatric Population
- 4.2.2 New Product Launches with Accelerated Drug Approval Initiative by USFDA
- 4.3 Market Restraints
- 4.3.1 High Risk of Complications such as Heart Failure, Liver Injury, Neurological Disorders, Psoriasis and many Allergic Reactions
- 4.3.2 High Challenges while in the Development Stage and Expensive Patented Products
- 4.4 Porter Five Forces
- 4.4.1 Threat of New Entrants
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Bargaining Power of Suppliers
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5 MARKET SEGMENTATION (Market Size by Value - USD Million)
- 5.1 By Application
- 5.1.1 Hematological Diseases
- 5.1.2 Autoimmune Diseases
- 5.1.3 Cancer
- 5.1.4 Others
- 5.2 By Source
- 5.2.1 Human
- 5.2.2 Humanized
- 5.2.3 Chimeric
- 5.2.4 Others
- 5.3 By End-user
- 5.3.1 Hospitals
- 5.3.2 Private Clinics
- 5.3.3 Others
- 5.4 Geography
- 5.4.1 North America
- 5.4.1.1 United States
- 5.4.1.2 Canada
- 5.4.1.3 Mexico
- 5.4.2 Europe
- 5.4.2.1 Germany
- 5.4.2.2 United Kingdom
- 5.4.2.3 France
- 5.4.2.4 Italy
- 5.4.2.5 Spain
- 5.4.2.6 Rest of Europe
- 5.4.3 Asia-Pacific
- 5.4.3.1 China
- 5.4.3.2 Japan
- 5.4.3.3 India
- 5.4.3.4 Australia
- 5.4.3.5 South Korea
- 5.4.3.6 Rest of Asia-Pacific
- 5.4.4 Middle East and Africa
- 5.4.4.1 GCC
- 5.4.4.2 South Africa
- 5.4.4.3 Rest of Middle East and Africa
- 5.4.5 South America
- 5.4.5.1 Brazil
- 5.4.5.2 Argentina
- 5.4.5.3 Rest of South America
- 5.4.1 North America
6 COMPETITIVE LANDSCAPE
- 6.1 Company Profiles
- 6.1.1 UCB S.A., Belgium (UCB Inc)
- 6.1.2 Pfizer Inc
- 6.1.3 Amgen Inc
- 6.1.4 AbbVie Inc
- 6.1.5 Johnson & Johnson
- 6.1.6 F. Hoffmann-La Roche Ltd
- 6.1.7 Boehringer Ingelheim International GmbH
- 6.1.8 Novartis AG
- 6.1.9 Merck KGaA
- 6.1.10 Daiichi Sankyo Company Limited
7 MARKET OPPORTUNITIES AND FUTURE TRENDS




