PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1444669

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1444669
Precision Medicine - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts (2024 - 2029)
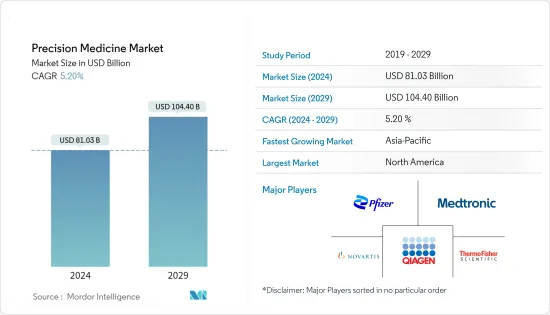
The Precision Medicine Market size is estimated at USD 81.03 billion in 2024, and is expected to reach USD 104.40 billion by 2029, growing at a CAGR of 5.20% during the forecast period (2024-2029).
The COVID-19 pandemic affected the market for precision medicine. For instance, an article published in the JAMA Network in May 2021 reported precision medicine approaches to emphasize more precise diagnosis and treatment based on a range of biomarkers, including genetic variants and data about patients' environment, lifestyle, and behaviors, which are useful in understanding variations in individuals' susceptibility and responses to COVID-19. Thus, the COVID-19 pandemic sparked a surge in demand for precision medicine. However, in the current scenario, it is anticipated that with the emergence of other SARS-CoV-2 strains and the presence of other diseases, the demand for precision medicine may keep on increasing over the forecast period.
The factors that are driving the growth of the studied market are increasing online collaborative forums, increased efforts to characterize genes, and advancements in cancer biology. In the past few years, several gene therapies, diagnostics, and medicines have been introduced in this field due to the rising incidence of life-threatening medical conditions, such as cancer and rare diseases. For instance, according to the Globocan 2021 fact sheet, an estimated 21.8 million new cancer cases are expected to be reported by 2025. The increasing incidence of cancer cases is expected to drive the need for precision medicine for the effective treatment of patients and thus contribute to the growth of the studied market.
Furthermore, technological advancements and advances in cell biology are fueling the growth of the market under consideration.For instance, in May 2021, AdventHealth expanded its genomics program with the launch of a data-driven precision medicine initiative that will structure and integrate longitudinal patient data with molecular testing data, creating a dataset and disease network models to help the health system better predict the development of diseases and responses to treatments. Thus, such advancements are driving the growth of the studied market.
Moreover, the creation of online forums for specialized areas like oncology and neurology will also boost the market. For instance, there are online forums planned for May 2022 in New Jersey, United States, and for November 2022 in Berlin, Germany, which will provide a platform for healthcare professionals, industry, patient groups, researchers, payers, and governments to share their experiences and vision for a future of improved cancer patient outcomes. Thus, such forums demand the development of precision medicine for the growing number of cancer cases in the country, thus driving the growth of the studied market.
Innovation and the availability of new technology are other key factors for the market's growth. For instance, in November 2021, the Cleveland Clinic expanded its advanced genomic testing platform as the standard of care for patients with cancer. This expanded capability to test tumor samples involves whole-exome sequencing, which examines all the portions of DNA in the genes responsible for making proteins in the body, and RNA sequencing, which assesses how these proteins are encoded. With advancements in precision therapies and diagnostics, coupled with breakthrough innovations such as Next Generation Sequencing (NGS) and big genomic data, the industry is expected to witness rapid growth during the forecast period.
Thus, due to the increasing number of online collaborative forums, increasing efforts to characterize genes, and advancements in cancer biology, the market is expected to witness significant growth over the forecast period. However, the perils of data sharing, the shift from treatment-based to preventive healthcare, and declining trends in the FDA pharmacotherapy approval rate may slow down the growth of the studied market.
Precision Medicine Market Trends
Oncology Segment is Expected to Witness a Significant Growth Over the Forecast Period.
The oncology segment is expected to witness significant growth over the forecast period owing to increasing cases of oncology and technological developments. Precision medicine applications are primarily directed toward better treatment of oncological diseases and are expected to witness significant growth over the forecast period. The key factor behind the significant growth of the segment is the rising prevalence of cancer and the rising number of drug candidates for the same in clinical trials.
The increasing use of precision oncology by patients with cancer is driving the growth of this segment. For instance, in April 2022, an article published in the journal Nature Medicine reported that the increasing use of genomic profiling for diagnosis and therapy guidance in many tumor types has increased the use of precision medicine in cancer patients. According to the article, precision medicine is used for people who are at a higher risk of developing certain cancers, particularly those who have a family history of cancer.Thus, the increasing efficiency of precision medicine and the increasing number of cancer cases are driving the growth of this segment.
Similarly, the collaborations between key market players are another factor propelling the market's growth. For instance, in September 2021, AstraZeneca and Thermo Fisher Scientific entered a multi-year strategic collaboration to co-develop next-generation sequencing (NGS)-based companion diagnostics (CDx). AstraZeneca said that more than 90% of its clinical pipeline contains targeted precision therapies, including oncology.
The high support from the government through funding and the rapid growth of genomic analysis are expected to augment the growth of the precision medicine market at a fast rate over the forecast period.
North America is Expected to Witness a Significant Growth Over the Forecast Period.
The presence of better healthcare infrastructure and huge expenditures on R&D are expected to boost the growth of the precision medicine market in the United States. For certain types of cancer treatments, like colorectal cancer and breast cancer, there have been significant advances in precision medicine.
The increasing number of cancer cases in North American countries like Canada is driving the growth of the studied market. For instance, in June 2022, Canada's Cancer Society reported that 233,900 people in Canada were expected to be diagnosed with cancer by the end of 2022. The source also reported that the most commonly diagnosed cancers are expected to be lung, breast, prostate, and colorectal cancers. Thus, increasing cancer cases are increasing the demand for oncology precision medicine and thus driving the growth of the studied market in the region.
In a similar way, the National Cancer Institute (NIC), based on data from March 2022, was given USD 6.9 billion by the Consolidated Appropriations Act, 2022. This is a net increase of USD 353 million over FY 2021.Included in the FY 2022 allocation are USD 194 million in funding for the Cancer Moonshot and USD 50 million for the Childhood Cancer Data Initiative. Thus, increasing investments in enabling a precision medicine approach across the country are expected to drive the market studied during the forecast period.
Furthermore, the growing number of precision medicine researchers in North American countries is driving the growth of the studied market.For instance, an article published in the journal Gaceta Medica de Mexico in March 2022 reported that a research team in Mexico has developed precision medicine for RET gene mutational diagnosis for medullary thyroid cancer and multiple endocrine neoplasia (MEN) 2. The article also quoted that RET pathogenic variants in the Mexican population are consistent with reported data and that the most common mutations are 634/NEM2 and 918/NEM3, which have been reported in the Mexican population, and that the researchers are developing precision for the diagnosis and treatment of this mutation.
Furthermore, in April 2021, MCI Onehealth Technologies Inc. (MCI) entered a strategic partnership with Ariel Precision Medicine Inc. to accelerate MCI's precision medicine and technology roadmap for both clinical and commercial projects with pharmaceutical, medical device, and life sciences companies, thereby driving the market. Similarly, in March 2021, the National Institute of Health (NIH) invested in the next iteration of a public-private partnership to advance precision medicine research for Alzheimer's disease. Therefore, increasing partnerships in advancing precision medicine for different diseases is expected to lead to lucrative growth of the market in the country.
Hence, the increasing number of cancer cases and technological developments are driving the growth of the studied market in the region over the forecast period.
Precision Medicine Industry Overview
There are a lot of different companies that work both globally and locally in the precision medicine market. Some of the companies are Astarte Medical, AstraZeneca PLC, Biogen, Bristol-Myers Squibb, F Hoffmann-La Roche AG, Laboratory Corporation of America Holdings, Medtronic PLC, Myriad Genetics Inc., Novartis AG, Pfizer Inc., Qiagen NV, Quest Diagnostics, and Thermo Fisher Scientific Inc.
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope of the Study
2 RESEARCH METHODOLOGY
3 EXECUTIVE SUMMARY
4 MARKET DYNAMICS
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Increasing Online Collaborative Forums
- 4.2.2 Increasing Efforts to Characterize Genes
- 4.2.3 Advancement in Cancer Biology
- 4.3 Market Restraints
- 4.3.1 Perils of Data Sharing
- 4.3.2 Shift from Treatment-based to Preventive Healthcare
- 4.3.3 Declining Trends in FDA Pharmacotherapy Approval Rate
- 4.4 Porter's Five Forces Analysis
- 4.4.1 Threat of New Entrants
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Bargaining Power of Suppliers
- 4.4.4 Threat of Substitute Products and Services
- 4.4.5 Intensity of Competitive Rivalry
5 MARKET SEGMENTATION (Market Size by Value - USD million)
- 5.1 Technology
- 5.1.1 Big Data Analytics
- 5.1.2 Bioinformatics
- 5.1.3 Gene Sequencing
- 5.1.4 Drug Discovery
- 5.1.5 Companion Diagnostics
- 5.1.6 Other Technologies
- 5.2 Application
- 5.2.1 Oncology
- 5.2.2 CNS
- 5.2.3 Immunology
- 5.2.4 Respiratory
- 5.2.5 Other Applications
- 5.3 Geography
- 5.3.1 North America
- 5.3.1.1 United States
- 5.3.1.2 Canada
- 5.3.1.3 Mexico
- 5.3.2 Europe
- 5.3.2.1 Germany
- 5.3.2.2 United Kingdom
- 5.3.2.3 France
- 5.3.2.4 Italy
- 5.3.2.5 Spain
- 5.3.2.6 Rest of Europe
- 5.3.3 Asia-Pacific
- 5.3.3.1 China
- 5.3.3.2 Japan
- 5.3.3.3 India
- 5.3.3.4 Australia
- 5.3.3.5 South Korea
- 5.3.3.6 Rest of Asia-Pacific
- 5.3.4 Middle East and Africa
- 5.3.4.1 GCC
- 5.3.4.2 South Africa
- 5.3.4.3 Rest of Middle East and Africa
- 5.3.5 South America
- 5.3.5.1 Brazil
- 5.3.5.2 Argentina
- 5.3.5.3 Rest of South America
- 5.3.1 North America
6 COMPETITIVE LANDSCAPE
- 6.1 Company Profiles
- 6.1.1 Astarte Medical
- 6.1.2 AstraZeneca PLC
- 6.1.3 Biogen
- 6.1.4 Bristol-Myers Squibb
- 6.1.5 F Hoffmann-La Roche Ltd.
- 6.1.6 Laboratory Corporation of America Holdings
- 6.1.7 Medtronic PLC
- 6.1.8 Myriad Genetics Inc.
- 6.1.9 Novartis AG
- 6.1.10 Pfizer Inc.
- 6.1.11 Qiagen NV
- 6.1.12 Quest Diagnostics
- 6.1.13 Thermo Fisher Scientific Inc.
7 MARKET OPPORTUNITIES AND FUTURE TRENDS




