PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1850270

PUBLISHER: Mordor Intelligence | PRODUCT CODE: 1850270
Immunotherapy Drugs - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts (2025 - 2030)
Global immunotherapy drugs market revenues reach USD 156.52 billion in 2025 and are set to climb to USD 262.68 billion by 2030, locking in a 10.91% CAGR.
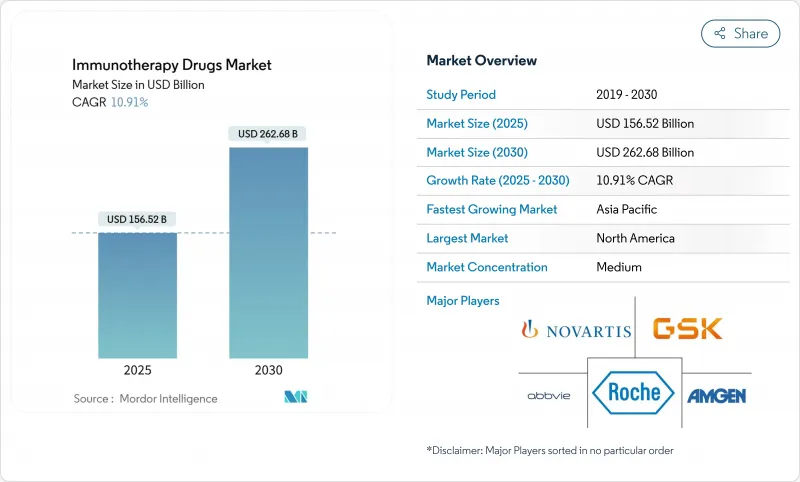
The surge originates from deeper checkpoint-inhibitor penetration, rapid maturation of T-cell engineering, and the routine use of artificial-intelligence platforms that shorten discovery cycles. Multi-modal regimens that merge monoclonal antibodies with precision biomarkers continue to dominate first-line protocols in solid tumors, while bespoke neoantigen vaccines graduate from early trials into pivotal studies in melanoma and lung cancer. Regulatory agencies simplify combination-therapy reviews, and outcomes-based payment pilots reduce upfront cost exposure for health systems. Manufacturing scale-ups in Asia-Pacific lower per-dose costs, and real-time pharmacovigilance networks improve adverse-event tracking, supporting broader geographic access for complex biologics.
Global Immunotherapy Drugs Market Trends and Insights
Explosive Pipeline of Immune-Checkpoint Inhibitors
Approvals of tislelizumab-jsgr and cosibelimab-ipdl in 2024 confirmed that PD-1, PD-L1, and CTLA-4 programs now pivot toward tumor-agnostic and rare-cancer settings, often as part of doublet or triplet regimens. Late-phase trials involving LAG-3, TIGIT, and TIM-3 candidates signal access to previously resistant populations, while adaptive trial designs shrink development timelines. Combination strategies such as nivolumab + ipilimumab secure higher complete-response rates in melanoma, underlining why regulators continue to grant breakthrough designations for synergistic protocols that deliver clear survival advantages over monotherapies.
Expanding Dominance of Monoclonal Antibodies
Iterative engineering upgrades-bispecific, trispecific, and antibody-drug conjugate (ADC) formats-preserve the 77.55% revenue grip held by antibodies. Teclistamab delivers a 63% overall response rate in late-line multiple myeloma, and Pfizer's AI-guided ADC collaboration compresses lead-optimization windows from months to days. CD3-engaging bispecifics redirect T-cell cytotoxicity against solid-tumor antigens that remain out of reach for current CAR-T delivery mechanisms.
High Treatment Costs & Reimbursement Hurdles
CAR-T price tags ranging from USD 373,000 to USD 4.25 million strain payer budgets; in response, CMS proposes a 17% base-payment increase and pilots outcomes-based contracts that refund non-responders. Medicaid's CGT Access Model ties reimbursement to real-world durability metrics, yet emerging economies still struggle to afford premium biologics despite high unmet needs.
Other drivers and restraints analyzed in the detailed report include:
- Rising Incidence of Cancer & Chronic Diseases
- Biomarker-Driven Precision-Medicine Adoption
- Manufacturing Complexity of Biologics Supply Chains
For complete list of drivers and restraints, kindly check the Table Of Contents.
Segment Analysis
The immunotherapy drugs market continues to be dominated by monoclonal antibodies, yet T-cell therapies represent the fastest-rising pillar with an 18.25% CAGR through 2030. The immunotherapy drugs market size allocated to T-cell therapies is forecast to expand from USD 15.2 billion in 2025 to USD 35.3 billion by 2030, underscoring their transformative impact on relapsed multiple myeloma and aggressive lymphomas. FDA approvals of idecabtagene vicleucel and ciltacabtagene autoleucel produce minimal-residual-disease negativity rates above 70%, while dual-target CAR constructs broaden tumor-antigen coverage. Checkpoint inhibitors gather steady share via combination approvals, and the pipeline for mRNA cancer vaccines accelerates on the back of breakthrough-therapy designations. Oncolytic viruses such as LOAd703 post 44% response rates when paired with atezolizumab, while IL-15 super-agonists like Anktiva secure approvals for lymphopenic settings, signaling gains for cytokine-based products within the broader immunotherapy drugs market.
Progress in generative chemistry compresses ADC payload discovery, raising potency without parallel toxicity. The immunotherapy drugs market thus transitions from standalone biologics to integrated, multimodal regimens that pair cell therapies with oncolytic platforms and targeted cytokines. Manufacturing automation reduces vein-to-vein time for autologous constructs, suggesting that living products will secure larger slices of future immunotherapy drugs market share once cost curves decline.
The Immunotherapy Drugs Market is Segmented by Drug Type (Monoclonal Antibodies, Cancer Vaccines, Interferons Alpha and Beta, Interleukins, and More), Therapy Area (Cancer, Autoimmune and Inflammatory Diseases, Infectious Diseases, and More), End-User (Hospitals & Clinics, Research Laboratories, and More), and Geography (North America, Europe, Asia-Pacific, and More). The Market Forecasts are Provided in Terms of Value (USD).
Geography Analysis
North America keeps a 48.72% revenue stake through 2025, anchored by a mature reimbursement framework, dense innovation clusters, and early adoption of outcomes-based contracting. The region's biopharma incumbents acquire AI start-ups and manufacturing specialists, consolidating platform control while reducing batch-failure rates. Strategic onshoring incentives lower supply-chain exposure to geopolitical shocks, strengthening long-term competitiveness for the regional immunotherapy drugs market.
Asia-Pacific records a 14.22% CAGR, the fastest worldwide, powered by regulatory harmonization, lower labor costs, and government-backed capacity expansions. China's approval of Stapokibart, the first domestic IL-4Ra antagonist, reflects higher local regulatory sophistication and shortens launch lags versus Western benchmarks. Japan and South Korea refine conditional early-approval pathways, and India's biomanufacturing corridors lure Western sponsors seeking cost-effective production slots, collectively lifting regional share in the global immunotherapy drugs market.
Europe demonstrates steady mid-single-digit growth under the Advanced Therapy Medicinal Products regulation that standardizes cell- and gene-therapy review. Biosimilar competition tempers monoclonal-antibody pricing but widens access, keeping overall market value intact. Targeted digital-health initiatives link registries with pharmacovigilance portals, streamlining post-approval monitoring and reinforcing public confidence in immunotherapy safety profiles.
The Middle East and Africa gain momentum from flagship projects such as Saudi Arabia's Cancer BioShield platform, which delivers immune-restorative regimens tailored to local genetic backgrounds. Multinational partnerships with regional sovereign funds finance precision oncology centers that function as hubs for nearby emerging markets. Latin America focuses on licensing ready-to-fill formulations and implementing risk-sharing reimbursement, gradually lifting therapy uptake in high-burden cancers like gastric carcinoma. Together, these advancements broaden the global footprint of the immunotherapy drugs market and reduce dependency on a handful of mature territories.
- Abbvie
- Amgen
- AstraZeneca
- Bristol-Myers Squibb
- Roche
- GlaxoSmithKline
- Johnson & Johnson
- Merck
- Novartis
- Pfizer
- Bayer
- Sanofi
- Boehringer Ingelheim
- Gilead Sciences
- UbiVac
- Regeneron Pharmaceuticals
- Eli Lilly and Company
- Seagen
- BeiGene Ltd.
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 Introduction
- 1.1 Study Assumptions & Market Definition
- 1.2 Scope of the Study
2 Research Methodology
3 Executive Summary
4 Market Landscape
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Explosive Pipeline of Immune-Checkpoint Inhibitors
- 4.2.2 Expanding Dominance of Monoclonal Antibodies
- 4.2.3 Rising Incidence of Cancer & Chronic Diseases
- 4.2.4 Biomarker-Driven Precision-Medicine Adoption
- 4.2.5 Bispecific Antibodies & Adcs Unlocking New Indications
- 4.2.6 AI-Enabled In-Silico Discovery Accelerating R&D
- 4.3 Market Restraints
- 4.3.1 High Treatment Costs & Reimbursement Hurdles
- 4.3.2 Manufacturing Complexity Of Biologics Supply Chains
- 4.3.3 Immune-Related Adverse-Event Management Burden
- 4.3.4 Price Erosion From Biosimilar Competition
- 4.4 Porter's Five Forces
- 4.4.1 Threat of New Entrants
- 4.4.2 Bargaining Power of Buyers
- 4.4.3 Bargaining Power of Suppliers
- 4.4.4 Threat of Substitutes
- 4.4.5 Intensity of Competitive Rivalry
5 Market Size & Growth Forecasts (Value, USD)
- 5.1 By Drug Type
- 5.1.1 Monoclonal Antibodies
- 5.1.2 Checkpoint Inhibitors
- 5.1.3 Cancer Vaccines
- 5.1.4 Interferons Alpha/Beta
- 5.1.5 Interleukins
- 5.1.6 Oncolytic Viral Therapies
- 5.1.7 T-Cell Therapies (CAR-T, TCR, TIL)
- 5.1.8 Cytokines & Immunomodulators
- 5.1.9 Other Drug Types
- 5.2 By Therapy Area
- 5.2.1 Cancer
- 5.2.2 Autoimmune & Inflammatory Diseases
- 5.2.3 Infectious Diseases
- 5.2.4 Other Therapy Areas
- 5.3 By End-User
- 5.3.1 Hospitals & Clinics
- 5.3.2 Research Laboratories & Academic Institutes
- 5.3.3 Pharmaceutical & Biotechnology Companies
- 5.4 Geography
- 5.4.1 North America
- 5.4.1.1 United States
- 5.4.1.2 Canada
- 5.4.1.3 Mexico
- 5.4.2 Europe
- 5.4.2.1 Germany
- 5.4.2.2 United Kingdom
- 5.4.2.3 France
- 5.4.2.4 Italy
- 5.4.2.5 Spain
- 5.4.2.6 Rest of Europe
- 5.4.3 Asia-Pacific
- 5.4.3.1 China
- 5.4.3.2 Japan
- 5.4.3.3 India
- 5.4.3.4 South Korea
- 5.4.3.5 Australia
- 5.4.3.6 Rest of Asia-Pacific
- 5.4.4 Middle East and Africa
- 5.4.4.1 GCC
- 5.4.4.2 South Africa
- 5.4.4.3 Rest of Middle East and Africa
- 5.4.5 South America
- 5.4.5.1 Brazil
- 5.4.5.2 Argentina
- 5.4.5.3 Rest of South America
- 5.4.1 North America
6 Competitive Landscape
- 6.1 Market Concentration
- 6.2 Market Share Analysis
- 6.3 Company Profiles (includes Global level Overview, Market level overview, Core Segments, Financials as available, Strategic Information, Market Rank/Share for key companies, Products & Services, and Recent Developments)
- 6.3.1 AbbVie Inc.
- 6.3.2 Amgen Inc.
- 6.3.3 AstraZeneca PLC
- 6.3.4 Bristol-Myers Squibb
- 6.3.5 F. Hoffmann-La Roche AG
- 6.3.6 GSK PLC
- 6.3.7 Johnson & Johnson
- 6.3.8 Merck & Co. Inc.
- 6.3.9 Novartis AG
- 6.3.10 Pfizer Inc.
- 6.3.11 Bayer AG
- 6.3.12 Sanofi
- 6.3.13 Boehringer Ingelheim
- 6.3.14 Gilead Sciences Inc.
- 6.3.15 UbiVac
- 6.3.16 Regeneron Pharmaceuticals
- 6.3.17 Eli Lilly & Co.
- 6.3.18 Seagen Inc.
- 6.3.19 BeiGene Ltd.
7 Market Opportunities & Future Outlook
- 7.1 White-Space & Unmet-Need Assessment




